Advanced Zinc-Catalyzed Synthesis of C3-Carbonyl Acetyl Indole Derivatives for Commercial API Production
Advanced Zinc-Catalyzed Synthesis of C3-Carbonyl Acetyl Indole Derivatives for Commercial API Production
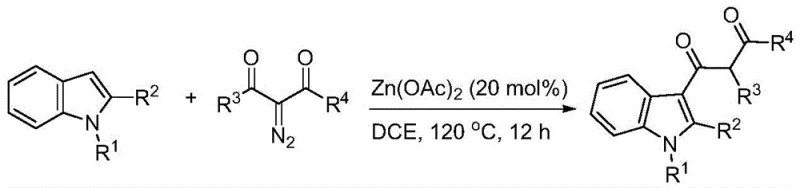
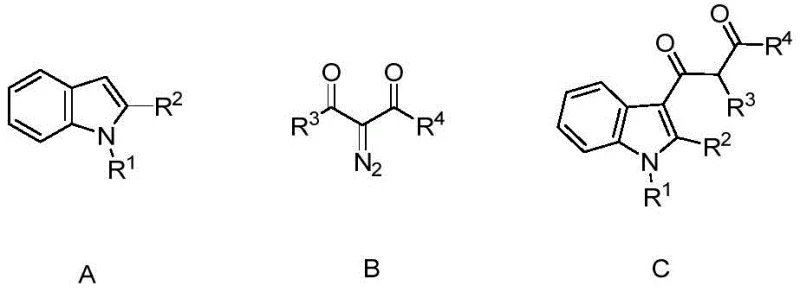
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds, particularly indole derivatives which serve as critical building blocks for numerous bioactive molecules. Patent CN109232361B introduces a transformative approach to synthesizing C3-carbonyl acetyl indole derivatives, addressing long-standing challenges in regioselectivity and operational simplicity. This technology leverages a zinc acetate-catalyzed coupling between N-substituted indoles and alpha-diazo-beta-keto esters, providing a direct route to highly functionalized products under relatively mild thermal conditions. For R&D directors and process chemists, this represents a significant leap forward in efficiency, eliminating the need for expensive transition metals like palladium or rhodium while maintaining high yields and purity profiles essential for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acylation of indoles at the C3 position has relied heavily on classical Friedel-Crafts reactions using acyl chlorides or anhydrides in the presence of strong Lewis acids. These traditional pathways often suffer from significant drawbacks, including poor regioselectivity leading to mixtures of C2 and C3 isomers that are difficult and costly to separate. Furthermore, the use of corrosive acylating agents and stoichiometric amounts of metal halides generates substantial hazardous waste, complicating environmental compliance and increasing disposal costs. Alternative methods involving organometallic reagents or palladium catalysis, while effective, introduce prohibitive costs due to the price of precious metals and the rigorous requirement for removing trace metal residues to meet pharmaceutical safety standards. These legacy processes often require multiple steps, stringent anhydrous conditions, and complex workup procedures that hinder rapid scale-up and increase the overall lead time for producing high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN109232361B utilizes a straightforward one-pot reaction strategy that dramatically simplifies the synthetic workflow. By employing alpha-diazo-beta-keto esters as the acylating source and zinc acetate as a benign, earth-abundant catalyst, the process achieves excellent C3-selectivity without the need for cryogenic temperatures or inert atmosphere gloveboxes. The reaction proceeds efficiently in 1,2-dichloroethane at 120°C, a condition that is easily manageable in standard stainless steel reactors used in commercial chemical plants. This novel approach not only streamlines the operation by reducing the number of unit operations but also enhances the safety profile by avoiding the handling of highly reactive acid chlorides. The result is a cleaner reaction profile with fewer byproducts, facilitating easier purification via standard column chromatography or crystallization, which is a critical factor for ensuring the consistent quality required by global supply chains.
Mechanistic Insights into Zinc Acetate-Catalyzed C3-Acylation
The success of this transformation lies in the unique ability of the zinc catalyst to activate the diazo compound towards nucleophilic attack by the electron-rich indole ring. Mechanistically, the zinc species likely coordinates with the carbonyl oxygen of the diazo precursor, enhancing the electrophilicity of the adjacent carbon and facilitating the loss of nitrogen gas to form a reactive metal-carbenoid or zwitterionic intermediate. This activated species is then intercepted by the C3 position of the indole, which is the most nucleophilic site, ensuring high regioselectivity. Unlike noble metal catalysts that might promote cyclopropanation or other side reactions, zinc directs the pathway specifically towards C-H insertion and subsequent acylation. This mechanistic pathway is robust against moisture and air to a significant degree, as noted in the patent data, which reduces the sensitivity of the process to minor fluctuations in reactor conditions, a key advantage for large-scale manufacturing where perfect exclusion of atmospheric elements is challenging.
