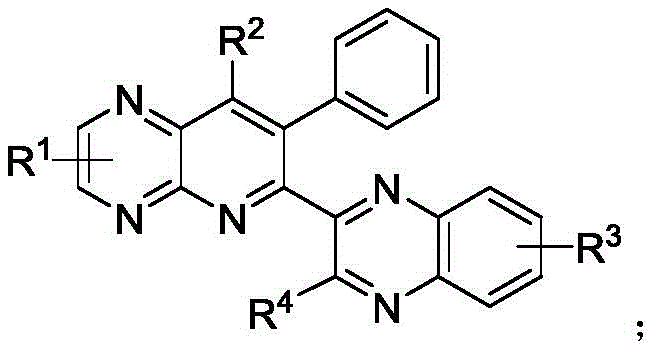
Advanced One-Pot Synthesis of Quinoxalinyl Pyridopyrazine for Commercial Antibacterial Drug Production
Advanced One-Pot Synthesis of Quinoxalinyl Pyridopyrazine for Commercial Antibacterial Drug Production
The pharmaceutical industry is constantly seeking robust synthetic routes for novel heterocyclic scaffolds that exhibit potent biological activity, particularly in the realm of antibacterial agents. Patent CN111892597A discloses a groundbreaking methodology for the preparation of quinoxalinyl pyridopyrazine compounds, which serve as critical intermediates in the development of next-generation therapeutics targeting resistant bacterial strains. This technical insight report analyzes the proprietary one-pot synthesis strategy detailed in the patent, highlighting its potential to revolutionize the manufacturing of high-purity pharmaceutical intermediates. The disclosed method leverages a ruthenium-catalyzed cascade reaction to construct complex polycyclic frameworks with remarkable efficiency and substrate universality. For R&D directors and procurement specialists, understanding the nuances of this chemistry is essential for evaluating supply chain reliability and cost-effectiveness in antibacterial drug production. The ability to synthesize these compounds directly from readily available alkynones and diamines represents a significant leap forward in process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing fused pyrazine and quinoxaline systems often suffer from significant inefficiencies that hinder commercial viability. Conventional methods typically require multi-step sequences involving the isolation and purification of unstable intermediates, which leads to substantial material loss and increased operational costs. These stepwise approaches frequently demand harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents, which complicate safety protocols and waste management in a manufacturing setting. Furthermore, the accumulation of impurities at each isolation step necessitates rigorous and costly purification processes to meet the stringent purity specifications required for pharmaceutical applications. The reliance on precious metal catalysts that are difficult to remove from the final product also poses a major challenge for regulatory compliance and cost reduction in pharmaceutical intermediate manufacturing. Consequently, there is a pressing need for a more streamlined approach that mitigates these bottlenecks while maintaining high yield and selectivity.
The Novel Approach
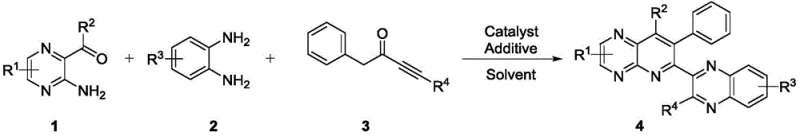
The methodology described in patent CN111892597A introduces a transformative one-pot synthesis strategy that effectively bypasses the drawbacks of traditional multi-step procedures. This novel approach utilizes a RuCl3-catalyzed system in the presence of N-bromosuccinimide (NBS) as an additive to facilitate the direct condensation of amino pyrazinyl ketones, phenylenediamines, and alkynones. By integrating the formation of the pyridopyrazine core and the quinoxaline moiety into a single reaction vessel, the process drastically simplifies the operational workflow and reduces the overall production time. The use of acetic acid as a solvent provides a mild yet effective medium that supports the cascade cyclization without degrading sensitive functional groups. This strategy not only enhances the overall yield but also significantly improves the atom economy of the reaction, making it an attractive option for cost reduction in antibacterial drug manufacturing. The versatility of this method is further demonstrated by its tolerance to a wide range of substituents, allowing for the rapid generation of diverse compound libraries for biological screening.
Mechanistic Insights into RuCl3-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the intricate catalytic cycle driven by ruthenium trichloride, which activates the alkyne functionality for nucleophilic attack. The mechanism likely involves the coordination of the ruthenium species to the triple bond of the alkynone compound, increasing its electrophilicity and facilitating the initial cyclization with the amino pyrazinyl ketone. The presence of NBS plays a pivotal role as an oxidant or halogen source that promotes the subsequent aromatization and C-H activation steps required to close the second ring system. This dual-catalyst system ensures that the reaction proceeds smoothly under reflux conditions, avoiding the need for external oxidants that might introduce unwanted byproducts. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction parameters such as temperature, catalyst loading, and reaction time to maximize efficiency. The precise control over the catalytic cycle allows for the consistent production of the target quinoxalinyl pyridopyrazine scaffold with minimal batch-to-batch variation.
