Advanced Mn-Catalyzed Synthesis of Photoinitiator UV-184 for Commercial Scale-Up
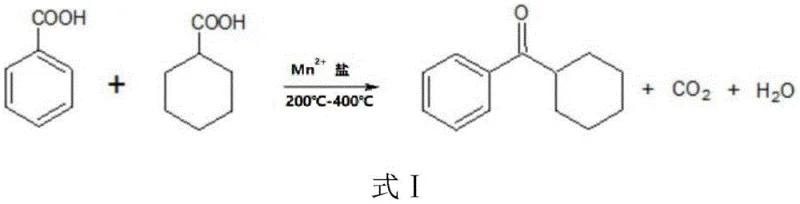
The global demand for high-performance ultraviolet curing agents continues to surge, driven by the rapid expansion of the electronics, printing, and advanced coatings sectors. Within this competitive landscape, Patent CN114835569A introduces a transformative condensation method for producing Photoinitiator UV-184, addressing critical bottlenecks in traditional manufacturing. This technical breakthrough utilizes a manganese-based catalytic system to facilitate the direct condensation of benzoic acid and cyclohexanecarboxylic acid, bypassing the hazardous and waste-intensive steps associated with legacy Friedel-Crafts acylation processes. For R&D Directors and Supply Chain Heads, this patent represents a pivotal shift towards greener, more sustainable chemical engineering, offering a pathway to significantly reduce environmental liabilities while maintaining rigorous purity standards. The methodology not only enhances reaction efficiency but also ensures long-term catalyst stability, which is essential for reliable photoinitiator supplier operations in a volatile market. By adopting this advanced synthesis route, manufacturers can achieve substantial cost savings in UV-curable coatings manufacturing through reduced waste disposal and optimized raw material utilization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 1-hydroxycyclohexyl phenyl ketone have long been plagued by severe environmental and operational inefficiencies that hinder scalable production. Conventional methods typically rely on Friedel-Crafts acylation, which necessitates the use of highly corrosive and dangerous reagents such as phosphorus trichloride, aluminum trichloride, and chlorine gas. These hazardous chemicals not only pose significant safety risks to plant personnel but also generate substantial quantities of toxic by-products, including high-phosphorus wastewater and aluminum-containing sludge. The disposal of such three wastes requires complex and expensive treatment protocols, drastically inflating the operational expenditure for any facility attempting commercial scale-up of complex photoinitiators. Furthermore, the legacy processes often suffer from inconsistent catalyst performance, requiring frequent shutdowns for catalyst replacement, which disrupts supply continuity and extends lead times for high-purity photoinitiators. The cumulative effect of these limitations is a production model that is increasingly unsustainable under modern environmental regulations and cost pressures.
The Novel Approach
In stark contrast, the innovative condensation method detailed in the patent data offers a streamlined, environmentally friendly alternative that fundamentally restructures the production workflow. By employing Mn2+ salts as a robust catalyst system, the process enables a direct one-step condensation reaction between benzoic acid and cyclohexanecarboxylic acid at elevated temperatures ranging from 200°C to 400°C. This approach effectively eliminates the need for hazardous acylation agents, thereby removing the source of phosphorus and aluminum waste streams entirely from the production cycle. The reaction mechanism facilitates the continuous removal of water and carbon dioxide as gas phases, driving the equilibrium towards the desired intermediate, cyclohexyl phenyl ketone, with exceptional efficiency. This novel approach not only simplifies the reactor design but also allows for continuous operation cycles lasting hundreds of days, ensuring a stable and predictable output for procurement managers. The result is a manufacturing process that aligns perfectly with the goals of cost reduction in electronic chemical manufacturing while adhering to strict global environmental compliance standards.
