Advanced Synthesis of Thiadiazole Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as key building blocks for novel therapeutic agents. Patent CN113767094A discloses a significant advancement in the synthesis of 3-methyl-1,2,4-thiadiazole-5-carbohydrazide and its methyl-d3 deuterated analog, which are critical intermediates for the production of Fezolinetant, a selective neurokinin-3 (NK-3) receptor antagonist. This technology addresses long-standing challenges in the manufacturing of these compounds, specifically regarding safety, yield, and isotopic purity. By shifting away from hazardous reagents utilized in prior art, such as chlorocarbonyl sulfinyl chloride, the disclosed method offers a safer and more efficient pathway suitable for commercial scale-up. The innovation lies in the strategic application of palladium-catalyzed alkoxycarbonylation, a transformation that has historically been difficult to achieve on less robust heterocycles like the 1,2,4-thiadiazole ring due to susceptibility to ring opening.
For procurement managers and supply chain directors, the implications of this technological shift are profound. The ability to source high-purity intermediates without the baggage of dangerous chemical handling translates directly into reduced operational risk and potential cost optimization. As a reliable pharmaceutical intermediate supplier, understanding these underlying process improvements is essential for evaluating long-term supply continuity. The patent demonstrates that through careful catalyst selection and reaction condition optimization, it is possible to achieve overall yields exceeding 67%, a substantial improvement over the less than 30% yields reported in previous methodologies. This efficiency gain is not merely a laboratory curiosity but a fundamental driver for industrial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-methyl-1,2,4-thiadiazole-5-carbohydrazide has relied on pathways that involve highly reactive and hazardous reagents. Specifically, prior art methods, such as those described in WO2013/050424, utilize chlorocarbonyl sulfinyl chloride and methyl cyanoformate to construct the thiadiazole core. These reagents present severe raw material problems for large-scale production, including toxicity, corrosivity, and difficult waste disposal requirements. Furthermore, these conventional routes are prone to generating sulfur-containing impurities that can persist through downstream processing, adversely affecting the quality and safety profile of the final drug product. Even with extensive optimization efforts, the overall yield of the intermediate via these traditional methods remains stubbornly low, typically falling below 30%. This inefficiency results in significant material loss, increased production costs, and a larger environmental footprint, making the conventional approach suboptimal for modern green chemistry standards and cost-sensitive manufacturing environments.
The Novel Approach
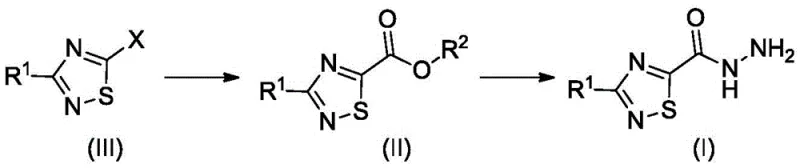
The methodology presented in CN113767094A represents a paradigm shift by employing a modular strategy centered on alkoxycarbonylation. Instead of constructing the ring from scratch using dangerous precursors, the new process starts with a pre-formed halogenated thiadiazole intermediate. This halide is then subjected to a palladium-catalyzed carbonylation reaction in the presence of carbon monoxide and an alcohol solvent to generate the corresponding ester. This ester is subsequently converted to the target carbohydrazide via hydrazinolysis. This approach completely bypasses the need for chlorocarbonyl sulfinyl chloride, thereby eliminating the associated safety hazards. Moreover, the process has been demonstrated to be robust even upon large-scale up, maintaining satisfactory yields. The versatility of this route is further highlighted by its applicability to both non-deuterated and deuterated forms, ensuring that the supply chain for isotopically labeled drugs can also benefit from these safety and efficiency improvements.
Mechanistic Insights into Palladium-Catalyzed Alkoxycarbonylation
The core of this technological breakthrough is the successful application of alkoxycarbonylation on the 1,2,4-thiadiazole ring, a transformation previously considered challenging due to the ring's instability. The reaction typically proceeds using a palladium catalyst, such as palladium acetate or bis(triphenylphosphine)palladium(II) chloride, in combination with an organophosphorus ligand like Xantphos. The mechanism involves the oxidative addition of the palladium catalyst to the carbon-halogen bond of the thiadiazole intermediate, followed by the insertion of carbon monoxide to form an acyl-palladium complex. Subsequent nucleophilic attack by the alcohol solvent releases the ester product and regenerates the active catalyst species. Crucially, the selection of ligands and reaction conditions, such as temperature (typically 50°C to 150°C) and pressure (1 to 20 bar CO), is optimized to prevent ring-opening side reactions that would lead to mercaptoamidine by-products. This precise control over the catalytic cycle ensures high selectivity for the desired ester formation.