Impurity control is another critical aspect where this mechanism offers distinct advantages over conventional routes. In stepwise syntheses, intermediates are often prone to decomposition or side reactions during isolation, leading to complex impurity profiles that are difficult to purge. The one-pot nature of this RuCl3-catalyzed process minimizes the exposure of reactive intermediates to the external environment, thereby suppressing the formation of degradation products. Furthermore, the specific selectivity of the catalyst system ensures that the cyclization occurs regioselectively, preventing the formation of isomeric byproducts that could compromise the biological activity of the final drug substance. This inherent purity advantage reduces the burden on downstream purification steps, such as chromatography or recrystallization, leading to significant savings in time and resources. For supply chain managers, this translates to a more predictable and reliable production schedule with fewer delays caused by out-of-specification batches.
How to Synthesize Quinoxalinyl Pyridopyrazine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized conditions outlined in the patent data to ensure reproducibility and safety. The process begins with the precise weighing and charging of the amino pyrazinyl ketone and alkynone starting materials into a reaction vessel equipped with a reflux condenser and inert gas protection. The addition of the RuCl3 catalyst and NBS additive must be controlled carefully to initiate the reaction without causing exothermic spikes, followed by the gradual introduction of the phenylenediamine component after the initial induction period. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the high yields and purity reported in the patent examples. Proper monitoring of the reaction progress via thin-layer chromatography (TLC) is essential to determine the exact endpoint and prevent over-reaction which could lead to product degradation.
- Combine amino pyrazinyl ketone and alkynone compounds in acetic acid with RuCl3 catalyst and NBS additive, then reflux for 3-5 hours to initiate cyclization.
- Introduce phenylenediamine compound to the reaction mixture and continue stirring under reflux conditions for an additional 4-6 hours until TLC indicates completion.
- Cool the reaction to room temperature, filter through diatomite, concentrate the filtrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot synthesis technology offers substantial benefits for procurement and supply chain operations within the pharmaceutical sector. The elimination of multiple isolation steps and the use of a single solvent system significantly reduce the consumption of raw materials and utilities, directly impacting the cost of goods sold. This streamlined process also shortens the manufacturing cycle time, allowing for faster response to market demands and reducing the inventory holding costs associated with work-in-progress materials. For supply chain heads, the robustness of the reaction conditions ensures a stable supply of critical intermediates, mitigating the risk of production stoppages due to process failures. The ability to scale this chemistry from gram to kilogram quantities without significant re-optimization further enhances its value proposition for commercial partnerships.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive transition metal removal steps often required in traditional catalytic methods. Since the reaction utilizes a relatively low loading of ruthenium catalyst and avoids the use of exotic ligands, the downstream processing costs associated with metal scavenging are drastically simplified. Additionally, the high atom economy of the one-pot reaction means that a larger proportion of the starting materials are converted into the desired product, reducing waste disposal costs and raw material procurement expenses. This qualitative improvement in process efficiency translates to a more competitive pricing structure for the final pharmaceutical intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, such as phenylenediamines and alkynones, are commercially available from multiple global suppliers, ensuring a resilient supply chain. Unlike proprietary reagents that may be subject to single-source bottlenecks, these commodity chemicals can be sourced with short lead times, reducing the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream drug manufacturers.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by the use of standard reactor equipment and common solvents like acetic acid, which are easily handled in existing manufacturing facilities. The reduction in the number of unit operations also minimizes the generation of solvent waste and hazardous byproducts, aligning with increasingly stringent environmental regulations. The simplified workup procedure, involving filtration and concentration, reduces the energy consumption associated with extensive drying and purification steps. These factors collectively contribute to a more sustainable manufacturing footprint, which is a key consideration for modern pharmaceutical supply chains aiming to reduce their environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoxalinyl pyridopyrazine synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production pipelines. The responses cover critical aspects such as reaction conditions, impurity management, and scalability to ensure a comprehensive understanding of the process capabilities.
Q: What are the critical reaction conditions for the RuCl3-catalyzed synthesis?
A: The process requires acetic acid as the solvent under reflux conditions, utilizing RuCl3 as the catalyst and N-bromosuccinimide (NBS) as a crucial additive to drive the one-pot cyclization efficiently.
Q: How does this one-pot method improve impurity profiles compared to stepwise synthesis?
A: By avoiding the isolation of unstable intermediates and performing the cascade reaction in a single vessel, the method minimizes side reactions and decomposition, resulting in a cleaner crude product and higher overall purity.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the use of commercially available starting materials, standard reflux conditions, and simple workup procedures like filtration and column chromatography makes the process highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxalinyl Pyridopyrazine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing, possessing the technical expertise to translate complex patent methodologies like CN111892597A into commercial reality. Our team of experienced chemists is well-versed in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinoxalinyl pyridopyrazine intermediate meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for pharmaceutical companies seeking to secure a stable supply of critical antibacterial intermediates.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain through a Customized Cost-Saving Analysis. By leveraging our manufacturing capabilities, you can access specific COA data and route feasibility assessments tailored to your project requirements. Let us help you accelerate your drug development timeline with our reliable and efficient production solutions. Contact us today to request a quote and discover how our expertise can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
