Scalable Manufacturing of Dehydroequol: A Technical Breakthrough for API Intermediates
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective routes for synthesizing bioactive molecules, particularly those with potent anticancer properties. Patent CN103224481A introduces a groundbreaking preparation method for Dehydroequol, also known scientifically as Haginin E or 3-(4-hydroxyphenyl)-2H-1-chromen-7-ol. This compound has garnered significant attention due to its ability to induce apoptosis in tumor cells, specifically showing efficacy against ovarian, cervical, and prostate cancers, while sparing normal cells. The structural integrity and purity of this molecule are paramount for its biological activity, making the development of efficient synthetic pathways a critical priority for R&D teams globally. 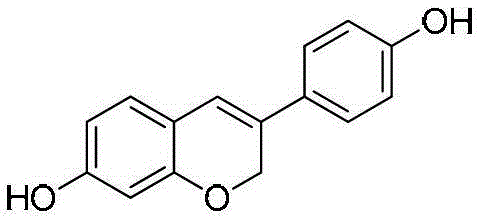 As illustrated in the molecular structure, the presence of specific hydroxyl groups and the chromene ring system defines its pharmacological profile. The patented technology addresses the historical challenges of complex processing and low yields associated with earlier methods, offering a streamlined four-step sequence that begins with the abundant and inexpensive natural product, Daidzein.
As illustrated in the molecular structure, the presence of specific hydroxyl groups and the chromene ring system defines its pharmacological profile. The patented technology addresses the historical challenges of complex processing and low yields associated with earlier methods, offering a streamlined four-step sequence that begins with the abundant and inexpensive natural product, Daidzein.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Dehydroequol was fraught with inefficiencies that hindered its commercial viability and widespread adoption in drug discovery pipelines. Existing literature, such as the method disclosed in Tetrahedron Letters (2009), relied on a multi-step sequence involving Wittig reactions and olefin metathesis using Grubbs 2nd generation catalysts. 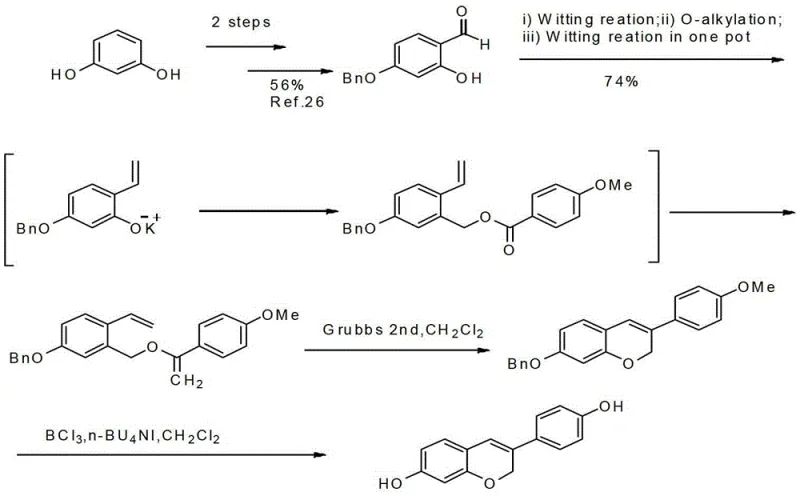 This conventional approach presents several critical bottlenecks for industrial application. Firstly, the reliance on Grubbs catalysts introduces a substantial cost burden, as these ruthenium-based complexes are expensive and often required in stoichiometric or near-stoichiometric amounts for difficult transformations. Secondly, the removal of residual heavy metals from the final API intermediate is a rigorous and costly process, necessitating specialized scavenging resins and extensive analytical testing to meet regulatory limits. Furthermore, the overall yield of such multi-step sequences tends to degrade significantly with each additional transformation, leading to poor atom economy and increased waste generation. The production cycle is inherently longer due to the sensitivity of the reagents and the need for inert atmosphere handling throughout multiple stages, making it unsuitable for the rapid, large-scale manufacturing required by the global supply chain.
This conventional approach presents several critical bottlenecks for industrial application. Firstly, the reliance on Grubbs catalysts introduces a substantial cost burden, as these ruthenium-based complexes are expensive and often required in stoichiometric or near-stoichiometric amounts for difficult transformations. Secondly, the removal of residual heavy metals from the final API intermediate is a rigorous and costly process, necessitating specialized scavenging resins and extensive analytical testing to meet regulatory limits. Furthermore, the overall yield of such multi-step sequences tends to degrade significantly with each additional transformation, leading to poor atom economy and increased waste generation. The production cycle is inherently longer due to the sensitivity of the reagents and the need for inert atmosphere handling throughout multiple stages, making it unsuitable for the rapid, large-scale manufacturing required by the global supply chain.