From an impurity control perspective, the mechanism inherently limits the formation of poly-acylated products or C2-isomers due to the steric and electronic directing effects of the catalyst-substrate complex. The use of a slight excess of the diazo component (1:1.25 molar ratio) ensures complete consumption of the more valuable indole starting material, minimizing the presence of unreacted starting materials in the final crude mix. Furthermore, the stability of the zinc catalyst prevents the decomposition of the sensitive diazo reagent into tar-like polymers, a common issue in uncatalyzed thermal decompositions. This controlled reactivity results in a simpler impurity profile, allowing quality control teams to establish tighter specifications for the final active pharmaceutical ingredient intermediates with greater confidence and less analytical overhead.
How to Synthesize C3-Carbonyl Acetyl Indole Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and thermal parameters outlined in the patent to maximize yield and reproducibility. The process is designed to be operationally simple, utilizing standard laboratory or plant equipment without the need for specialized high-pressure or low-temperature apparatus. The following guide summarizes the critical operational phases derived from the patented examples, serving as a foundational reference for process development teams aiming to adapt this chemistry for specific target molecules. Detailed standardized synthesis steps see the guide below.
- Charge a sealed reaction vessel with N-substituted indole, alpha-diazo-beta-keto ester, and zinc acetate catalyst in 1,2-dichloroethane.
- Heat the reaction mixture to 120°C and maintain stirring for 12 hours to ensure complete conversion.
- Cool the mixture, remove solvent, and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-catalyzed technology offers compelling economic and logistical benefits that directly impact the bottom line. The shift away from precious metal catalysts to zinc acetate represents a drastic reduction in raw material costs, as zinc salts are commodity chemicals available in bulk quantities at a fraction of the price of palladium or rhodium complexes. Additionally, the simplified workup procedure reduces the consumption of solvents and silica gel during purification, further driving down the cost of goods sold. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by sensitive reagent failures or strict environmental controls, ensuring a more reliable flow of materials to downstream synthesis units.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts removes the necessity for costly metal scavenging steps, which are often required to meet strict residual metal limits in pharmaceutical products. This simplification of the purification train significantly lowers processing time and consumable usage. Furthermore, the high atom economy of the reaction, combined with the use of inexpensive and readily available diazo precursors, ensures that the overall manufacturing cost is substantially optimized compared to traditional multi-step acylation routes.
- Enhanced Supply Chain Reliability: The starting materials, including various N-substituted indoles and alpha-diazo-beta-keto esters, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The insensitivity of the reaction to air and water, as highlighted in the patent, allows for more flexible storage and handling protocols, reducing the likelihood of batch failures due to minor environmental excursions. This resilience translates to shorter lead times for high-purity pharmaceutical intermediates, enabling faster response to market demands and reducing inventory holding costs.
- Scalability and Environmental Compliance: The reaction operates at a moderate temperature of 120°C in a closed system, which is easily scalable from benchtop to multi-ton production without significant engineering hurdles. The use of 1,2-dichloroethane, while requiring proper venting, is a well-understood solvent in the industry with established recovery and recycling protocols. The absence of heavy metal waste streams simplifies wastewater treatment and aligns with increasingly stringent green chemistry regulations, positioning manufacturers as responsible partners in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential licensees or manufacturing partners. Understanding these nuances is crucial for evaluating the feasibility of integrating this route into existing production portfolios.
Q: What are the primary advantages of this zinc-catalyzed method over traditional Friedel-Crafts acylation?
A: Unlike traditional methods requiring harsh Lewis acids or sensitive acyl chlorides, this protocol utilizes stable diazo compounds and a non-toxic zinc catalyst, offering superior regioselectivity at the C3 position and simplified purification.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process operates in a closed system at moderate temperatures (120°C) using commercially available solvents and catalysts, making it highly suitable for scale-up from gram to multi-kilogram batches without complex equipment.
Q: What is the substrate scope for the indole and diazo components?
A: The method demonstrates broad tolerance, accommodating various N-substituents (Me, Ph, Bn) on the indole and diverse aryl or alkyl groups on the diazo component, including electron-withdrawing and electron-donating substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C3-Carbonyl Acetyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN109232361B for accelerating drug development timelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and risk-free. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of C3-carbonyl acetyl indole derivative meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with our technical team to explore how this zinc-catalyzed technology can be tailored to your specific project needs. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your specific supply chain. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial reliability.
