Scalable Preparation of High-Purity 17 Alpha-Pregnenolone for Pharmaceutical Quality Control
Scalable Preparation of High-Purity 17 Alpha-Pregnenolone for Pharmaceutical Quality Control
The pharmaceutical landscape for neuroactive steroids has shifted dramatically with the approval of allopregnanolone (Zulresso®) for postpartum depression, creating an urgent demand for rigorous impurity profiling and high-quality reference standards. As regulatory agencies enforce stricter limits on related substances, typically requiring known impurities to be controlled below 0.15%, the ability to synthesize authentic standards like 17 alpha-pregnenolone becomes a critical bottleneck for quality control laboratories. The patent CN114031660A discloses a groundbreaking preparation method that bypasses traditional chromatographic limitations, offering a robust chemical route to this challenging stereoisomer. This technical insight analyzes the novel derivatization strategy that enables the production of 17 alpha-pregnenolone with purity exceeding 99.80%, providing a viable solution for reliable pharmaceutical intermediate supplier networks seeking to support the growing allopregnanolone market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
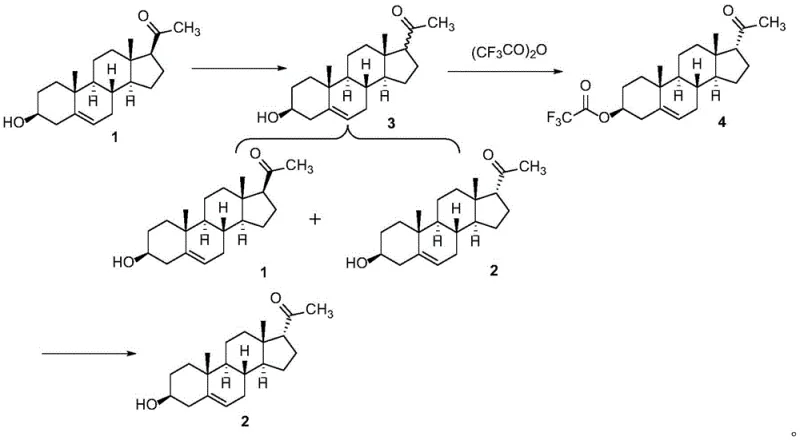
In the conventional production of allopregnanolone, the formation of 17 alpha-pregnenolone as a process-related impurity is inevitable due to the stereochemical similarity between the 17-alpha and 17-beta configurations. Historically, isolating this specific impurity for use as a reference marker has relied heavily on preparative chiral high-performance liquid chromatography (HPLC). While effective on a milligram scale for analytical method development, chiral HPLC presents severe scalability issues for commercial manufacturing. The technique is characterized by low sample loading capacities, excessive consumption of expensive chiral stationary phases, and high volumes of organic solvents, leading to prohibitive operational costs. Furthermore, the physical properties of 17 alpha-pregnenolone and its precursor pregnenolone are so similar that standard recrystallization or normal phase chromatography fails to achieve the necessary resolution, leaving manufacturers dependent on inefficient separation technologies that cannot meet the demands of cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative approach detailed in the patent data utilizes a clever chemical derivatization strategy to break the symmetry of the separation problem. Instead of attempting to separate the two stereoisomers directly, the process converts the mixture into a derivative where the steric environment of the 17-hydroxyl group dictates reactivity. By reacting the isomeric mixture with trifluoroacetic anhydride, the process selectively targets the 17-alpha hydroxyl group to form a trifluoroacetate ester (Compound 4), while the 17-beta isomer reacts differently or remains separable due to the bulkiness of the trifluoroacetyl group. This chemical modification introduces a significant difference in polarity and crystallization behavior, allowing for the isolation of the derivative in high purity. Subsequent mild hydrolysis regenerates the free alcohol, yielding the target 17 alpha-pregnenolone without the need for chiral columns, thereby drastically simplifying the workflow and enhancing supply chain reliability for high-purity steroid intermediates.
