Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The integration of trifluoromethyl groups into nitrogen-containing heterocycles represents a cornerstone strategy in modern medicinal chemistry, significantly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As illustrated in the structural diversity of bioactive molecules below, the 1,2,4-triazole scaffold is ubiquitous in high-value pharmaceuticals ranging from antifungal agents like fluconazole to aromatase inhibitors like letrozole. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses critical bottlenecks in current synthetic methodologies. By utilizing a non-metallic iodine-promoted cyclization strategy, this technology offers a streamlined pathway for producing high-purity pharmaceutical intermediates, positioning it as a vital asset for reliable API intermediate suppliers seeking to optimize their manufacturing portfolios.
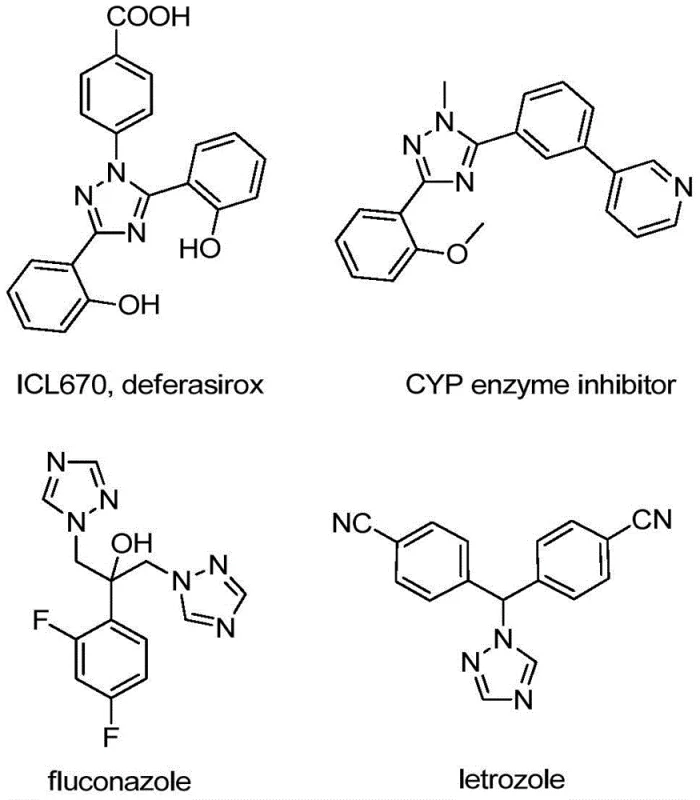
The strategic incorporation of the trifluoromethyl moiety into the triazole ring system is not merely a structural modification but a functional imperative for next-generation therapeutics. The electron-withdrawing nature of the CF3 group modulates the pKa of adjacent nitrogens and alters the electronic distribution of the heterocycle, often leading to improved binding affinity with biological targets. However, traditional methods for installing this group often suffer from harsh reaction conditions, limited substrate scope, or the use of hazardous reagents. The methodology described in CN110467579B circumvents these issues by employing readily available hydrazones and trifluoroethylimidoyl chlorides as building blocks. This approach not only simplifies the supply chain by relying on commodity chemicals but also ensures that the resulting high-purity OLED material or pharmaceutical precursor meets stringent regulatory standards for impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethylated nitrogen heterocycles has relied heavily on two primary strategies, both of which present significant logistical and economic challenges for cost reduction in pharmaceutical manufacturing. The first approach involves the direct trifluoromethylation of pre-synthesized triazole rings using specialized reagents such as Togni or Umemoto reagents; these reagents are notoriously expensive, thermally unstable, and often require stoichiometric amounts of precious metal catalysts. The second mainstream method utilizes trifluorodiazoethane, a highly reactive and potentially explosive synthon that demands rigorous safety protocols, specialized equipment, and strictly anhydrous conditions. Furthermore, many existing protocols rely on transition metal catalysis (e.g., copper or palladium), which introduces the risk of heavy metal contamination in the final Active Pharmaceutical Ingredient (API). Removing these trace metals to meet ICH Q3D guidelines necessitates additional purification steps, such as column chromatography or scavenger treatment, which drastically reduces overall yield and increases production lead time.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel iodine-promoted cyclization described in the patent offers a remarkably efficient and operationally simple alternative. The core innovation lies in the tandem use of sodium acetate and elemental iodine to drive the cyclization of trifluoroethylimidoyl chloride with hydrazones. As depicted in the general reaction scheme below, this method proceeds smoothly in common organic solvents like dichloroethane (DCE) at moderate temperatures (80°C). Crucially, the reaction does not require inert atmosphere techniques or rigorously dried solvents, significantly lowering the barrier to entry for commercial scale-up of complex pharmaceutical intermediates. The avoidance of transition metals means the crude product is inherently cleaner, reducing the burden on downstream purification teams and facilitating a more sustainable manufacturing process with a lower environmental footprint.
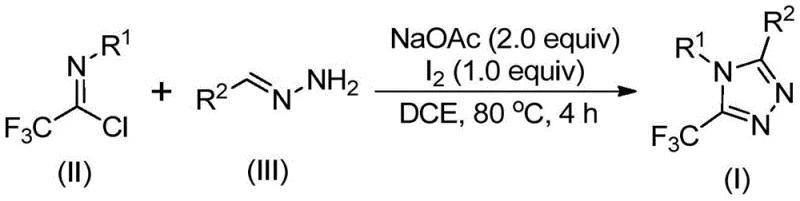
Mechanistic Insights into Iodine-Promoted Oxidative Cyclization
The mechanistic pathway of this transformation is a sophisticated example of base-promoted oxidative heterocyclization, offering R&D directors valuable insights into reaction control and impurity management. The process likely initiates with a nucleophilic attack by the hydrazone nitrogen on the electrophilic carbon of the trifluoroethylimidoyl chloride, facilitated by sodium acetate acting as a mild base. This step forms a transient trifluoroacetamidine intermediate via intermolecular carbon-nitrogen bond formation. Subsequent isomerization aligns the reactive centers for cyclization. The pivotal role of elemental iodine is then realized in the oxidative iodination step, where iodine acts as a mild oxidant to generate an iodo-intermediate. This species undergoes rapid intramolecular electrophilic substitution, followed by spontaneous aromatization and elimination of HI to yield the stable 5-trifluoromethyl-1,2,4-triazole core. Understanding this mechanism allows chemists to fine-tune reaction parameters, such as the molar ratio of iodine (optimized at 1.0 equivalent) and base (2.0 equivalents), to maximize conversion and minimize side products.
