Advanced Synthesis of 4-Iodo-3-Nitrobenzamide for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical antineoplastic intermediates, and patent CN103274946A presents a significant advancement in the preparation of 4-iodo-3-nitrobenzamide. This compound serves as a pivotal building block for poly ADP-ribose polymerase (PARP) inhibitors like AG14361, which are currently in clinical studies for cancer treatment. The disclosed methodology addresses long-standing challenges in medicinal chemistry by offering a pathway that drastically enhances both productive rate and product quality. By leveraging a specific sequence of cyanation, diazotization-iodination, and hydrolysis, this technology provides a reliable foundation for high-purity pharmaceutical intermediates. For R&D directors and procurement specialists, understanding this patent is crucial for securing a stable supply of high-value chemical entities. The process utilizes standard industrial reagents, making it an attractive candidate for commercial scale-up and cost-effective manufacturing in the competitive fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex nitrobenzamide derivatives has been plagued by significant technical hurdles that impede efficient commercial production. Conventional methods often suffer from poor reaction selectivity, leading to the formation of numerous by-products that complicate downstream purification processes. These impurities not only lower the overall yield but also pose risks to the safety profile of the final pharmaceutical product. Furthermore, traditional routes frequently require harsh reaction conditions or expensive catalysts that are difficult to remove, resulting in elevated manufacturing costs and extended production timelines. The low purity associated with these older methods necessitates multiple recrystallization steps, which further erodes material throughput and increases waste generation. For supply chain managers, these inefficiencies translate into unpredictable lead times and higher procurement costs, making the reliable sourcing of such intermediates a persistent challenge in the global market.
The Novel Approach
In stark contrast to legacy techniques, the novel approach detailed in the patent introduces a streamlined three-step sequence that optimizes both chemical efficiency and operational simplicity. The process begins with a highly selective nitrile grouping reaction, followed by a controlled diazotization-iodo-elimination, and concludes with a high-yield hydrolysis step. This strategic design allows for precise control over reaction parameters, such as temperature and molar ratios, ensuring consistent product quality batch after batch. The use of commercially available solvents like DMF and water simplifies the procurement of raw materials, reducing dependency on specialized supply chains. By achieving yields as high as 90.1% in the final step, this method significantly maximizes material utilization, directly contributing to substantial cost savings in large-scale manufacturing. This robust methodology represents a paradigm shift towards more sustainable and economically viable production of critical oncology intermediates.
Mechanistic Insights into CuCN-Catalyzed Cyanation and Hydrolysis
The core of this synthetic strategy lies in the initial Rosenmund-von Braun type reaction where a bromine substituent is replaced by a cyano group using cuprous cyanide. This transformation is critical as it sets the stage for subsequent functionalization while maintaining the integrity of the nitro group. The reaction is typically conducted in polar aprotic solvents like DMF at temperatures ranging from 80°C to 90°C, which facilitates the nucleophilic substitution without degrading the sensitive aromatic structure. Careful control of the molar ratio between the starting aniline derivative and cuprous cyanide is essential to minimize the formation of homocoupling by-products. Following this, the amino group is converted to an iodo substituent via a diazonium salt intermediate, a classic transformation that requires precise temperature control to prevent decomposition. 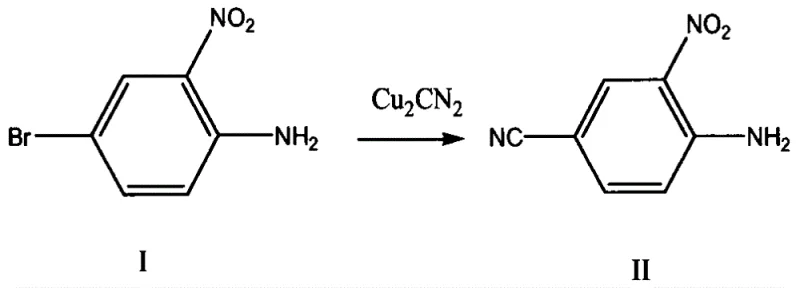
The final mechanistic step involves the acid-catalyzed hydrolysis of the nitrile group to the corresponding primary amide, a transformation that is often kinetically slow but is accelerated here under optimized conditions. By employing concentrated hydrochloric acid at elevated temperatures, the reaction proceeds to completion with minimal formation of carboxylic acid by-products, which are common impurities in nitrile hydrolysis. The mechanism likely proceeds through an imidate intermediate that is rapidly hydrolyzed to the amide, driven by the excess of water and acid. This step is particularly noteworthy for its high yield of 90.1%, indicating that the reaction equilibrium strongly favors the product under the specified conditions. For quality control teams, understanding these mechanistic nuances is vital for establishing effective in-process controls that ensure the impurity profile remains within strict pharmacopeial limits. 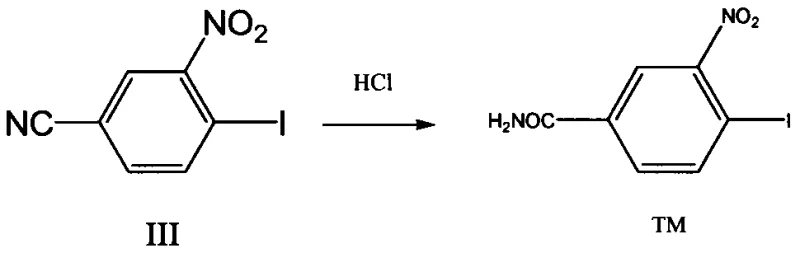
How to Synthesize 4-Iodo-3-Nitrobenzamide Efficiently
Implementing this synthesis route requires strict adherence to the optimized reaction conditions outlined in the patent to ensure maximum efficiency and safety. The process is designed to be scalable, utilizing standard reactor equipment and common chemical reagents that are easily sourced from global suppliers. Operators must focus on maintaining precise temperature profiles during the diazotization step to ensure safety and product consistency. The detailed standardized synthesis steps, including specific work-up procedures and purification methods, are provided in the guide below to facilitate technology transfer.
