Optimizing Agrochemical Intermediate Production via Advanced Copper-Catalyzed Coupling Strategies
The global agrochemical industry faces continuous pressure to develop novel active ingredients that overcome pest resistance while maintaining economic viability in large-scale manufacturing. Patent CN110139853B addresses a critical bottleneck in the synthesis of high-value pesticidal thioethers and sulfoxides, specifically targeting the cost and efficiency of the key N-arylation step. This intellectual property discloses a refined synthetic methodology that optimizes the copper-catalyzed coupling of pyrazole derivatives with halopyridines, a transformation central to producing next-generation insecticides. By re-engineering the catalyst system and ligand environment, the patent offers a pathway to substantially lower production costs without compromising yield or purity, making it a vital reference for manufacturers seeking competitive advantages in the agrochemical intermediate market.
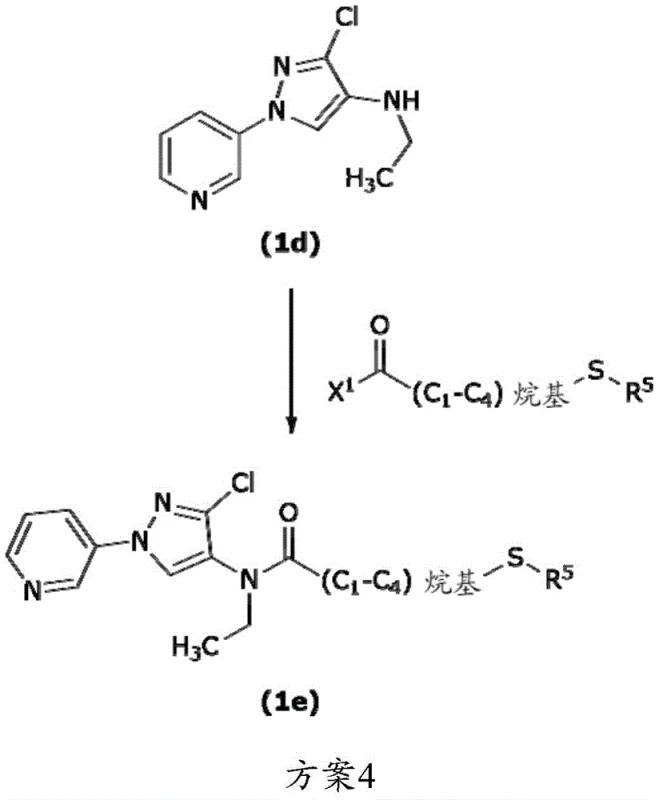
Traditional manufacturing routes for these complex heterocyclic scaffolds often rely on the Ullmann coupling reaction, which historically requires stoichiometric or near-stoichiometric amounts of expensive ligands to drive the reaction to completion. As illustrated in the prior art methodology, the reliance on N,N'-dimethylethane-1,2-diamine (DMEDA) presents a significant economic burden due to its high market price, approximately $256 per kilogram. Furthermore, the conventional process typically necessitates ligand loadings between 0.4 and 0.6 molar equivalents relative to the substrate, inflating the raw material costs for multi-ton production campaigns. This dependency creates a vulnerability in the supply chain, where fluctuations in ligand pricing directly impact the margin of the final active pharmaceutical or agrochemical ingredient.
The novel approach detailed in the patent fundamentally shifts the economic model of this synthesis by introducing a flexible ligand and additive system that drastically reduces dependency on costly amines. Instead of relying solely on high loadings of DMEDA, the invention demonstrates that the reaction can proceed efficiently with significantly reduced ligand concentrations when specific additives such as tetramethylethylenediamine (TMEDA), benzonitrile, or various alkylamines are employed. Alternatively, the process allows for the substitution of DMEDA with much more economical ligands like triethylenetetramine (TETA), which costs approximately $4.8 per kilogram, representing a massive reduction in input material expenses. This strategic modification not only lowers the direct cost of goods sold but also simplifies the downstream purification processes by reducing the amount of organic amine residues that must be removed from the final product.