Mechanistic Insights into Mn2+-Catalyzed Condensation
The core of this technological advancement lies in the unique catalytic activity of manganese salts, which facilitate a high-temperature decarboxylative condensation mechanism. Under thermal conditions between 200°C and 400°C, the Mn2+ ions coordinate with the carboxyl groups of the reactants, lowering the activation energy required for the formation of the ketone bond. This catalytic cycle promotes the simultaneous elimination of carbon dioxide and water, which are vented from the reaction system, effectively pushing the chemical equilibrium towards the formation of cyclohexyl phenyl ketone. The stability of the manganese catalyst under these harsh thermal conditions is remarkable, allowing for prolonged reaction periods without significant degradation of catalytic activity. This mechanistic robustness is critical for maintaining consistent product quality and yield over extended production runs, a key factor for supply chain reliability. The ability to operate continuously without frequent catalyst regeneration or replacement distinguishes this method from traditional batch processes that suffer from rapid catalyst deactivation.
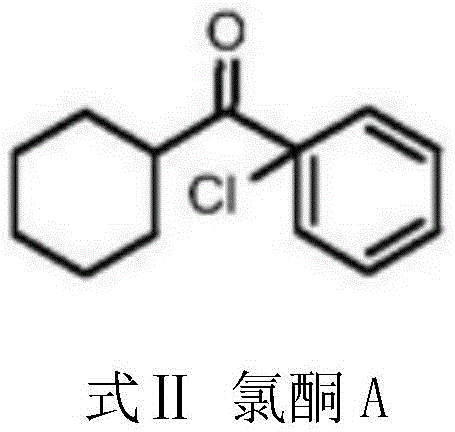
Following the initial condensation, the intermediate cyclohexyl phenyl ketone undergoes a subsequent chlorination and alkaline hydrolysis sequence to yield the final photoinitiator. The chlorination step introduces a chlorine atom at the alpha position relative to the carbonyl group, forming a reactive chloroketone intermediate that is crucial for the final structural configuration. This intermediate is then subjected to alkaline hydrolysis, where the chlorine atom is replaced by a hydroxyl group, completing the synthesis of 1-hydroxycyclohexyl phenyl ketone. The precision of this two-step conversion ensures that impurity profiles are tightly controlled, resulting in a final product with purity levels exceeding 95% after distillation. Understanding this mechanistic pathway is vital for R&D teams aiming to optimize reaction parameters for maximum yield and minimal by-product formation. The clarity of this reaction pathway provides a solid foundation for scaling the process from laboratory benchtop to multi-ton industrial reactors.
How to Synthesize Photoinitiator UV-184 Efficiently
Implementing this synthesis route requires careful attention to reaction temperatures, catalyst loading, and feed rates to ensure optimal performance and safety. The process begins with the preparation of a raw material mixture where benzoic acid and cyclohexanecarboxylic acid are combined in a specific molar ratio, typically with a slight excess of the cyclohexane derivative to drive the reaction forward. This mixture is then introduced into a reactor containing the activated manganese catalyst, where temperature control is maintained strictly within the 200°C to 400°C window to prevent thermal degradation while ensuring sufficient reaction kinetics. The dropwise addition of the raw material mixture must be carefully regulated to match the catalytic capacity, preventing accumulation of unreacted acids that could complicate downstream separation. Detailed standardized synthesis steps are essential for replicating the high yields and purity reported in the patent data, ensuring that the transition from pilot scale to full commercial production is seamless and efficient.
- Mix benzoic acid and cyclohexanecarboxylic acid in a molar ratio of 1: 1.05-1.15 to prepare the raw material mixture.
- Conduct condensation reaction at 200°C-400°C using Mn2+ salt catalyst to generate cyclohexyl phenyl ketone intermediate.
