Advanced Dibenzofuran Phosphine Oxides for Low-Voltage High-Efficiency OLED Displays
The landscape of organic light-emitting diode (OLED) technology is constantly evolving, driven by the relentless demand for higher efficiency and lower power consumption in portable display terminals and lighting equipment. A pivotal advancement in this domain is documented in patent CN102898472A, which introduces a novel class of multifunctional modified dibenzofuryl bisphosphine oxide compounds. These materials address a critical bottleneck in phosphorescent organic light-emitting diodes (PHOLEDs): the incompatibility between maintaining a high triplet energy level and achieving sufficient carrier injection and transport capabilities. Traditionally, host materials with high triplet energies often suffer from wide energy gaps that impede charge transport, leading to excessively high driving voltages. The innovation presented here strategically modifies the 4,6-bis(diphenylphosphinooxy)dibenzofuran core to balance these competing properties, resulting in devices with significantly reduced start-up voltages.
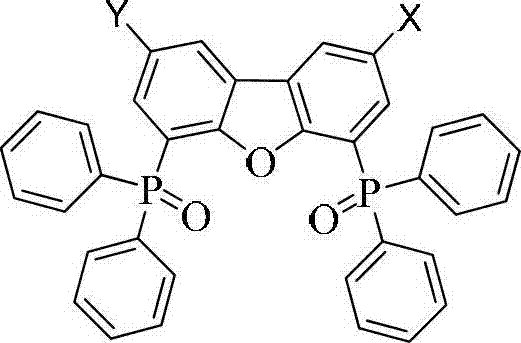
This breakthrough is particularly relevant for manufacturers seeking a reliable OLED material supplier capable of delivering next-generation host materials. The core structure, as illustrated in the patent, allows for versatile functionalization where substituents X and Y can be tailored to optimize electronic properties. By integrating groups known for superior carrier mobility onto a scaffold that inherently possesses a high triplet energy level (approximately 3.0 eV), the invention ensures efficient energy transfer from the host to the guest emitter while facilitating smooth charge flow. This dual functionality is the key to overcoming the limitations of previous generations of host materials, paving the way for more energy-efficient and stable display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the conventional fabrication of electrophosphorescent devices, the selection of host materials has always been a compromise. To prevent reverse energy transfer and ensure effective confinement of excitons within the guest emitter, the host material must possess a first triplet energy level (T1) higher than that of the guest, typically around 3.0 eV. However, molecules engineered to have such wide bandgaps frequently exhibit poor charge carrier transport properties. This deficiency forces device engineers to apply higher driving voltages to achieve the necessary current density for illumination. In many existing preparations, the start-up voltage of electroluminescent devices exceeds 5V, which is detrimental to battery life in portable electronics and increases heat generation. Furthermore, the lack of balanced hole and electron transport often leads to efficiency roll-off at high brightness levels, limiting the practical utility of these materials in commercial applications.
The Novel Approach
The novel approach detailed in CN102898472A circumvents these issues by employing a modular synthetic strategy centered on the 4,6-bis(diphenylphosphinooxy)dibenzofuran skeleton. This core is chemically robust and inherently possesses the requisite high triplet energy. The innovation lies in the subsequent functionalization steps where specific moieties, such as carbazole derivatives or aryl groups linked via boronic acid esters, are introduced at the 2 or 2,8 positions. This modification does not significantly lower the triplet energy of the core but drastically enhances the hole or electron injection capabilities. Consequently, the resulting multifunctional modified compounds enable devices to operate at start-up voltages as low as 2.4V. This represents a paradigm shift in host material design, moving from static wide-bandgap molecules to dynamic, multifunctional systems that actively participate in charge transport without sacrificing energy confinement.
Mechanistic Insights into Suzuki-Miyaura and Ullmann-Type Couplings
The synthesis of these advanced materials relies on precise transition-metal catalyzed cross-coupling reactions, specifically leveraging the reactivity of brominated intermediates. The process begins with the electrophilic aromatic substitution of the parent dibenzofuran derivative. As shown in the structural analysis of the intermediates, the introduction of bromine atoms is critical for enabling subsequent bond formation.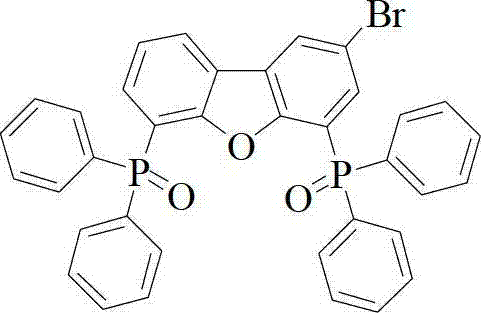 The bromination is carefully controlled using N-bromosuccinimide (NBS) in concentrated sulfuric acid at room temperature. This mild condition prevents the degradation of the sensitive phosphine oxide groups while ensuring regioselective substitution at the activated positions of the dibenzofuran ring. The resulting 2-bromo or 2,8-dibromo intermediates serve as versatile electrophiles for the second stage of the synthesis.
The bromination is carefully controlled using N-bromosuccinimide (NBS) in concentrated sulfuric acid at room temperature. This mild condition prevents the degradation of the sensitive phosphine oxide groups while ensuring regioselective substitution at the activated positions of the dibenzofuran ring. The resulting 2-bromo or 2,8-dibromo intermediates serve as versatile electrophiles for the second stage of the synthesis.
