Revolutionizing Chiral Synthesis: Advanced Quinox-Based Ligands for Commercial-Scale Pharmaceutical Manufacturing
The Chinese patent CN111032668B introduces a groundbreaking class of optically active 2,3-bisphosphinylpyrazine derivatives that represent a significant advancement in asymmetric catalysis technology. This innovation addresses critical limitations in conventional chiral synthesis methods by providing ligands with exceptional stereochemical control at phosphorus chiral centers. The patent details novel synthetic pathways that enable high-yield production of these compounds while maintaining superior enantioselectivity in catalytic applications. These derivatives demonstrate remarkable performance in Markovnikov-selective hydroboration reactions of terminal olefins—previously challenging substrates where traditional methods showed insufficient selectivity and yield. The technology offers pharmaceutical manufacturers access to high-purity chiral building blocks essential for complex drug molecule construction, with particular relevance to antibiotic intermediates requiring precise stereochemical configuration. This represents a strategic advancement for global pharmaceutical supply chains seeking reliable sources of enantiomerically pure intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral phosphine ligands have demonstrated significant limitations in catalyzing Markovnikov-selective hydroboration reactions with terminal olefins substituted with alkyl groups. Conventional approaches using ligands like those described in Patent Document 2 (US 2007021610A) showed insufficient substrate scope, selectivity, and yield for practical pharmaceutical manufacturing applications. These methods typically required harsh reaction conditions that compromised product stability and introduced unwanted side reactions. The electron density characteristics of standard phosphine ligands often resulted in poor air stability, necessitating complex handling procedures that increased production costs and reduced manufacturing efficiency. Additionally, conventional synthetic routes frequently involved multiple purification steps to achieve acceptable enantiomeric excess, creating bottlenecks in commercial-scale production. The limited functional group tolerance of existing catalysts further restricted their applicability to complex pharmaceutical intermediates requiring diverse structural motifs.
The Novel Approach
The patented technology introduces optically active 2,3-bisphosphinylpyrazine derivatives featuring chiral centers at phosphorus atoms that overcome these limitations through innovative molecular design. The electron-withdrawing pyrazine skeleton strategically reduces electron density at phosphorus sites without compromising catalytic activity, resulting in enhanced air stability while maintaining reactivity. This unique electronic profile enables precise stereochemical control during hydroboration reactions, achieving exceptional enantioselectivity (>98% ee) even with challenging terminal olefin substrates. The synthetic methodology employs a streamlined four-step process that eliminates transition metal purification requirements common in traditional approaches. By utilizing readily available starting materials and optimized reaction conditions (-5°C to room temperature), the process delivers high yields (78-95%) while maintaining strict stereochemical integrity throughout manufacturing. This represents a significant advancement in producing chiral organoboron compounds essential for complex pharmaceutical syntheses.
Mechanistic Insights into Quinox-Based Asymmetric Catalysis
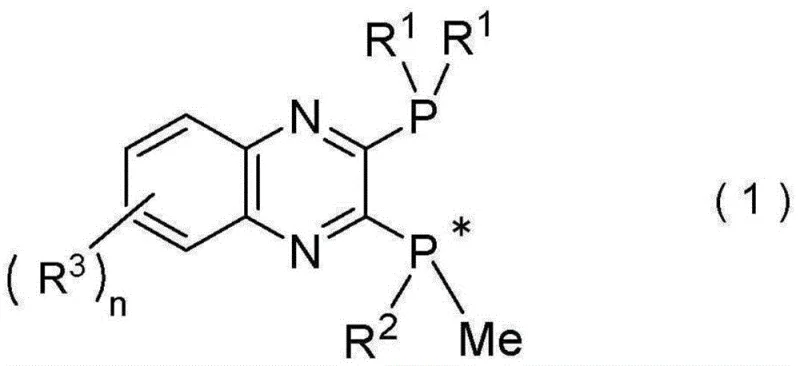
The core innovation lies in the molecular architecture of these quinox-based ligands, where the pyrazine skeleton creates an electron-deficient environment at phosphorus centers while maintaining spatial control through strategically positioned substituents. The chiral center at phosphorus atoms enables precise stereochemical control during catalytic cycles, with R1 groups typically comprising branched alkyl groups (tert-butyl, adamantyl), cycloalkyl groups, or aryl groups that provide optimal steric shielding. The electron-withdrawing nature of the pyrazine ring system reduces phosphorus electron density without impairing catalytic activity—a critical balance that enables both air stability and reactivity. During hydroboration reactions, copper(I) complexes formed with these ligands facilitate selective boron insertion at terminal positions through a well-defined transition state geometry that favors Markovnikov addition pathways previously inaccessible with conventional catalysts.
