Revolutionizing Aromatic Hydroxylamine Production With Green Zinc-CO2 Reduction Technology
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies that balance high efficiency with environmental responsibility. Patent CN101357894A introduces a groundbreaking approach for the preparation of aromatic hydroxylamine through the selective reduction of aromatic nitro compounds. This technology fundamentally shifts the paradigm from traditional ammonium chloride-based systems to a greener zinc-carbon dioxide aqueous system. By leveraging the acidic properties of dissolved CO2, this method achieves complete conversion of nitro compounds while maintaining exceptional selectivity for the hydroxylamine product. For global procurement leaders and R&D directors, this represents a critical opportunity to optimize supply chains for high-purity pharmaceutical intermediates. The elimination of toxic byproducts and the ability to recycle the aqueous phase position this technology as a cornerstone for future-proofing chemical manufacturing processes against tightening environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic hydroxylamines has relied heavily on the zinc powder reduction method in ammonium chloride aqueous solutions. While effective in terms of conversion, this legacy technology suffers from severe environmental and operational drawbacks that impact the total cost of ownership for chemical manufacturers. The primary issue lies in the massive generation of wastewater containing high concentrations of ammonium salts and zinc residues, which necessitates complex and expensive treatment protocols before discharge. Furthermore, the inability to recycle the reaction medium means that every batch consumes fresh water and chemicals, driving up raw material costs and increasing the facility's water footprint. Alternative methods such as catalytic hydrogenation pose significant safety risks due to the use of flammable hydrogen gas and expensive noble metal catalysts that are difficult to recover. These factors collectively create bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, limiting the agility of supply chains to respond to market demands.
The Novel Approach
The innovative method disclosed in the patent data replaces the problematic ammonium chloride with a carbon dioxide atmosphere in an aqueous zinc powder system. This strategic substitution utilizes the in-situ formation of carbonic acid to drive the reduction reaction, effectively eliminating the source of nitrogenous wastewater. The process operates under mild conditions with CO2 pressures ranging from 0.1 to 15MPa and temperatures between 0 and 120°C, ensuring robust control over the reaction kinetics. Crucially, the aqueous phase remains chemically stable after the reaction, allowing for direct recycling and reuse in subsequent batches without extensive purification. This closed-loop capability drastically simplifies the downstream processing workflow and reduces the burden on waste management infrastructure. By adopting this novel approach, manufacturers can achieve significant cost reduction in fine chemical manufacturing while adhering to the highest standards of green chemistry and sustainability.
Mechanistic Insights into Zinc-CO2 Catalyzed Selective Reduction
The core mechanism of this transformation relies on the unique interaction between metallic zinc, water, and carbon dioxide under pressure. When CO2 is dissolved in the aqueous reaction medium, it forms carbonic acid, which provides the necessary protons to facilitate the electron transfer from zinc to the nitro group. This proton source is self-regulating and avoids the accumulation of non-volatile salts that plague traditional methods. The zinc powder acts as the electron donor, reducing the nitro group stepwise to the hydroxylamine stage without proceeding to the amine, provided the reaction conditions are optimized. The selectivity is governed by the specific molar ratio of zinc to the nitro compound, typically maintained between 0.5 and 20:1, and the mass ratio of water to substrate. This precise control ensures that the reduction stops at the hydroxylamine stage, preventing over-reduction which is a common failure mode in catalytic hydrogenation processes.
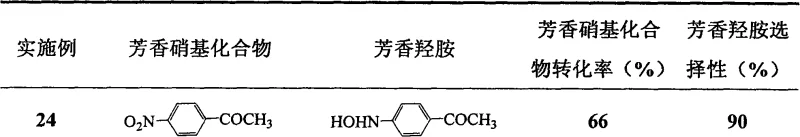
Impurity control is inherently superior in this system due to the absence of extraneous ions like ammonium or chloride which can form difficult-to-remove complexes with the product. The reaction pathway favors the formation of the desired aromatic hydroxylamine with selectivity reaching as high as 98% for certain substrates like p-chloronitrobenzene. The clean reaction profile minimizes the formation of azo or azoxy byproducts, which are typical impurities in nitro reductions. This high purity profile is essential for downstream applications in drug synthesis where impurity thresholds are strictly regulated. The ability to tune the reaction by adjusting CO2 pressure and temperature allows for further optimization of the impurity spectrum, ensuring that the final product meets stringent quality specifications required by global regulatory bodies.