The Novel Approach
In stark contrast, the methodology outlined in patent CN103224481A represents a paradigm shift towards green and economical chemistry. 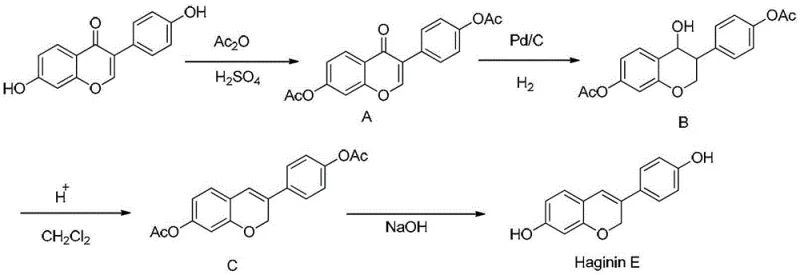 The novel approach leverages Daidzein, a readily available isoflavone derived from soy, as the foundational building block. This strategic choice immediately lowers the barrier to entry regarding raw material costs and supply chain security. The synthesis proceeds through a logical sequence of protection, reduction, cyclization, and deprotection. Instead of expensive metathesis catalysts, the core transformation utilizes catalytic hydrogenation with Palladium on Carbon (Pd/C), a standard, recyclable, and cost-effective heterogeneous catalyst widely available in the fine chemical industry. The process eliminates the need for complex organometallic manipulations, replacing them with robust acid-catalyzed cyclization and base-mediated hydrolysis steps. This simplification not only enhances the safety profile by avoiding high-risk reagents but also drastically improves the operational simplicity, allowing for easier post-treatment via filtration and crystallization rather than tedious chromatographic separations. The result is a process where each step consistently achieves yields exceeding 80%, culminating in a highly efficient overall production capability.
The novel approach leverages Daidzein, a readily available isoflavone derived from soy, as the foundational building block. This strategic choice immediately lowers the barrier to entry regarding raw material costs and supply chain security. The synthesis proceeds through a logical sequence of protection, reduction, cyclization, and deprotection. Instead of expensive metathesis catalysts, the core transformation utilizes catalytic hydrogenation with Palladium on Carbon (Pd/C), a standard, recyclable, and cost-effective heterogeneous catalyst widely available in the fine chemical industry. The process eliminates the need for complex organometallic manipulations, replacing them with robust acid-catalyzed cyclization and base-mediated hydrolysis steps. This simplification not only enhances the safety profile by avoiding high-risk reagents but also drastically improves the operational simplicity, allowing for easier post-treatment via filtration and crystallization rather than tedious chromatographic separations. The result is a process where each step consistently achieves yields exceeding 80%, culminating in a highly efficient overall production capability.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Acid Cyclization
The heart of this synthetic strategy lies in the precise manipulation of the isoflavone skeleton to construct the chromene ring system found in Dehydroequol. The initial acetylation step serves a dual purpose: it protects the phenolic hydroxyl groups from unwanted side reactions during the subsequent reduction and activates the molecule for the specific regioselective transformations required later. Following protection, the catalytic hydrogenation step using 10% Pd/C under hydrogen pressure (5-15 atm) is critical. This step selectively reduces the alkene functionality within the heterocyclic ring without affecting the aromatic systems or the ester protecting groups. The use of a heterogeneous catalyst like Pd/C allows for easy removal via simple filtration, ensuring that the reaction mixture remains free of dissolved metal contaminants that could complicate downstream processing. The mechanism involves the adsorption of hydrogen onto the palladium surface, followed by syn-addition to the double bond, resulting in the saturated intermediate O,O-diacetyl-4-hydroxydaidzein with high stereochemical fidelity.
Subsequent to hydrogenation, the acid-catalyzed cyclization is the key step that forms the core chromene structure of the target molecule. By treating the reduced intermediate with an acidic solution, such as trifluoroacetic acid or hydrochloric acid, the molecule undergoes dehydration and intramolecular etherification. The acidic conditions protonate the oxygenated functionalities, facilitating the elimination of water and the formation of the new carbon-oxygen bond that closes the pyran ring. This step is meticulously controlled by temperature (25°C to 70°C) to prevent polymerization or degradation of the sensitive phenolic core. Finally, the removal of the acetyl protecting groups via saponification with sodium hydroxide restores the free phenolic hydroxyls, yielding the final Dehydroequol. This mechanistic pathway ensures that impurities are minimized at every stage, as the crystallization protocols specified in the patent effectively purge by-products, resulting in a final product with stringent purity specifications essential for pharmaceutical applications.