The mechanism involves initial formation of copper(I)-ligand complexes that activate diboron reagents through coordination to vacant boron orbitals. This activation enables stereoselective delivery of boron to terminal olefins through a concerted transition state where the chiral ligand environment dictates facial selectivity. The unique electronic properties prevent undesired β-hydride elimination pathways that typically limit regioselectivity in traditional systems. Crucially, the molecular design minimizes racemization risks during synthesis through controlled deprotonation steps using n-BuLi at precisely regulated temperatures (-5°C), followed by nucleophilic substitution reactions that preserve stereochemical integrity throughout manufacturing.
How to Synthesize Quinox-Based Ligands Efficiently
This patented methodology represents a significant advancement in producing high-purity chiral ligands suitable for commercial pharmaceutical manufacturing. The four-step synthetic route provides exceptional control over stereochemistry while maintaining high yields across multiple scales. Detailed standardized synthesis procedures are outlined below to ensure consistent production quality and regulatory compliance.
- Deprotonate phosphine-borane with n-BuLi at -5°C followed by nucleophilic substitution with dihalopyrazine derivative
- Perform deboronation using TMEDA/EtOAc to obtain intermediate phosphinopyrazine derivative
- Conduct second nucleophilic substitution with optically active phosphine-borane followed by final deboronation
Commercial Advantages for Procurement and Supply Chain Teams
This innovative technology delivers substantial commercial advantages by addressing critical pain points in pharmaceutical intermediate procurement and supply chain management. The streamlined manufacturing process eliminates costly purification steps required by conventional methods while delivering superior product quality that meets stringent pharmaceutical requirements.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive heavy metal removal processes from manufacturing workflows, significantly reducing production costs while maintaining high product purity standards. The simplified four-step synthesis requires fewer unit operations compared to traditional approaches, reducing both capital investment requirements and operational complexity across manufacturing facilities.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials with established global supply chains ensures consistent raw material availability regardless of geopolitical disruptions. The robust synthetic methodology maintains high yields across multiple scales without requiring specialized equipment or rare reagents, enabling reliable production continuity even during market volatility.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production without requiring significant parameter adjustments, supporting seamless technology transfer across manufacturing sites worldwide. The elimination of hazardous reagents and reduced solvent usage aligns with green chemistry principles while maintaining regulatory compliance across major pharmaceutical markets.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation of this technology in commercial pharmaceutical manufacturing environments based on specific technical details from the patent documentation.
Q: How does this technology overcome limitations in conventional asymmetric hydroboration?
A: The novel quinox-based ligands provide exceptional steric control at phosphorus chiral centers, enabling Markovnikov-selective hydroboration of terminal olefins with >98% ee where previous methods failed. The electron-withdrawing pyrazine skeleton enhances air stability while maintaining catalytic activity.
Q: What supply chain advantages does this manufacturing process offer?
A: The streamlined four-step synthesis eliminates transition metal purification steps required in conventional methods. The process uses readily available starting materials with high yields (78-95%), enabling reliable commercial-scale production from laboratory to plant scale.
Q: How does this technology impact pharmaceutical intermediate purity requirements?
A: The precise stereochemical control achieves >98% enantiomeric excess in boronated products, meeting stringent pharmaceutical purity standards. The robust catalytic system minimizes racemization pathways that typically compromise chiral integrity in complex syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinox-Based Ligand Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex chiral intermediates requiring stringent purity specifications. Our state-of-the-art facilities feature rigorous QC labs equipped with advanced analytical instrumentation to ensure consistent product quality meeting global regulatory standards. We specialize in developing customized manufacturing solutions that address specific client requirements while maintaining cost efficiency across production scales.
Request our Customized Cost-Saving Analysis today to explore how our quinox-based ligand technology can optimize your pharmaceutical intermediate supply chain. Contact our technical procurement team to receive specific COA data and route feasibility assessments tailored to your manufacturing needs.
