Advanced Catalyst-Free Synthesis of Substituted Cyclic Acid Anhydrides for Commercial Scale-Up
Advanced Catalyst-Free Synthesis of Substituted Cyclic Acid Anhydrides for Commercial Scale-Up
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes that balance high purity with environmental sustainability. Patent CN101519407B introduces a transformative methodology for the synthesis of substituted cyclic acid anhydrides, a critical class of intermediates used in the production of complex heterocycles including Biotin (Vitamin H) derivatives. This technology replaces hazardous acidic dehydration agents with a clean, azeotropic distillation process, achieving yields exceeding 99% and purity levels greater than 99.5%. For R&D directors and procurement specialists, this represents a significant leap forward in process safety and cost efficiency, eliminating the need for corrosive reagents while simplifying downstream purification.
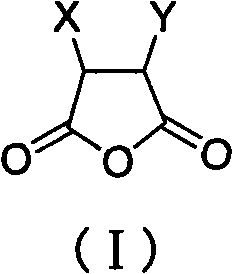
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the cyclization of dicarboxylic acids to form anhydrides has relied heavily on aggressive dehydrating agents such as acetic anhydride or acid chlorides. Prior art, including USP 2489232 and various academic studies, indicates that these traditional pathways often require stoichiometric excesses of reagents, leading to molar ratios as high as 1:3. This not only inflates raw material costs but generates substantial quantities of acidic byproducts like acetic acid and acetate mixtures, which are notoriously difficult to separate from the target product. Furthermore, the corrosive nature of these reaction media necessitates the use of specialized, expensive corrosion-resistant equipment, thereby increasing capital expenditure and maintenance overheads for manufacturing facilities.
The Novel Approach
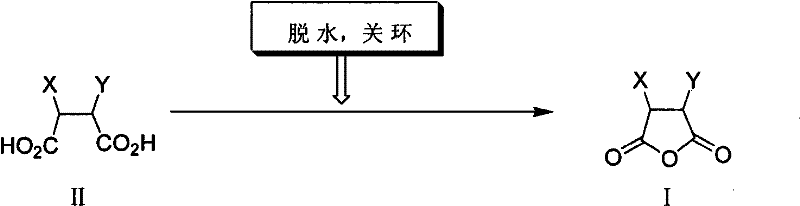
In stark contrast, the method disclosed in CN101519407B utilizes a physical-chemical approach driven by thermodynamics rather than harsh chemical activation. By employing high-boiling organic solvents such as toluene or xylene, the process facilitates the continuous removal of water via azeotropic distillation. This shifts the equilibrium entirely towards the formation of the cyclic anhydride without the addition of any acidic catalyst. The result is a remarkably clean reaction profile where the product precipitates directly upon cooling, allowing for simple filtration. This eliminates the complex aqueous workups and neutralization steps typical of older methods, drastically reducing waste generation and operational complexity.
Mechanistic Insights into Thermal Azeotropic Cyclization
The core mechanism of this synthesis relies on the intramolecular nucleophilic attack of one carboxylic acid group onto the carbonyl carbon of the adjacent carboxylic acid group, facilitated by thermal energy. In the absence of an acid catalyst, the reaction rate is governed by the efficient removal of the water byproduct. The use of solvents like toluene, which form low-boiling azeotropes with water, ensures that water is physically removed from the reaction zone as it is formed. This prevents the reverse hydrolysis reaction, driving the conversion to completion. The structural integrity of sensitive substituents (X and Y groups), such as the imidazoline rings found in Biotin precursors, is preserved because the reaction environment remains neutral throughout the process.
From an impurity control perspective, this catalyst-free approach offers superior selectivity. Traditional acid-catalyzed dehydrations often promote side reactions such as decarboxylation or polymerization, especially at elevated temperatures. By avoiding strong mineral acids or Lewis acids, the novel method minimizes the formation of colored impurities and degradation products. The patent data confirms that the crude product obtained after filtration possesses a purity of >99.5%, indicating that the crystallization from the reaction solvent acts as an effective self-purification step. This high level of chemical fidelity is crucial for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals are enforced.
