Advanced Purification Technology for High-Purity (E,Z)-7,9-Dodecadienyl Acetate Production
Advanced Purification Technology for High-Purity (E,Z)-7,9-Dodecadienyl Acetate Production
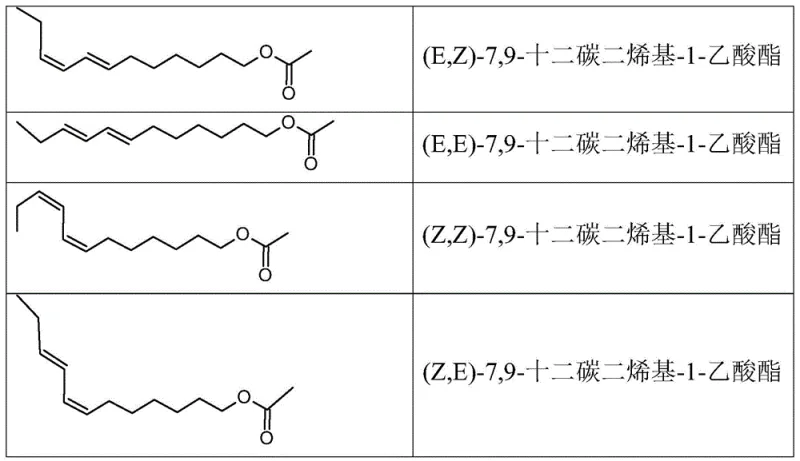
The global demand for environmentally sustainable pest control solutions has intensified the focus on semiochemicals, specifically sex pheromones for mating disruption strategies. Patent CN110891957A introduces a groundbreaking methodology for the preparation of 7,9-dodecadienyl-1-acetate isomers, specifically targeting the economically vital (E,Z) isomer used against the European grapevine moth, Lobesia botrana. Traditional synthetic routes often struggle with the co-formation of the thermodynamically stable but biologically inactive (E,E) isomer, which significantly dilutes the potency of the final agrochemical formulation. This patent discloses a novel two-step process that leverages a selective Diels-Alder reaction to chemically sequester and remove the unwanted (E,E) impurity, achieving an unprecedented isomeric purity of greater than 98% for the active (E,Z) component. For procurement specialists and R&D directors in the agrochemical sector, this technology represents a pivotal shift towards more efficient, cost-effective, and high-performance pheromone manufacturing capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lepidopteran pheromones containing conjugated diene systems has been plagued by stereochemical challenges. Conventional approaches, such as those utilizing Wittig reactions or copper-catalyzed couplings, frequently result in mixtures where the desired (E,Z) isomer constitutes only 70% to 75% of the total mass, with the (E,E) isomer appearing as a major contaminant in proportions of 20% to 30%. Previous attempts to purify these mixtures, such as urea inclusion complexation, have proven to be operationally tedious, requiring multiple crystallization cycles that still fail to reduce the (E,E) content below 5%. Furthermore, these traditional purification methods often lead to significant product degradation and yield losses, rendering the final active ingredient prohibitively expensive for widespread agricultural deployment. The reliance on chromatographic separation for high purity is economically unviable for bulk agrochemical intermediates, creating a bottleneck in the supply chain for effective mating disruption products.
The Novel Approach
The methodology described in CN110891957A circumvents these historical bottlenecks by introducing a chemical purification strategy rather than a physical one. By reacting the crude mixture of enol phosphate intermediates with a hydrolyzable dienophile, such as maleic anhydride, the process selectively targets the (E,E) isomer for a Diels-Alder cycloaddition. This reaction is remarkably specific; the active (E,Z), (Z,E), and (Z,Z) isomers remain largely unreacted under the optimized conditions. The resulting Diels-Alder adduct of the (E,E) isomer is then subjected to alkaline hydrolysis, converting it into a water-soluble salt that can be effortlessly removed via simple aqueous extraction. This elegant solution not only eliminates the need for complex chromatography but also preserves the integrity of the valuable active isomers, ensuring that the subsequent coupling steps proceed with maximum efficiency and minimal waste of expensive Grignard reagents.