- Perform chlorination and alkaline hydrolysis on the intermediate to finalize the synthesis of 1-hydroxycyclohexyl phenyl ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Mn-catalyzed condensation process offers profound strategic advantages that extend beyond simple chemical efficiency. The elimination of hazardous reagents like phosphorus trichloride and aluminum trichloride translates directly into a drastic simplification of the supply chain, as there is no longer a need to source, store, and handle these dangerous materials. This reduction in hazardous inventory significantly lowers insurance premiums and safety compliance costs, contributing to substantial cost savings in the overall manufacturing budget. Furthermore, the continuous nature of the production process, supported by a catalyst that remains active for hundreds of days, ensures a steady and reliable flow of product, mitigating the risks of supply disruptions that often plague batch-based chemical manufacturing. The ability to maintain long production cycles without frequent shutdowns for maintenance or catalyst replacement enhances the overall equipment effectiveness, allowing manufacturers to meet tight delivery deadlines with greater confidence. These operational improvements collectively strengthen the position of a reliable photoinitiator supplier in the global market.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous acylation agents eliminates the need for complex waste treatment systems designed to handle phosphorus and aluminum by-products. This simplification of the waste stream leads to significantly reduced disposal costs and lowers the capital expenditure required for environmental protection facilities. Additionally, the high yield of the condensation reaction ensures that raw material utilization is maximized, minimizing the cost per kilogram of the final photoinitiator product. The continuous operation mode further reduces energy consumption per unit of output compared to intermittent batch processes, driving down utility costs over the long term. These factors combine to create a highly cost-competitive production model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The exceptional stability of the Mn2+ catalyst allows for continuous production runs lasting over 300 days, which drastically reduces the frequency of production stoppages. This continuity is critical for maintaining consistent inventory levels and ensuring that customer orders are fulfilled on time, every time. By minimizing the need for catalyst replacement and reactor cleaning, the process reduces the operational downtime that often leads to supply bottlenecks in the fine chemical industry. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. For buyers, this translates into a more predictable procurement experience with reduced risk of delays or quality deviations.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with data showing successful operation in reactors capable of producing hundreds of tons of intermediate product. The significant reduction in solid waste generation, down to merely a few kilograms per ton of product, ensures that the manufacturing facility remains well within environmental regulatory limits. This low waste profile simplifies the permitting process for new production lines and reduces the risk of regulatory fines or shutdowns due to non-compliance. The environmentally friendly nature of the process also aligns with the sustainability goals of major multinational corporations, making the product more attractive to eco-conscious buyers. Scalability is further supported by the use of common and stable raw materials, ensuring that supply can be ramped up quickly to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These answers are derived directly from the patent specifications and experimental data, providing a factual basis for decision-making. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The information covers catalyst performance, waste management, and product quality, offering a comprehensive overview of the technology's value proposition. Stakeholders are encouraged to review these points to gain a deeper appreciation of the operational advantages offered by this method.
Q: How does the Mn2+ catalyzed route improve environmental compliance compared to Friedel-Crafts?
A: The novel condensation method eliminates the need for hazardous reagents like phosphorus trichloride and aluminum trichloride, significantly reducing hazardous wastewater and solid waste generation.
Q: What is the expected catalyst stability for continuous production of UV-184?
A: Industrial data indicates that Mn2+ salt catalysts can maintain activity for over 320 days of continuous operation, ensuring long production cycles without frequent replacement.
Q: Can this synthesis method be scaled for high-purity photoinitiator manufacturing?
A: Yes, the process supports continuous feeding and distillation, achieving yields over 90% and intermediate purity exceeding 99%, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoinitiator UV-184 Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Mn-catalyzed condensation route for the global photoinitiator market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying that every batch of Photoinitiator UV-184 meets the highest international standards. We understand the critical importance of supply continuity for our partners, and our advanced manufacturing facilities are equipped to handle the continuous processing requirements of this novel technology. By leveraging our technical expertise and infrastructure, we can help you secure a stable supply of high-purity photoinitiators while optimizing your overall production costs.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your supply chain efficiency and product competitiveness. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to request specific COA data and route feasibility assessments to verify the compatibility of this process with your current formulation strategies. Partnering with us means gaining access to cutting-edge chemical technology backed by a reliable and experienced manufacturing partner dedicated to your success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
