Advanced Synthetic Strategy For 2-Bromo-6-Fluorobenzoic Acid: Technical Breakthroughs And Commercial Scalability
The pharmaceutical industry continuously demands robust, scalable, and cost-effective pathways for complex intermediates, particularly those serving as key building blocks for kinase inhibitors and antiviral agents. Patent CN102795993A, published on November 28, 2012, introduces a technically sophisticated preparation method for 2-bromo-6-fluorobenzoic acid, a critical scaffold in modern medicinal chemistry. This document outlines a transformative synthetic strategy that departs from conventional cryogenic methodologies, utilizing o-fluorobenzonitrile as a commercially accessible starting material. The process integrates nitration, nitro-reduction, bromination, and a combined diazo-deamination hydrolysis sequence, achieving a final purity of 98.8%. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediates supplier options, this patent represents a significant leap forward in process chemistry, offering a viable alternative to resource-intensive legacy routes while maintaining stringent quality standards required for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromo-6-fluorobenzoic acid has relied heavily on organometallic strategies that impose severe operational burdens on manufacturing facilities. Traditional literature, such as the work by DiMichele et al., describes a Grignard approach involving 1,2-dibromo-6-fluorobenzene reacted with isopropylmagnesium chloride. This methodology necessitates cryogenic conditions, typically around -40°C, to manage the exothermic nature of the halogen-metal exchange and prevent decomposition. Furthermore, alternative routes utilizing lithium diisopropylamide (LDA) require even more extreme temperatures, often dropping to -78°C, to achieve the necessary lithiation before carboxylation with dry ice. These low-temperature requirements demand specialized refrigeration equipment, significantly increasing capital expenditure and energy consumption. Additionally, the handling of pyrophoric reagents and moisture-sensitive organometallic intermediates introduces substantial safety risks and complicates waste management protocols, making these routes less attractive for large-scale commercial production where safety and cost efficiency are paramount concerns for supply chain heads.
The Novel Approach
In stark contrast, the methodology disclosed in CN102795993A leverages a classical yet highly optimized electrophilic aromatic substitution strategy that operates under significantly milder conditions. By initiating the synthesis with o-fluorobenzonitrile, the process bypasses the need for cryogenic organometallic chemistry entirely. The nitration step is conducted at temperatures ranging from -5°C to 20°C, which can be achieved using standard chilled water or brine systems rather than expensive glycol loops. The subsequent reduction utilizes iron powder and ammonium chloride, a classic Béchamp reduction variant that is inherently safer and generates less hazardous waste compared to catalytic hydrogenation or hydride reductions. This shift from exotic, high-energy reagents to commodity chemicals drastically simplifies the engineering requirements for the reactor setup. The integration of deamination and hydrolysis into a single pot operation further streamlines the workflow, eliminating isolation steps and reducing solvent consumption. This holistic approach not only enhances the safety profile but also aligns perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing by minimizing utility usage and maximizing throughput.
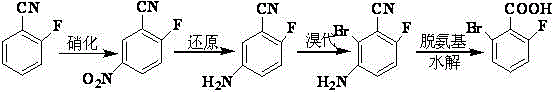
Mechanistic Insights into Electrophilic Substitution and Diazotization
The core of this synthetic route lies in the precise control of regioselectivity during the initial functionalization of the aromatic ring. The nitration of o-fluorobenzonitrile is an electrophilic aromatic substitution where the nitrile group acts as a meta-director and the fluorine atom acts as an ortho/para director. However, due to the strong deactivating nature of the nitrile group and the specific reaction conditions employing potassium nitrate in concentrated sulfuric acid, the nitro group is selectively introduced at the 5-position relative to the nitrile, yielding 2-fluoro-5-nitrobenzonitrile. The patent specifies a molar ratio of sulfuric acid to substrate between 5.0:1 and 15.0:1, with an optimal ratio of 11.3:1, ensuring sufficient protonation of the nitrile to direct the incoming electrophile correctly. Following this, the reduction of the nitro group to an amine using iron powder proceeds via a single electron transfer mechanism. The presence of ammonium chloride serves to maintain the acidity of the medium, preventing the precipitation of iron oxides and ensuring the continuous availability of the reducing surface. This step achieves conversion rates approaching 100%, demonstrating the robustness of the redox chemistry involved.
The subsequent bromination step utilizes N-bromosuccinimide (NBS) as a mild brominating agent, which reacts with the electron-rich aniline derivative formed in the previous step. The amino group is a powerful activator, directing the bromine atom to the ortho position relative to itself, which corresponds to the 3-position of the benzonitrile ring, resulting in 2-bromo-3-amino-6-fluorobenzonitrile. The use of NBS avoids the generation of hydrobromic acid byproducts associated with elemental bromine, simplifying the workup procedure. The final transformation involves the formation of a diazonium salt using sodium nitrite and sulfuric acid at 0 to 5°C. This unstable intermediate is then subjected to a reductive deamination in the presence of copper sulfate and sodium hypophosphite. Crucially, the acidic and heated conditions (50 to 60°C) simultaneously facilitate the hydrolysis of the nitrile group to the carboxylic acid. This tandem deamination-hydrolysis mechanism is a masterstroke of process efficiency, as it accomplishes two distinct bond-breaking and bond-forming events in a single operational unit, thereby reducing the overall processing time and minimizing the potential for impurity accumulation between steps.