- Perform a nitrile grouping reaction on 4-bromo-2-N-methyl-p-nitroaniline with cuprous cyanide in an organic solvent like DMF at 80-90°C.
- Conduct a diazotization-iodo-elimination reaction on the intermediate using sodium nitrite and potassium iodide in aqueous acid at 20-30°C.
- Hydrolyze the resulting nitrile intermediate using concentrated hydrochloric acid at 80-90°C to obtain the final 4-iodo-3-nitrobenzamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement and supply chain management in the fine chemical industry. The elimination of complex catalytic systems and the use of abundant raw materials significantly reduce the risk of supply disruptions, ensuring a steady flow of intermediates for downstream drug manufacturing. The high yields achieved at each step translate to lower raw material consumption per kilogram of product, which is a primary driver for cost reduction in chemical manufacturing. Furthermore, the simplified purification requirements reduce the demand for expensive chromatography resins or extensive solvent exchanges, lowering the overall operational expenditure. For supply chain heads, the robustness of this method means reduced lead times and greater predictability in delivery schedules, which is critical for just-in-time manufacturing models.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by maximizing material throughput through high-yield reaction steps, particularly the final hydrolysis which exceeds 90% efficiency. By avoiding the use of precious metal catalysts that require expensive recovery or removal processes, the overall cost of goods sold is drastically reduced. The use of common solvents like DMF and water further minimizes procurement costs compared to specialized proprietary solvents. Additionally, the reduced formation of by-products lowers the cost associated with waste treatment and disposal, contributing to a leaner manufacturing budget. These factors combine to create a highly competitive cost structure for the production of this valuable pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly enhanced because all reagents and starting materials specified in the patent are commercially available from multiple global vendors. This diversity in sourcing options mitigates the risk of single-supplier dependency, which is a common vulnerability in the pharmaceutical supply chain. The reaction conditions are not overly sensitive to minor fluctuations in raw material quality, allowing for greater flexibility in vendor selection. Consequently, procurement managers can negotiate better terms and secure long-term contracts with confidence, knowing that production will not be halted due to material shortages. This reliability is essential for maintaining continuous production schedules for critical antineoplastic agents.
- Scalability and Environmental Compliance: The synthesis route is inherently scalable, having been demonstrated in multi-gram to kilogram scales with consistent results, making it suitable for commercial tonnage production. The process avoids the generation of hazardous heavy metal waste, as the copper residues can be managed through standard wastewater treatment protocols. The use of water as a solvent in the diazotization step aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental regulations simplifies the permitting process for new production facilities and reduces the risk of regulatory shutdowns. For companies aiming to expand capacity, this technology offers a clear path to increasing output without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of 4-iodo-3-nitrobenzamide based on the patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders make informed decisions about process adoption and supplier qualification.
Q: What are the key advantages of the patent CN103274946A synthesis route?
A: The patent describes a method that significantly improves yield and purity compared to conventional methods, utilizing commercially available reagents and manageable reaction conditions suitable for scale-up.
Q: What is the overall yield potential for this synthesis pathway?
A: The patent data indicates high individual step yields, with the final hydrolysis step achieving over 90% yield, suggesting a highly efficient overall process for commercial manufacturing.
Q: Are the raw materials for this process readily available?
A: Yes, the patent explicitly states that the reagents and raw materials used, such as cuprous cyanide and standard organic solvents, are commercially available, ensuring supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Iodo-3-Nitrobenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antineoplastic therapies. Our team of expert chemists has extensively evaluated the route described in patent CN103274946A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of 4-iodo-3-nitrobenzamide meets the highest industry standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions to optimize your drug development pipeline.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to this optimized route can improve your bottom line. Please contact us to request specific COA data and route feasibility assessments tailored to your volume needs. By partnering with us, you gain access to a secure supply chain and a wealth of chemical expertise dedicated to your success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