How to Synthesize Aromatic Hydroxylamine Efficiently
Implementing this synthesis route requires careful attention to the loading sequence and pressure management within the reactor system. The process begins with the precise weighing of the aromatic nitro compound and zinc powder, followed by the addition of deionized water to establish the reaction slurry. Once the reactor is sealed, it is critical to perform multiple purge cycles with carbon dioxide to remove atmospheric oxygen, which could otherwise oxidize the sensitive hydroxylamine product or pose safety hazards. The detailed standardized synthesis steps see the guide below.
- Load the reactor with aromatic nitro compounds, zinc powder, and water according to specific molar and mass ratios.
- Seal the reactor and purge with carbon dioxide gas three times to ensure an inert and acidic atmosphere.
- Pressurize with CO2 to 0.1-15MPa, heat to 0-120°C, and stir for 0.25-80 hours to complete the selective reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-CO2 reduction technology offers transformative benefits that extend beyond simple yield improvements. The elimination of ammonium chloride removes a significant variable cost associated with raw material procurement and waste disposal fees. Since the aqueous phase can be recycled, the consumption of fresh water is dramatically lowered, contributing to substantial cost savings in utilities and environmental compliance. The process avoids the use of high-pressure hydrogen gas, thereby reducing the insurance premiums and safety infrastructure costs associated with hazardous material handling. These operational efficiencies translate directly into a more competitive pricing structure for the final intermediate, enhancing the margin potential for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The removal of ammonium chloride from the recipe eliminates the need for purchasing this bulk chemical and the subsequent costs of treating nitrogen-rich wastewater. The recyclability of the water phase means that solvent recovery costs are negligible compared to traditional organic solvent-based processes. Additionally, the use of zinc powder, a commodity chemical with stable pricing, ensures predictable raw material costs without exposure to the volatility of noble metal markets. This stability allows for long-term supply contracts with fixed pricing models, providing financial certainty for procurement planning.
- Enhanced Supply Chain Reliability: The simplicity of the reagent profile, consisting mainly of zinc, water, and CO2, ensures that supply chain disruptions are minimized as these materials are globally available and easy to source. The robustness of the reaction conditions allows for flexible production scheduling without the need for specialized cryogenic or ultra-high-pressure equipment. This flexibility enables manufacturers to respond quickly to fluctuations in demand for high-purity aromatic hydroxylamines, reducing lead times for critical pharmaceutical projects. The reduced dependency on complex catalysts also mitigates the risk of production stoppages due to catalyst supply shortages.
- Scalability and Environmental Compliance: The aqueous nature of the reaction makes it inherently safer and easier to scale from pilot plant to multi-ton commercial production without significant engineering redesigns. The absence of toxic sulfur or heavy metal contaminants simplifies the regulatory approval process for new manufacturing sites, accelerating time-to-market for new products. Environmental compliance is significantly streamlined as the process generates minimal hazardous waste, aligning with global initiatives for sustainable chemical manufacturing. This alignment reduces the administrative burden on EHS teams and lowers the risk of regulatory fines or shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green reduction technology. These answers are derived directly from the patent specifications and practical engineering considerations for scaling this chemistry. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing production lines.
Q: How does the CO2 atmosphere improve the environmental profile of hydroxylamine synthesis?
A: The CO2 atmosphere generates carbonic acid in situ, replacing the need for ammonium chloride. This eliminates the generation of large volumes of nitrogen-containing wastewater, allowing the aqueous phase to be recycled directly.
Q: What is the selectivity advantage of this zinc-based method over catalytic hydrogenation?
A: Unlike catalytic hydrogenation which often requires strict control to prevent over-reduction to amines, this method achieves high selectivity for the hydroxylamine intermediate, with conversion rates reaching 100% and selectivity up to 98% for specific substrates.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses standard industrial reagents like zinc powder and water, operates at moderate temperatures, and avoids hazardous hydrogen gas, making it highly scalable and safer for commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Hydroxylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this patented zinc-CO2 reduction technology in modernizing the production of critical pharmaceutical intermediates. Our CDMO expertise allows us to adapt and optimize this green chemistry route for diverse client needs, ensuring seamless technology transfer from lab to plant. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply requirements are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of aromatic hydroxylamine delivered meets the highest international standards for drug substance manufacturing.
We invite you to collaborate with us to leverage this advanced synthesis method for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this technology can enhance your product portfolio. Let us help you build a more resilient and sustainable supply chain for your high-value chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