In the second stage, the patent outlines two distinct pathways depending on the desired functional group. For aryl substitutions, a Suzuki-Miyaura coupling is employed using functionalized boronic acid pinacol esters, tetrakis(triphenylphosphine)palladium, and a base like sodium hydroxide in tetrahydrofuran at 85°C to 95°C. Alternatively, for nitrogen-containing heterocycles like carbazole, an Ullmann-type condensation is utilized involving cuprous iodide, 18-crown-6, and potassium carbonate at higher temperatures of 170°C to 200°C. These mechanistic pathways allow for the fine-tuning of the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) levels. By selecting specific coupling partners, chemists can align the energy levels of the host with the adjacent charge transport layers, thereby reducing the injection barrier and minimizing the operating voltage of the final OLED device.
How to Synthesize Multifunctional Modified Dibenzofuryl Bisphosphine Oxide Efficiently
The preparation method described in the patent offers a robust route for producing these high-performance host materials with high purity and yield. The process is designed to be scalable, utilizing readily available reagents and standard laboratory equipment suitable for pilot and commercial production. The initial bromination step is straightforward, requiring only careful control of stoichiometry to achieve mono- or di-substitution as needed. The subsequent coupling reactions are well-established in organic synthesis, ensuring reproducibility. For those interested in the specific operational parameters, stoichiometry, and workup procedures required to replicate this synthesis in a GMP environment, the detailed standardized synthesis steps are provided in the guide below.
- Dissolve 4,6-bis(diphenylphosphinooxy)dibenzofuran in concentrated sulfuric acid and react with N-bromosuccinimide at room temperature for 2-6 hours to obtain brominated intermediates.
- Perform a coupling reaction using the brominated intermediate with functionalized boronic acid pinacol esters or carbazole derivatives under palladium or copper catalysis at elevated temperatures.
- Purify the final multifunctional modified dibenzofuryl bisphosphine oxide compound using column chromatography with ethyl acetate as the eluent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers significant strategic advantages beyond mere technical performance. The synthesis route avoids the use of exotic or highly unstable reagents, relying instead on commodity chemicals like NBS, sulfuric acid, and common palladium or copper catalysts. This reliance on established chemistry translates directly into cost reduction in electronic chemical manufacturing. By eliminating the need for complex, multi-step protection-deprotection sequences often required in other high-triplet energy hosts, the overall process mass intensity is reduced. Furthermore, the high thermal stability of the final products (decomposition temperatures up to 544°C) implies a longer shelf life and reduced risk of degradation during storage and transport, enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The synthetic pathway is streamlined, utilizing direct bromination and one-pot coupling reactions that minimize solvent usage and purification steps. The elimination of expensive transition metal scavengers, often required when using less stable catalysts, further drives down the cost of goods sold. Additionally, the high thermal stability reduces waste associated with material degradation, contributing to substantial cost savings in the long run.
- Enhanced Supply Chain Reliability: The precursors, such as 4,6-bis(diphenylphosphinooxy)dibenzofuran and various boronic esters, are commercially accessible or easily synthesized from bulk petrochemical feedstocks. This reduces dependency on single-source specialty suppliers and mitigates the risk of raw material shortages. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in environmental controls, ensuring consistent batch-to-batch quality.
- Scalability and Environmental Compliance: The reactions are conducted in common solvents like tetrahydrofuran and dichloromethane, which have well-established recovery and recycling protocols in modern chemical plants. The absence of highly toxic heavy metals in the final product structure simplifies waste disposal and regulatory compliance. The process is inherently scalable from gram to kilogram quantities without significant re-optimization, facilitating rapid commercial scale-up of complex OLED materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these multifunctional dibenzofuran derivatives in OLED production lines. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: How does this new material reduce the start-up voltage of OLED devices?
A: By attaching groups with excellent carrier injection and transport capabilities to the 4,6-bis(diphenylphosphinooxy)dibenzofuran core, the material maintains a high triplet energy level while significantly improving charge mobility, lowering the start-up voltage to approximately 2.4V.
Q: What is the thermal stability of these phosphine oxide derivatives?
A: The multifunctional modified dibenzofuryl bisphosphine oxides exhibit exceptional thermodynamic stability, with decomposition temperatures ranging from 398°C to 544°C, ensuring robust performance during device operation and vacuum deposition.
Q: Can these materials be used for different color emissions in PHOLEDs?
A: Yes, the patent demonstrates successful application as host materials for blue, green, yellow, and red electrophosphorescent devices by doping with appropriate iridium complexes, showing versatile compatibility across the visible spectrum.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-bis(diphenylphosphinooxy)dibenzofuran Derivative Supplier
The transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying the stringent purity specifications required for electronic grade materials. We understand that even trace impurities can quench phosphorescence or alter charge transport, so our purification protocols are designed to meet the highest standards of the optoelectronic industry.
We invite you to collaborate with us to evaluate the feasibility of integrating these low-voltage host materials into your product roadmap. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and device architecture. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate the development of high-efficiency, low-power OLED displays that define the future of visual technology.