Mechanistic Insights into Selective Acylation and Hydrolysis
The core of this synthesis lies in the differential reactivity of the stereoisomers during the acylation step. When the mixture of pregnenolone (Compound 1) and 17 alpha-pregnenolone (Compound 2) is treated with trifluoroacetic anhydride (TFAA) in the presence of a catalyst like 4-dimethylaminopyridine (DMAP) and an acid scavenger like triethylamine, the reaction kinetics favor the formation of the 17-alpha-trifluoroacetate. The 17-alpha configuration places the hydroxyl group in a less sterically hindered environment compared to the 17-beta isomer, or alternatively, the resulting 17-alpha-ester possesses distinct crystallization properties that allow it to be separated from the reaction matrix. The use of dichloromethane as a solvent at low temperatures (0 to 10°C) further controls the reaction selectivity, minimizing side reactions and ensuring that the trifluoroacetyl group is installed cleanly. This step effectively acts as a chemical filter, converting the difficult-to-separate alcohol mixture into a separable ester derivative.
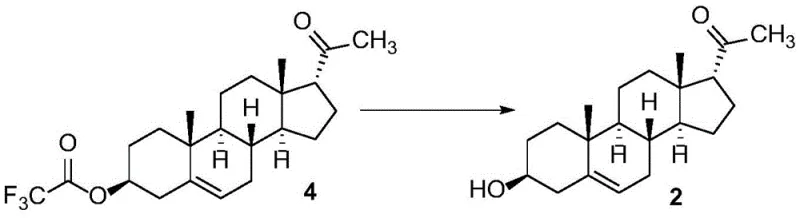
The final and perhaps most critical mechanistic step is the selective hydrolysis of Compound 4 back to the alcohol. The patent data highlights a surprising discovery regarding the choice of base for this deprotection. While strong bases like sodium hydroxide or sodium methoxide might seem intuitive for ester hydrolysis, they pose a risk of epimerizing the sensitive steroid backbone or causing elimination reactions. The inventors found that using a weak base, specifically sodium acetate, in a protic solvent like ethanol or methanol under reflux conditions provides the perfect balance of reactivity and selectivity. Sodium acetate is sufficiently nucleophilic to cleave the activated trifluoroacetate ester but mild enough to preserve the stereochemical integrity of the 17-alpha center. This specific catalytic condition prevents the reformation of the 17-beta isomer (Compound 1), ensuring that the final product is obtained with a purity of greater than 99.80%, a level of quality essential for accurate impurity quantification in drug substance testing.
How to Synthesize 17 Alpha-Pregnenolone Efficiently
The synthesis protocol outlined in the patent offers a streamlined pathway that integrates isomerization, derivatization, and hydrolysis into a cohesive manufacturing process suitable for scale-up. The procedure begins with the base-catalyzed equilibration of commercially available pregnenolone to generate the requisite mixture of isomers, followed by the selective protection and final deprotection steps. This route eliminates the dependency on specialized chromatographic equipment, relying instead on standard unit operations such as reflux, filtration, and crystallization. For process chemists looking to implement this technology, the detailed standardized synthetic steps see the guide below.
- Perform base-catalyzed isomerization of pregnenolone (Compound 1) in methanol to generate a mixture of stereoisomers (Mixture 3).
- React Mixture 3 with trifluoroacetic anhydride in the presence of DMAP and triethylamine to selectively form the trifluoroacetate derivative (Compound 4).
- Hydrolyze Compound 4 using sodium acetate in ethanol or methanol under reflux to selectively recover pure 17 alpha-pregnenolone (Compound 2).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chromatographic separation to chemical synthesis represents a significant strategic advantage in securing the supply of critical reference standards. The traditional reliance on chiral HPLC creates a fragile supply chain vulnerable to column shortages, solvent price volatility, and long lead times associated with low-throughput processing. By adopting this chemical derivatization route, manufacturers can leverage existing reactor infrastructure to produce large batches of 17 alpha-pregnenolone, significantly reducing lead time for high-purity pharmaceutical intermediates. The ability to produce kilogram quantities through simple filtration and crystallization, rather than iterative chromatographic runs, ensures a consistent and reliable supply that can scale with the growing demand for allopregnanolone formulations.