Impurity control is another critical aspect addressed by this mechanistic understanding. In conventional syntheses, sulfur impurities are a major concern. The new route minimizes these by avoiding sulfur-introducing reagents in the final steps and utilizing purification techniques like distillation or crystallization that are compatible with the ester intermediate. For the deuterated variants, the process begins with d3-acetonitrile, which is converted to a Pinner salt and then to d3-acetamidine. This amidine is cyclized to the deuterated amine using bromine or hypochlorite routes, preserving the isotopic label with high fidelity (>95% purity). The subsequent Sandmeyer reaction converts this amine to the bromo-intermediate without scrambling the deuterium atoms, ensuring that the final deuterated drug substance meets strict regulatory specifications for isotopic enrichment.
How to Synthesize 3-Methyl-1,2,4-Thiadiazole-5-Carbohydrazide Efficiently
The synthesis of this valuable intermediate is streamlined into a logical sequence that prioritizes safety and yield. The process begins with the preparation of the halogenated precursor via a Sandmeyer reaction, followed by the key carbonylation step, and concludes with hydrazide formation. Detailed operational parameters, including specific catalyst loadings, solvent choices like ethanol or methyl tert-butyl ether, and temperature profiles, are critical for success. The following guide outlines the standardized synthesis steps derived from the patent examples, providing a roadmap for R&D teams looking to implement this technology.
- Perform a Sandmeyer reaction on 3-methyl-1,2,4-thiadiazol-5-amine using sodium nitrite and hydrogen bromide to generate the corresponding bromo-intermediate.
- Subject the bromo-intermediate to alkoxycarbonylation using carbon monoxide, a palladium catalyst (e.g., Pd(OAc)2), Xantphos ligand, and ethanol to form the ethyl ester.
- React the resulting ethyl ester with hydrazine monohydrate in isopropanol at controlled low temperatures to yield the final carbohydrazide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond simple chemical yield. The elimination of hazardous reagents like chlorocarbonyl sulfinyl chloride significantly reduces the regulatory burden and safety infrastructure costs associated with manufacturing. Facilities no longer need to invest in specialized containment systems for these dangerous chemicals, leading to substantial cost savings in capital expenditure and operational maintenance. Furthermore, the robustness of the palladium-catalyzed step ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. This reliability is paramount for maintaining a steady supply of critical intermediates for global pharmaceutical clients.
- Cost Reduction in Manufacturing: The new process achieves significantly higher overall yields compared to conventional methods, which directly translates to better material utilization and lower cost of goods sold. By avoiding expensive and hazardous reagents, the raw material costs are optimized, and the waste treatment costs are drastically simplified. The ability to use standard palladium catalysts and common solvents like ethanol further enhances the economic viability of the process, making it a superior choice for cost-sensitive large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 3-methyl-1,2,4-thiadiazol-5-amine and carbon monoxide, ensures a secure supply chain. Unlike specialized reagents that may face availability issues, the inputs for this process are commodity chemicals with robust global supply networks. This stability reduces lead times and mitigates the risk of supply disruptions, allowing manufacturers to commit to long-term supply agreements with greater confidence.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable, with examples showing successful execution on multi-kilogram scales. The avoidance of sulfur impurities and hazardous by-products simplifies the environmental compliance landscape, reducing the complexity of waste stream management. This aligns with modern green chemistry principles and helps pharmaceutical companies meet their sustainability goals, making the intermediate more attractive to environmentally conscious buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 3-methyl-1,2,4-thiadiazole-5-carbohydrazide. These answers are derived directly from the technical disclosures in patent CN113767094A, providing clarity on the process capabilities and limitations.
Q: What are the safety advantages of the new synthesis route compared to conventional methods?
A: The novel process eliminates the use of hazardous reagents such as chlorocarbonyl sulfinyl chloride and methyl cyanoformate, which pose significant raw material handling risks in large-scale production. Additionally, it avoids the generation of sulfur impurities that can adversely affect final drug quality.
Q: How does this patent address the challenge of synthesizing deuterated intermediates?
A: The patent provides specific routes for synthesizing the methyl-d3 deuterated form starting from d3-acetonitrile. It utilizes Pinner reactions or hydroxylamine routes to form deuterated amidines, ensuring high isotopic purity (>95%) before cyclization to the thiadiazole ring.
Q: What catalysts are employed in the key alkoxycarbonylation step?
A: The process utilizes palladium catalysts such as palladium acetate or bis(triphenylphosphine)palladium(II) chloride. These are often used in conjunction with organophosphorus ligands like Xantphos or triphenylphosphine to facilitate the conversion of the halogenated intermediate to the ester.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-1,2,4-Thiadiazole-5-Carbohydrazide Supplier
The synthesis route detailed in CN113767094A represents a significant leap forward in the manufacturing of thiadiazole intermediates, offering a blend of safety, efficiency, and scalability that is essential for modern pharmaceutical production. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this advanced chemistry can be seamlessly transferred to an industrial setting. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of intermediate meets the highest standards required for API synthesis. We understand the critical nature of these building blocks in the development of NK-3 receptor antagonists and are dedicated to supporting our partners with reliable supply.
We invite procurement teams and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging this improved synthetic route, we can help you optimize your supply chain and reduce overall manufacturing costs. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to bring safer and more efficient pharmaceutical solutions to the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