How to Synthesize Dehydroequol Efficiently
The synthesis of Dehydroequol via this patented route is designed for operational excellence, balancing high yield with procedural simplicity. The process begins with the acetylation of Daidzein using acetic anhydride and a catalytic amount of sulfuric acid, followed by a straightforward aqueous workup and recrystallization to secure the protected intermediate. This structured approach ensures that operators can reliably reproduce the results across different batch sizes, from laboratory benchtop to pilot plant scales, maintaining consistent quality and minimizing the risk of batch failure due to operator error or variable reaction conditions.
- Protect the hydroxyl groups of Daidzein using acetic anhydride and sulfuric acid to form O,O-diacetyl daidzein.
- Perform catalytic hydrogenation on the diacetyl intermediate using Pd/C under hydrogen pressure to reduce the double bond.
- Induce cyclization and dehydration using an acidic solution (e.g., trifluoroacetic acid) to form the chromene ring structure.
- Remove the acetyl protecting groups using sodium hydroxide (NaOH) to yield the final Dehydroequol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic advantages that directly impact the bottom line and operational resilience. The shift from complex, catalyst-intensive methodologies to a Daidzein-based hydrogenation route fundamentally alters the cost structure of manufacturing this valuable intermediate. By eliminating the dependency on precious metal catalysts like Ruthenium (Grubbs), the process removes a significant variable cost driver and mitigates the supply risk associated with scarce platinum group metals. Furthermore, the simplified workup procedures, which rely on filtration and crystallization rather than column chromatography, significantly reduce solvent consumption and waste disposal costs, aligning with modern sustainability goals and reducing the environmental footprint of production facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the utilization of Daidzein, a bulk commodity chemical with a stable and low market price, as the starting material. Unlike previous methods that required expensive, custom-synthesized precursors, this route leverages existing agricultural supply chains. Additionally, the replacement of homogeneous noble metal catalysts with heterogeneous Pd/C allows for catalyst recovery and reuse, further driving down the cost per kilogram of the final product. The avoidance of chromatographic purification steps translates to substantial savings in silica gel, solvents, and labor hours, creating a leaner and more cost-efficient manufacturing operation that can withstand market fluctuations.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions and the availability of raw materials. Daidzein is produced in large quantities globally, ensuring that feedstock shortages are unlikely to disrupt production schedules. The process conditions, involving standard hydrogenation and acid/base chemistry, are well-understood and can be executed in general-purpose chemical reactors without the need for specialized equipment required for air-sensitive organometallic chemistry. This flexibility allows for manufacturing to be distributed across multiple sites if necessary, reducing the risk of single-point failures and ensuring a steady flow of high-purity intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From a regulatory and environmental perspective, this method is superior. The avoidance of high-risk reagents and the generation of less hazardous waste streams simplify the permitting process for new manufacturing lines. The high yield per step (>80%) means that less raw material is wasted, improving the overall mass intensity of the process. The use of common solvents like ethanol, ethyl acetate, and methanol facilitates solvent recovery and recycling, further enhancing the environmental profile. This compliance-ready approach minimizes the administrative burden on EHS teams and accelerates the timeline for technology transfer from R&D to commercial production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common concerns regarding the practical application and quality assurance of this patented method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring that decision-makers have access to accurate and actionable information.
Q: What are the primary advantages of this Dehydroequol synthesis route over prior art?
A: The patented method utilizes Daidzein, a low-cost natural product, as the starting material and replaces expensive transition metal catalysts (like Grubbs catalyst) with standard Pd/C hydrogenation. This significantly simplifies the workflow and reduces raw material costs while maintaining yields above 80% per step.
Q: How does the process ensure high purity without chromatography?
A: The process relies on strategic recrystallization steps after each reaction stage. By utilizing solvents like ethanol, ethyl acetate, and methanol for purification, the method effectively removes impurities and by-products, ensuring high-purity specifications suitable for pharmaceutical applications without the need for costly column chromatography.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for scalability. It avoids hazardous reagents and complex operational conditions. The use of standard unit operations such as filtration, rotary evaporation, and crystallization makes it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroequol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the pathway described in CN103224481A and possesses the expertise to implement this efficient synthesis at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Dehydroequol meets the exacting standards required for oncology research and drug development.
We invite you to collaborate with us to optimize your supply chain for this valuable anticancer intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and budgetary goals efficiently.