Mechanistic Insights into Selective Diels-Alder Purification
The core innovation lies in the differential reactivity of the geometric isomers towards the dienophile. While all isomers possess a conjugated diene system, the spatial arrangement of the (E,E) isomer renders it uniquely susceptible to the cycloaddition under the specific thermal conditions employed (typically around 70°C). The reaction mechanism involves the [4+2] cycloaddition where the diene system of the enol phosphate reacts with the electron-deficient double bond of the maleic anhydride. Crucially, the patent data indicates that this reaction does not merely separate the isomers physically but chemically transforms the impurity into a distinct chemical entity. Following the cycloaddition, the addition of a basic aqueous solution, such as sodium hydroxide, hydrolyzes the anhydride moiety of the adduct. This hydrolysis generates a dicarboxylate salt species which exhibits high polarity and water solubility, contrasting sharply with the lipophilic nature of the unreacted enol phosphate isomers.
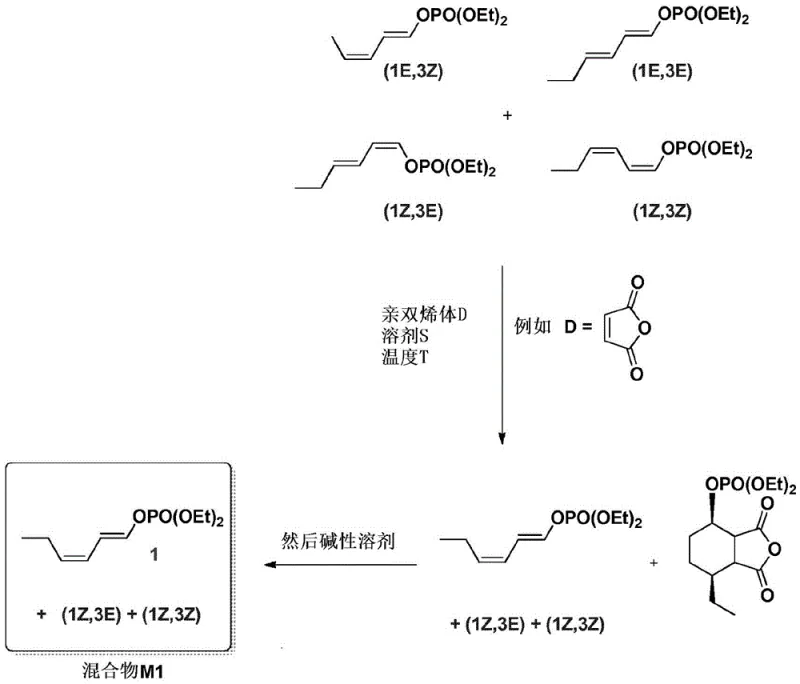
This phase separation allows for the recovery of the organic phase containing the purified mixture M1, which is now enriched to over 98% in the desired (E,Z) configuration relative to the total diene content. The preservation of the minor (Z,E) and (Z,Z) isomers is also maintained, which is beneficial as some studies suggest these minor components do not hinder the attractiveness of the pheromone blend. From a process chemistry perspective, this mechanism avoids the use of heavy metal catalysts or cryogenic conditions often required for high stereoselectivity in direct synthesis. The robustness of this chemical scavenging method ensures that variations in the initial feedstock quality can be corrected mid-process, providing a reliable buffer against batch-to-batch variability in the upstream synthesis of the enol phosphate precursors.
How to Synthesize (E,Z)-7,9-Dodecadienyl Acetate Efficiently
The implementation of this technology involves a streamlined workflow designed for industrial scalability. The process begins with the preparation of the enol phosphate intermediate, typically derived from hex-2-enal, which is then subjected to the selective purification protocol described above. Once the (E,E) impurity is scrubbed from the mixture to yield M1, the purified intermediate undergoes an iron-catalyzed cross-coupling reaction with a Grignard reagent, specifically a magnesium alkoxide derived from 6-chloro-hexan-1-ol. This coupling step is highly efficient due to the absence of the (E,E) isomer, which would otherwise consume the Grignard reagent unproductively. The final step involves acetylation to generate the target acetate ester. Detailed operational parameters, including solvent choices like methylcyclohexane and specific temperature gradients, are critical for maintaining the high isomeric ratio throughout the synthesis.