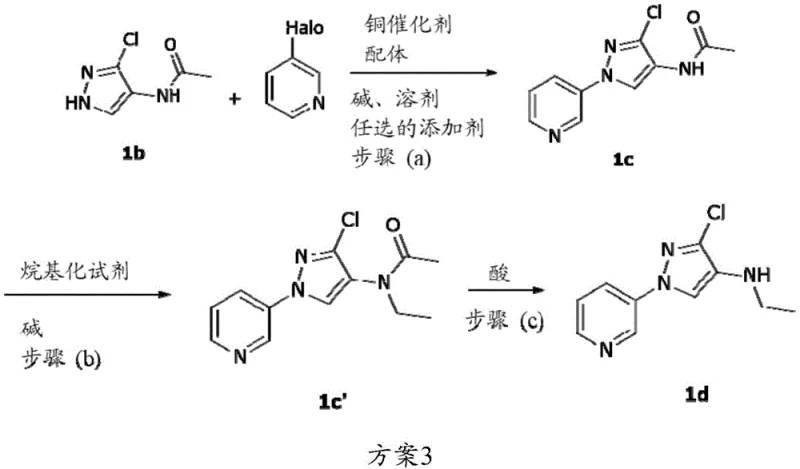
Mechanistically, the success of this optimized Ullmann coupling relies on the synergistic interaction between the copper catalyst, the primary ligand, and the newly introduced additives. The copper species, typically copper(I) chloride or iodide, facilitates the oxidative addition and reductive elimination cycles necessary for carbon-nitrogen bond formation. The presence of additives appears to stabilize the active catalytic species or modify the coordination sphere in a way that enhances turnover frequency, allowing for lower catalyst and ligand loadings. For instance, the patent describes conditions where DMEDA loading is reduced to 0.1 molar equivalents in the presence of additives, yet high conversion rates are maintained. This suggests that the additives may act as co-ligands or phase transfer agents that improve the solubility of the inorganic base or the copper complex in the reaction medium, thereby increasing the effective concentration of the active catalyst. Such mechanistic insights are crucial for R&D teams aiming to replicate these results on a pilot plant scale, ensuring that the delicate balance of reagents is maintained to prevent the formation of homocoupling byproducts or dehalogenated impurities.
Impurity control is another critical aspect addressed by this refined protocol, particularly regarding the removal of residual copper and amine ligands which are regulated contaminants in agrochemical products. The use of inorganic bases like potassium carbonate or potassium phosphate, combined with the specific solvent systems such as N-methyl-2-pyrrolidone (NMP) or dimethylformamide (DMF), facilitates a cleaner reaction profile. The patent outlines workup procedures involving aqueous quenching and filtration that effectively separate the solid product from soluble copper salts and excess ligands. By minimizing the initial loading of these auxiliaries, the burden on the purification train is significantly lightened, reducing the volume of solvent required for recrystallization or chromatography. This leads to a higher overall mass balance and reduced waste generation, aligning with green chemistry principles that are increasingly important for regulatory compliance and environmental sustainability in chemical manufacturing.
How to Synthesize 3-Chloro-N-ethyl-1-(pyridin-3-yl)-1H-pyrazol-4-amine Efficiently
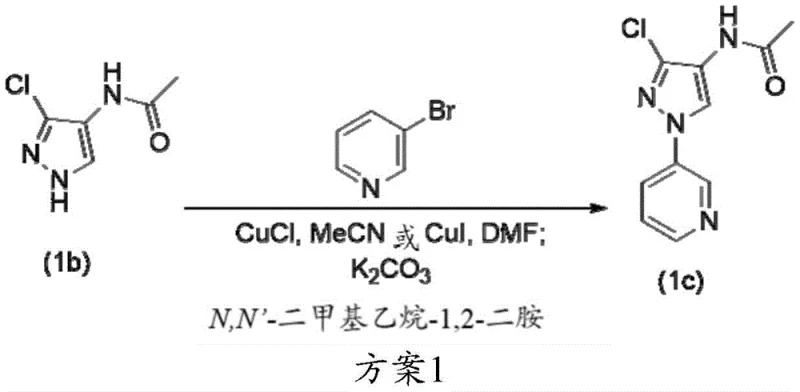
The synthesis of the key intermediate 3-chloro-N-ethyl-1-(pyridin-3-yl)-1H-pyrazol-4-amine (1d) serves as a prime example of the patent's utility, demonstrating a robust three-step sequence that can be adapted for commercial production. The process begins with the copper-catalyzed coupling of N-(3-chloro-1H-pyrazol-4-yl)acetamide with 3-bromopyridine, followed by N-alkylation with ethyl bromide, and concludes with acid hydrolysis to remove the acetyl protecting group. This telescoped approach minimizes the handling of intermediates and maximizes throughput, making it an ideal candidate for continuous manufacturing or large-batch processing. The detailed operational parameters, including temperature controls and reagent addition rates, are critical for ensuring safety and reproducibility, particularly during the exothermic alkylation and hydrolysis steps.