How to Synthesize 2-Bromo-6-Fluorobenzoic Acid Efficiently
The execution of this synthesis requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent data. The process begins with the controlled addition of nitrating agents to the sulfonated substrate, followed by a heterogeneous reduction that requires efficient stirring to maintain contact between the solid iron powder and the liquid phase. The bromination step must be kept cool to prevent poly-bromination, while the final diazotization requires precise pH and temperature control to stabilize the diazonium species before its decomposition. For detailed operational parameters, including specific addition rates, stirring speeds, and quenching protocols, operators should refer to the standardized technical documentation provided below. Adhering to these validated parameters ensures the consistent production of high-purity material suitable for downstream coupling reactions in API synthesis.
- Perform nitration of o-fluorobenzonitrile using potassium nitrate and concentrated sulfuric acid at -5 to 20°C to obtain 2-fluoro-5-nitrobenzonitrile.
- Reduce the nitro group using iron powder and ammonium chloride in water at 75 to 102°C to yield 2-fluoro-5-aminobenzonitrile.
- Conduct bromination using N-bromosuccinimide (NBS) in ethanol at 0 to 5°C to form 2-bromo-3-amino-6-fluorobenzonitrile.
- Execute diazotization with sodium nitrite followed by deamination and hydrolysis using sodium hypophosphite and copper sulfate to finalize 2-bromo-6-fluorobenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthetic route offers profound advantages for procurement managers tasked with optimizing the cost of goods sold (COGS) for complex intermediates. The primary driver of value is the substitution of expensive, specialty starting materials with o-fluorobenzonitrile, a commodity chemical available from multiple global suppliers, which mitigates supply risk and enhances negotiating leverage. Furthermore, the elimination of cryogenic operations removes a significant portion of the energy burden associated with manufacturing, leading to substantial cost savings in utility expenditures. The use of iron powder for reduction, rather than precious metal catalysts like palladium or platinum, not only reduces raw material costs but also eliminates the need for expensive metal scavenging steps to meet strict residual metal specifications in pharmaceutical products. This simplification of the purification train directly translates to higher overall equipment effectiveness (OEE) and faster batch cycle times.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive reagents such as potassium nitrate, iron powder, and N-bromosuccinimide, which are significantly cheaper than the organolithium or Grignard reagents required in alternative routes. By avoiding the need for specialized low-temperature reactors capable of reaching -78°C, manufacturers can utilize standard glass-lined or stainless steel vessels, drastically lowering capital depreciation costs. Additionally, the combined deamination and hydrolysis step reduces the number of isolation and purification stages, which minimizes solvent loss and labor hours. These cumulative efficiencies result in a markedly lower production cost per kilogram, allowing for more competitive pricing structures in the global market without compromising margin integrity.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available raw materials that are not subject to the same geopolitical or logistical constraints as specialty organometallic reagents. The robustness of the reaction conditions, particularly the tolerance of the reduction and bromination steps to minor variations in temperature, ensures high batch-to-batch consistency and reduces the likelihood of failed batches that could disrupt inventory levels. The high conversion rates reported, reaching nearly 100% in the reduction step, mean that raw material utilization is maximized, reducing the volume of waste that needs to be managed and disposed of. This reliability makes the process ideal for long-term supply agreements where consistent delivery schedules are critical for the downstream production of active pharmaceutical ingredients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous pyrophoric reagents and the use of aqueous workups in several steps. The generation of iron sludge from the reduction step is a well-understood waste stream that can be managed through established treatment protocols, unlike the complex organic waste generated by organometallic quenches. The process design inherently supports green chemistry principles by maximizing atom economy in the bromination step and reducing solvent intensity through the telescoped final step. For facilities operating under strict environmental regulations, this route offers a compliant pathway to manufacture high-purity pharmaceutical intermediates, reducing the regulatory burden associated with permitting and emissions monitoring.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on critical process parameters and quality attributes. Understanding these nuances is essential for process engineers and quality assurance teams when validating the method for commercial use.
Q: What are the critical temperature controls for the nitration step in this process?
A: The nitration reaction requires strict temperature control between -5°C and 20°C, with an optimal range of -2°C to 2°C, to ensure proper regioselectivity and prevent side reactions.
Q: How does this method improve upon traditional Grignard-based syntheses?
A: Unlike traditional methods requiring cryogenic conditions (-78°C) and expensive organolithium reagents, this route utilizes inexpensive o-fluorobenzonitrile and operates at much milder temperatures, significantly reducing operational costs.
Q: What is the reported purity and total yield of the final product?
A: The patent reports a final product purity of 98.8% with a total process yield reaching up to 16.9%, achieved through high conversion rates in each individual step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-6-Fluorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. Our facilities are equipped with advanced reactor systems capable of precise temperature control and safe handling of reactive intermediates, supported by rigorous QC labs that enforce stringent purity specifications. We are committed to delivering 2-bromo-6-fluorobenzoic acid that meets the highest standards of quality, with impurity profiles meticulously controlled to support your regulatory filings and clinical trials.
We invite you to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain resilience and cost structure. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project's unique requirements, ensuring a seamless integration of this critical intermediate into your manufacturing pipeline.