How to Synthesize Substituted Cyclic Acid Anhydride Efficiently
The operational simplicity of this route makes it highly attractive for process chemists aiming to transfer technology from the lab to the pilot plant. The procedure involves charging the dicarboxylic acid precursor and the solvent into a reactor equipped with a Dean-Stark trap or similar water separator. The mixture is heated to reflux, typically between 80°C and 120°C, and maintained until theoretical water evolution is observed. Detailed standard operating procedures and specific stoichiometric ratios for various substrates are outlined in the structured guide below.
- Charge the reactor with the substituted dicarboxylic acid starting material and a high-boiling organic solvent such as toluene or xylene in a weight ratio of approximately 1: 10 to 1:25.
- Heat the reaction mixture to reflux temperatures between 80°C and 120°C to initiate azeotropic distillation, continuously removing water formed during the cyclization.
- Maintain reflux for 6 to 12 hours until dehydration is complete, then cool the mixture to room temperature to precipitate the pure cyclic acid anhydride product for filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible economic and logistical benefits. The elimination of corrosive reagents reduces the dependency on specialized alloy reactors, allowing for the use of standard glass-lined or stainless steel equipment which is more readily available and cheaper to maintain. Furthermore, the ability to recover and recycle the high-boiling solvent significantly lowers the overall solvent consumption per kilogram of product, directly impacting the variable cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The removal of expensive dehydrating agents like acetic anhydride and the associated neutralization bases creates a leaner bill of materials. Additionally, the high yield (>99%) means less raw material is wasted, and the simplified purification process reduces labor and utility costs associated with distillation or chromatography. The qualitative reduction in waste disposal fees, due to the absence of acidic aqueous streams, further enhances the economic viability of the process.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis—substituted dicarboxylic acids and common solvents like toluene—are commodity chemicals with stable global supply chains. Unlike specialized catalysts that may have long lead times or single-source risks, the inputs for this process are widely available. This ensures consistent production scheduling and reduces the risk of supply disruptions for critical intermediates like those used in Vitamin synthesis.
- Scalability and Environmental Compliance: The process aligns perfectly with green chemistry principles by minimizing E-factors (mass of waste per mass of product). The lack of corrosive waste streams simplifies wastewater treatment compliance, a growing concern for manufacturers facing stricter environmental regulations. The mild reaction conditions and robust nature of the chemistry make it inherently scalable from kilogram to multi-ton production without significant re-engineering of the process parameters.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a reliable basis for feasibility assessments.
Q: What are the primary advantages of this azeotropic dehydration method over traditional acetic anhydride methods?
A: Unlike traditional methods requiring acetic anhydride or acid chlorides which generate corrosive acidic waste and require complex separation, this method uses simple thermal dehydration with solvent recovery, resulting in >99.5% purity and significantly reduced environmental impact.
Q: Can this process be scaled for industrial production of Biotin intermediates?
A: Yes, the process operates under mild conditions (80-120°C) without corrosive catalysts, making it highly suitable for large-scale stainless steel reactors and ideal for the commercial production of key intermediates like 1,3-dibenzyl imidazoline-2-ketone derivatives.
Q: What solvents are compatible with this synthesis route?
A: The patent specifies the use of high-boiling aromatic hydrocarbons such as toluene, ethylbenzene, or xylenes, as well as ethers like dioxane, which facilitate effective azeotropic water removal and can be easily recycled.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Acid Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that technologies like the one described in CN101519407B are translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cyclic acid anhydride meets the exacting standards required for API synthesis.
We invite potential partners to contact our technical procurement team to discuss how this catalyst-free technology can optimize your supply chain. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your project, and ask for specific COA data and route feasibility assessments tailored to your target molecules.