- Perform copper-catalyzed N-arylation of N-(3-chloro-1H-pyrazol-4-yl)acetamide with 3-halopyridine using optimized ligand loading.
- Execute N-alkylation of the resulting acetamide intermediate using ethyl bromide and a strong base like sodium tert-butoxide.
- Complete the sequence with acid-mediated hydrolysis to remove the acetyl protecting group and isolate the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the methodologies described in CN110139853B offers tangible benefits that extend beyond simple reaction yields. The primary advantage lies in the drastic reduction of raw material costs associated with the ligand system. By switching from exclusive use of high-cost DMEDA to lower-cost alternatives like TETA or by reducing DMEDA usage through additives, manufacturers can achieve substantial cost savings in the bill of materials. This is particularly impactful for high-volume agrochemical intermediates where even small per-kilogram savings translate into significant annual financial improvements. Furthermore, the reliance on commercially available and inexpensive starting materials enhances supply chain resilience, reducing the risk of production delays caused by shortages of specialized reagents.
- Cost Reduction in Manufacturing: The ability to reduce the loading of expensive ligands like DMEDA from 0.4-0.6 equivalents down to 0.1 equivalents, or substitute them entirely with ligands costing significantly less per kilogram, directly lowers the variable cost of production. This optimization eliminates the need for expensive metal scavenging resins that are often required to remove high levels of copper-ligand complexes, further reducing downstream processing costs. The cumulative effect of these changes results in a more lean and cost-effective manufacturing process that improves the overall margin profile of the final pesticidal active ingredient.
- Enhanced Supply Chain Reliability: The process utilizes widely available commodity chemicals such as 3-bromopyridine, copper(I) chloride, and common inorganic bases, which are sourced from a broad global supplier base. This diversification of the supply chain mitigates the risk of single-source dependency and ensures consistent availability of critical inputs. Additionally, the robustness of the reaction conditions allows for flexibility in sourcing, as the process tolerates slight variations in reagent quality without compromising the integrity of the final product, thereby securing long-term supply continuity for downstream formulation partners.
- Scalability and Environmental Compliance: The described methods are designed with scalability in mind, utilizing solvents and conditions that are compatible with standard stainless steel reactor infrastructure. The reduction in ligand loading and the potential for telescoping multiple steps into a single vessel decrease the total solvent consumption and waste generation per kilogram of product. This aligns with stricter environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste disposal, facilitating easier permitting and operation of manufacturing facilities while supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in an industrial setting. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does this patent address the high cost of DMEDA ligands in Ullmann coupling?
A: The patent demonstrates that expensive DMEDA loading can be significantly reduced from 0.4-0.6 equivalents to as low as 0.1 equivalents by introducing specific additives like TMEDA or benzonitrile, or by substituting with cheaper ligands like TETA.
Q: Can the synthesis steps be telescoped to improve manufacturing efficiency?
A: Yes, the disclosed methods allow for telescoping the Ullmann coupling, alkylation, and hydrolysis steps, potentially eliminating intermediate isolation and reducing overall processing time and solvent consumption.
Q: What are the primary applications of the synthesized thioether compounds?
A: The resulting pesticidal thioethers and sulfoxides are designed for agricultural use to control pest species that cause significant crop losses, offering a solution for resistance management in modern farming.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pesticidal Thioether Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in maintaining a competitive edge in the global agrochemical market. Our technical team has extensively analyzed the innovations presented in CN110139853B and possesses the expertise to implement these advanced copper-catalyzed protocols at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of intermediate meets the exacting standards required for pesticide registration and formulation.
We invite potential partners to engage with our technical procurement team to discuss how these process optimizations can be tailored to your specific supply chain needs. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis that quantifies the potential economic benefits of adopting this novel synthetic route. We encourage you to request specific COA data and route feasibility assessments to validate the quality and reliability of our production processes, ensuring a stable and cost-effective supply of high-purity agrochemical intermediates for your global operations.