- Cost Reduction in Manufacturing: The elimination of chiral stationary phases and the reduction in solvent usage directly translate to substantial cost savings. Chromatographic methods require expensive resins that degrade over time and vast amounts of high-grade solvents for mobile phases. In contrast, the reagents used in this chemical route, such as trifluoroacetic anhydride and sodium acetate, are commodity chemicals available at competitive prices. Furthermore, the high yield reported in the examples, reaching up to 93.3% in the hydrolysis step, minimizes material loss, ensuring that the cost per gram of the final reference standard is drastically lower than chromatographically purified material.
- Enhanced Supply Chain Reliability: Dependence on complex purification technologies often introduces single points of failure in the supply chain. If a specific chiral column is discontinued or backordered, production halts. This chemical synthesis route relies on robust, well-understood reactions that can be performed in any standard multipurpose pharmaceutical plant. The use of common solvents like methanol, ethanol, and dichloromethane ensures that raw material availability is never a bottleneck. This robustness allows suppliers to maintain safety stock and respond quickly to urgent requests for impurity standards from regulatory bodies or internal QC labs, thereby stabilizing the overall supply chain for neuroactive steroid products.
- Scalability and Environmental Compliance: From an environmental and safety perspective, moving away from large-volume chromatography reduces the facility's solvent footprint and waste generation. Crystallization and filtration generate solid waste that is often easier to handle and dispose of than liquid chromatographic fractions laden with complex solvent mixtures. The process described operates at atmospheric pressure and moderate temperatures, reducing energy consumption compared to high-pressure liquid chromatography systems. This alignment with green chemistry principles not only lowers operational costs but also simplifies regulatory compliance regarding waste disposal and emissions, making the commercial scale-up of complex steroid intermediates more sustainable and economically viable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and technical stakeholders.
Q: Why is traditional chiral HPLC unsuitable for large-scale isolation of 17 alpha-pregnenolone?
A: Traditional chiral HPLC is effective for analytical separation but suffers from low loading capacity, high solvent consumption, and significant operational costs, making it economically unviable for industrial mass production of reference standards.
Q: How does the selective acylation strategy improve purity compared to direct crystallization?
A: Direct crystallization fails because 17 alpha-pregnenolone and pregnenolone are stereoisomers with nearly identical physical properties. Selective acylation creates a chemical difference (trifluoroacetate ester) that allows for easy separation before converting back to the target alcohol.
Q: What represents the critical control point for achieving >99.8% purity in this synthesis?
A: The hydrolysis step using sodium acetate is critical. Unlike stronger bases like sodium hydroxide which may cause re-isomerization or degradation, sodium acetate provides mild conditions that selectively cleave the trifluoroacetate group without affecting the steroid backbone stereochemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17 Alpha-Pregnenolone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the availability of high-purity impurity standards is foundational to the safety and efficacy of modern pharmaceuticals. Our team of expert process chemists has thoroughly evaluated the route disclosed in CN114031660A and confirmed its potential for seamless technology transfer. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for method validation or kilograms for stability studies, our stringent purity specifications and rigorous QC labs can deliver material that meets the highest global regulatory standards. Our commitment to technical excellence ensures that every batch of 17 alpha-pregnenolone we supply is fully characterized and ready for immediate use in your quality control workflows.
We invite you to collaborate with us to optimize your supply chain for neuroactive steroid intermediates. By leveraging our manufacturing capabilities, you can secure a stable source of this critical reference standard while achieving significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our production capabilities can support your long-term strategic goals in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