- React crude enol phosphate mixture with a hydrolyzable dienophile like maleic anhydride at elevated temperatures to selectively trap the unwanted (E,E) isomer.
- Perform alkaline hydrolysis to convert the Diels-Alder adduct into a water-soluble salt, allowing for easy separation via aqueous extraction.
- Couple the purified (E,Z)-enriched enol phosphate with a Grignard reagent using an iron catalyst, followed by acetylation to yield the final pheromone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology offers substantial strategic advantages beyond mere chemical purity. The primary benefit is the drastic simplification of the downstream processing unit operations. By replacing energy-intensive and solvent-heavy chromatographic purifications or repetitive crystallizations with a single reactive extraction step, the overall manufacturing footprint is significantly reduced. This reduction in processing complexity translates directly into lower operational expenditures and a reduced environmental burden, aligning with modern green chemistry mandates. Furthermore, the ability to tolerate a cruder initial feedstock without compromising final quality provides greater flexibility in sourcing raw materials, thereby mitigating supply chain risks associated with high-purity precursor availability.
- Cost Reduction in Manufacturing: The elimination of the (E,E) isomer prior to the coupling step prevents the wasteful consumption of expensive organometallic reagents. In conventional processes, up to 25% of the Grignard reagent is wasted reacting with the inactive (E,E) impurity. By removing this impurity beforehand, the atom economy of the coupling step is maximized, leading to significant savings in reagent costs. Additionally, the avoidance of column chromatography removes a major cost center related to silica gel disposal and solvent recovery, further enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the Diels-Alder purification step ensures consistent output quality even when input material quality fluctuates. This reliability is crucial for maintaining uninterrupted supply contracts with agrochemical formulators who require strict adherence to specification limits. The process uses commodity chemicals like maleic anhydride and standard organic solvents, reducing dependency on specialized or scarce reagents that could cause supply bottlenecks. This stability allows for more accurate forecasting and inventory management, ensuring that critical pest control seasons are supported by adequate stock levels of the active pheromone ingredient.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in reactor sizes suitable for commercial production without the need for specialized equipment. The aqueous waste stream generated from the hydrolysis step contains water-soluble salts that are easier to treat compared to the complex organic waste streams generated by phosphine oxide byproducts from Wittig reactions. This simplifies wastewater treatment compliance and reduces the overall environmental impact of the manufacturing site. The high yield and purity achieved in fewer steps also mean less energy consumption per kilogram of product, contributing to a lower carbon footprint for the final agrochemical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel purification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial pheromone synthesis. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: Why is the (E,E) isomer considered a critical impurity in grape moth pheromones?
A: The (E,E) isomer is thermodynamically stable but biologically inactive for Lobesia botrana control. Its presence dilutes the efficacy of mating disruption dispensers, requiring higher application rates and increasing costs.
Q: How does the Diels-Alder purification method improve yield compared to urea inclusion?
A: Unlike urea inclusion which suffers from product degradation and limited purity ceilings, the Diels-Alder method chemically modifies the impurity into a water-soluble form, preserving the active (E,Z) isomer and preventing yield loss during separation.
Q: Is this purification method scalable for industrial agrochemical production?
A: Yes, the process utilizes standard organic solvents like methylcyclohexane and common reagents like maleic anhydride, avoiding complex chromatography or cryogenic conditions, making it highly suitable for multi-ton commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7,9-Dodecadienyl-1-Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation sustainable agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated purification techniques described in CN110891957A can be seamlessly translated from laboratory bench to industrial reactor. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced gas chromatography systems to verify isomeric ratios, guaranteeing that every batch of 7,9-dodecadienyl-1-acetate meets the exacting standards required for effective mating disruption dispensers. Our commitment to quality assurance ensures that our clients receive a product that maximizes field efficacy while minimizing application rates.
We invite potential partners to engage with our technical procurement team to discuss how this advanced purification technology can be tailored to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this high-efficiency process for your pheromone portfolio. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both performance and profitability in your agrochemical manufacturing operations. Let us collaborate to secure a sustainable and cost-effective supply of this vital pest control intermediate.
