Advanced Manufacturing Strategy for High-Purity Ketolide Compounds via Optimized Synthetic Routes
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex antibiotic intermediates, particularly ketolides, which represent a critical class of macrolide antibiotics with enhanced antibacterial properties. Patent CN110418797B discloses a sophisticated and highly efficient method for preparing ketolide compounds, specifically targeting the synthesis of Formula (I) through a series of well-defined chemical transformations. This technical breakthrough addresses long-standing challenges in the manufacturing of macrolide derivatives by introducing a streamlined sequence that utilizes selective protection and deprotection strategies alongside key cyclization and functionalization steps. The process is designed to maximize yield and purity while minimizing the formation of difficult-to-remove impurities, which is a paramount concern for R&D Directors overseeing process development. By leveraging specific reagents such as triethylsilyl chloride and trimethylsilyl cyanide under controlled thermal conditions, the methodology ensures consistent quality across batches. This report provides a deep technical and commercial analysis of this patent, offering valuable insights for procurement and supply chain leaders looking to secure reliable sources for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for ketolide compounds often suffer from significant drawbacks that hinder efficient commercial production and compromise final product quality. Conventional methods frequently rely on harsh reaction conditions that can lead to the degradation of the sensitive macrolide backbone, resulting in complex impurity profiles that are costly and time-consuming to purify. Many existing processes utilize non-selective reagents that fail to distinguish between multiple hydroxyl groups on the sugar moieties, leading to mixtures of regioisomers that drastically reduce overall yield. Furthermore, older methodologies often involve multiple isolation and purification steps between each transformation, which increases solvent consumption, waste generation, and overall processing time. The use of unstable intermediates in conventional routes can also pose safety risks during scale-up, limiting the ability of manufacturers to produce these compounds in metric ton quantities. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for pharmaceutical companies aiming to bring new antibiotic therapies to market quickly and cost-effectively.
The Novel Approach
The method disclosed in CN110418797B represents a significant evolution in ketolide synthesis by introducing a highly selective and modular approach that overcomes the limitations of prior art. This novel route employs a strategic silylation step using triethylsilyl chloride to protect specific hydroxyl groups, thereby preventing unwanted side reactions during subsequent transformations. The process utilizes triphosgene for cyclization, a reagent that offers superior control over reaction kinetics compared to traditional phosgene sources, enhancing operational safety and reproducibility. A key innovation lies in the cyanation step using trimethylsilyl cyanide, which allows for the efficient introduction of the nitrile functionality under mild conditions, preserving the stereochemical integrity of the molecule. By optimizing solvent systems and temperature ranges, such as maintaining reactions between 5°C to 30°C, the process minimizes thermal stress on the intermediate compounds. This results in a cleaner reaction profile with fewer by-products, simplifying downstream purification and significantly improving the overall economic viability of the manufacturing process for commercial partners.
Mechanistic Insights into Silylation and Cyanation Transformations
The core of this synthetic strategy relies on precise chemical mechanisms that ensure high fidelity in the construction of the ketolide scaffold. The initial silylation step involves the nucleophilic attack of hydroxyl groups on the silicon atom of triethylsilyl chloride, facilitated by a base such as triethylamine or 4-dimethylaminopyridine. This protection is crucial as it masks reactive sites that could otherwise interfere with the subsequent cyclization reaction. The use of triethylsilyl (TES) groups is particularly advantageous due to their stability under the reaction conditions yet ease of removal during the final deprotection stage. Following protection, the cyclization step utilizes triphosgene to activate specific positions on the macrolide ring, leading to the formation of a cyclic carbonate or similar intermediate which is then eliminated to form an unsaturated bond. This sequence is meticulously controlled to prevent over-reaction or decomposition.
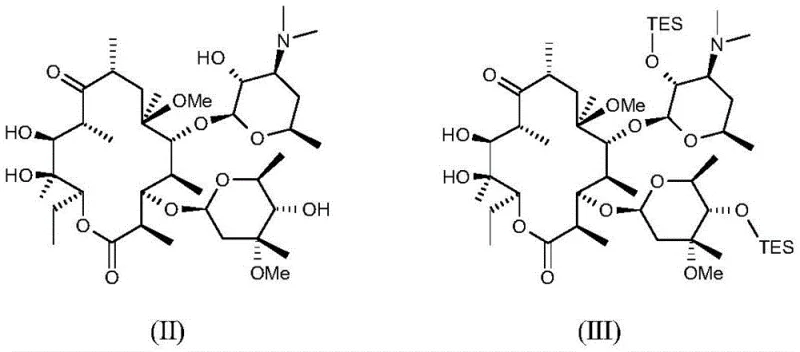
Subsequent transformations involve the introduction of key functional groups necessary for biological activity. The cyanation step is a pivotal moment in the synthesis, where trimethylsilyl cyanide reacts with an activated intermediate in the presence of a base and activator. This reaction proceeds through a nucleophilic addition mechanism, where the cyanide ion attacks an electrophilic center to form the nitrile group found in Formula (VII). The choice of activator, such as methanol or water, plays a critical role in facilitating this transformation without causing hydrolysis of other sensitive esters or lactones in the molecule. The final deprotection steps utilize acidic conditions to remove the silyl groups and other protecting moieties, revealing the final active ketolide structure. Each step is monitored via HPLC to ensure conversion rates exceed 90%, demonstrating the robustness of the chemical design.