From an impurity control perspective, the mildness of the iodine promoter is advantageous compared to strong oxidants like peroxides or hypervalent iodine species, which can lead to over-oxidation or decomposition of sensitive functional groups. The patent data indicates high yields across a broad range of substrates, including those with electron-donating groups (methyl, methoxy) and electron-withdrawing groups (bromo, nitro, trifluoromethyl). For instance, heteroaryl substrates like thiophene-derived hydrazones achieved yields as high as 97%, demonstrating exceptional functional group tolerance. This robustness suggests that the reaction mechanism is resilient to steric and electronic variations, making it a versatile platform for synthesizing diverse libraries of triazole derivatives. The ability to predictably manage the reaction trajectory ensures consistent batch-to-batch quality, a critical factor for maintaining supply chain reliability in the production of high-purity pharmaceutical intermediates.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazoles Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemistry teams aiming to transition from discovery to pilot scale. The procedure involves a straightforward one-pot sequence where reagents are combined in a standard reactor, heated, and then treated with iodine in a second stage. This minimizes unit operations and handling time. Detailed standardized synthetic steps for implementing this route in your facility are provided in the guide below.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone substrates in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and maintain stirring for 2 to 4 hours to facilitate initial condensation.
- Introduce elemental iodine to the system and continue heating for an additional 1 to 2 hours to drive oxidative cyclization, followed by standard purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The elimination of expensive noble metal catalysts and hazardous diazo compounds directly impacts the cost of goods sold (COGS). By utilizing commodity chemicals like hydrazones and imidoyl chlorides, which are widely available from bulk chemical suppliers, companies can mitigate supply risks associated with specialized reagents. Furthermore, the operational robustness of the method—specifically its tolerance to ambient moisture and oxygen—reduces the need for specialized glassware or reactor conditioning, allowing for faster turnaround times between batches. This efficiency is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that production schedules remain agile and responsive to market demand fluctuations.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of transition metal catalysts. In traditional cross-coupling or trifluoromethylation reactions, the cost of palladium or copper catalysts, combined with the subsequent expense of metal scavengers and extensive filtration processes, can constitute a substantial portion of production costs. By switching to an iodine-promoted system, manufacturers eliminate these line items entirely. Additionally, the use of inexpensive sodium acetate as a base instead of stronger, more costly organic bases further drives down raw material expenses. The high yields reported (often exceeding 80-90%) mean less raw material waste and higher throughput per batch, resulting in substantial cost savings without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of starting materials. The precursors for this reaction, specifically the hydrazones and trifluoroethylimidoyl chlorides, are derived from common aldehydes and amines, which are produced on a massive global scale. Unlike trifluorodiazoethane, which requires on-site generation due to instability, these solid or liquid precursors can be sourced, stored, and transported safely. This stability simplifies inventory management and reduces the risk of production stoppages due to reagent degradation. Moreover, the reaction's insensitivity to moisture means that solvent drying systems do not need to be pushed to their limits, reducing energy consumption and maintenance downtime, thereby ensuring a more continuous and reliable supply of critical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this method is inherently designed for expansion. The patent explicitly notes the ease of scaling from gram to kilogram levels, indicating that heat transfer and mixing issues are minimal. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and waste disposal, aligning with increasingly strict global environmental regulations. The use of dichloroethane (DCE) is standard in the industry, and solvent recovery systems are well-established. By avoiding the generation of heavy metal sludge, facilities can significantly reduce their hazardous waste disposal costs and improve their overall sustainability profile, making it easier to meet corporate ESG (Environmental, Social, and Governance) goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl-triazole synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear framework for evaluating the technology's fit within your existing manufacturing capabilities.
Q: What are the primary advantages of this iodine-promoted method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive and toxic transition metal catalysts (such as palladium or copper), thereby removing the costly and complex heavy metal scavenging steps typically required in API manufacturing. Additionally, it operates under mild conditions without strict anhydrous or anaerobic requirements.
Q: What is the substrate scope for the R1 and R2 groups in this triazole synthesis?
A: The process demonstrates excellent functional group tolerance. R1 can be various substituted phenyl groups (methyl, methoxy, bromo, trifluoromethyl), while R2 accommodates alkenyl, aryl, and heteroaryl groups (such as thiophene or pyridine), allowing for diverse molecular scaffolding.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is easily scalable from gram-level laboratory synthesis to kilogram-level industrial production. The use of cheap, commercially available starting materials and simple post-treatment procedures supports robust commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We understand the critical nature of stringent purity specifications in the pharmaceutical sector and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of novel triazole derivatives or large-scale supply of established intermediates, our infrastructure is designed to support your most demanding projects with precision and reliability.
We invite you to leverage our technical capabilities to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data for reference standards and comprehensive route feasibility assessments to demonstrate how this iodine-promoted technology can enhance your production efficiency. Let us partner with you to deliver high-quality chemical solutions that drive your business forward.