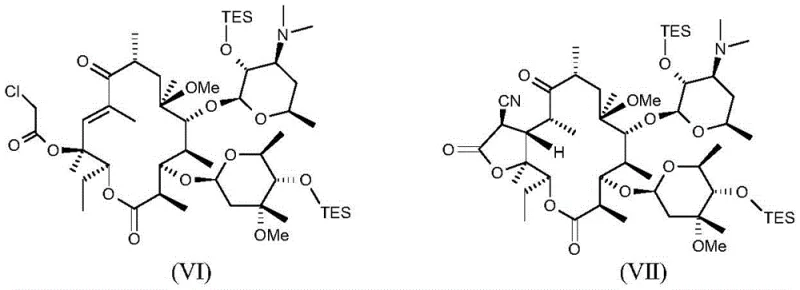
How to Synthesize Ketolide Compound Efficiently
The synthesis of the target ketolide compound requires a disciplined approach to reaction conditions and reagent quality to achieve the high yields and purity levels described in the patent. The process begins with the preparation of the silylated intermediate, where strict temperature control between 5°C to 10°C is maintained to prevent exothermic runaway and ensure selective mono- or di-silylation. Following isolation, the intermediate undergoes cyclization using triphosgene in dichloromethane, a step that demands careful handling of reagents to maintain safety and reaction efficiency. The subsequent elimination and functionalization steps utilize common organic solvents like acetone and DMF, which are easily recoverable and recyclable in a commercial setting. Detailed standardized synthetic steps are essential for reproducibility, particularly when scaling from laboratory glassware to industrial reactors.
- Protect hydroxyl groups using triethylsilyl chloride in the presence of base and solvent at 5°C to 30°C.
- Perform cyclization using triphosgene followed by elimination with a base to form the unsaturated intermediate.
- Execute cyanation using trimethylsilyl cyanide with an activator, followed by deprotection to yield the final ketolide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthetic route offers substantial strategic advantages that extend beyond mere technical feasibility. The high yields reported in the examples, such as 92.3% in the initial silylation step and 98% in the elimination step, indicate a highly efficient use of raw materials, which directly correlates to reduced cost of goods sold (COGS). By minimizing the number of purification steps and maximizing the purity of intermediates, the process significantly reduces solvent consumption and waste disposal costs, contributing to a more sustainable and cost-effective manufacturing operation. The use of commercially available and stable reagents like triethylsilyl chloride and triphosgene ensures a reliable supply chain, reducing the risk of production delays caused by specialty chemical shortages. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, adding another layer of cost optimization for large-scale production facilities.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route eliminates the need for expensive transition metal catalysts or complex chromatographic purifications that are often required in conventional methods. By achieving high HPLC purity levels, such as 98% in key intermediates, the process reduces the burden on quality control labs and minimizes the loss of valuable material during recrystallization or column chromatography. The high atom economy of the cyanation and cyclization steps ensures that a greater proportion of starting materials are converted into the final product, drastically lowering the raw material cost per kilogram. This efficiency allows for substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard solvents like methanol, dichloromethane, and acetone means that the supply chain is less vulnerable to disruptions associated with exotic or regulated reagents. The robustness of the reaction conditions, which tolerate a wide range of temperatures and slight variations in reagent quality, ensures consistent production output even in diverse manufacturing environments. This reliability is critical for maintaining continuous supply to pharmaceutical clients who require just-in-time delivery of intermediates for their own drug substance manufacturing. The scalability of the process from 100 kgs to multi-ton levels ensures that procurement teams can secure long-term contracts with confidence in the supplier's ability to meet demand fluctuations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry, such as filtration, distillation, and crystallization. The reduction in hazardous waste generation, due to higher yields and fewer purification steps, simplifies compliance with environmental regulations and reduces the cost of waste treatment. The use of triphosgene as a safer alternative to gaseous phosgene enhances workplace safety and reduces the regulatory burden associated with handling highly toxic gases. These factors combined make the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to expand their portfolio of antibiotic intermediates while maintaining strict environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ketolide synthesis process. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing manufacturing frameworks. The focus is on clarity regarding reaction parameters, safety considerations, and quality outcomes to ensure all parties have a unified understanding of the process capabilities.
Q: What are the critical reaction conditions for the silylation step in this ketolide synthesis?
A: The silylation step requires precise temperature control between 5°C to 30°C, utilizing triethylsilyl chloride with bases like triethylamine or DMAP in solvents such as DMF or dichloromethane to ensure high conversion rates.
Q: How does this patent process improve impurity control compared to conventional methods?
A: By employing specific protecting group strategies and mild reaction conditions, the process minimizes side reactions and degradation, resulting in HPLC purity levels reaching up to 98% in intermediate steps.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the use of common solvents like methanol and dichloromethane, along with scalable reagents like triphosgene and TMSCN, makes the process highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketolide Compound Supplier
The technical potential of the synthetic route described in CN110418797B is immense, offering a pathway to high-quality ketolide intermediates that are essential for the next generation of antibacterial therapies. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this patent from the laboratory to the marketplace. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating every step of this complex synthesis, ensuring that the final product meets the exacting standards of global pharmaceutical regulators. We understand the critical nature of antibiotic supply chains and are committed to delivering consistency and reliability in every batch we produce.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this optimized process can enhance your supply chain resilience. By partnering with us, you gain access to a manufacturing partner that values technical excellence and commercial integrity, ensuring a seamless transition from development to commercial supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
