Advanced Asymmetric Synthesis of Chiral 1,3-Alkamine Intermediates for Commercial Dapoxetine Production
The pharmaceutical landscape for treating premature ejaculation has been significantly shaped by the introduction of Dapoxetine, a short-acting selective serotonin reuptake inhibitor (SSRI). As demand for this active pharmaceutical ingredient (API) continues to grow globally, the efficiency of its supply chain relies heavily on the availability of high-quality chiral intermediates. Patent CN101875666B introduces a groundbreaking methodology for the preparation of optically pure 1,3-alkamine compounds, which serve as critical precursors in the synthesis of Dapoxetine and its analogues. This technology addresses long-standing challenges in stereochemical control, offering a robust alternative to traditional resolution techniques that have historically plagued the manufacturing of chiral amines. By leveraging chiral sulfinamide auxiliaries, this process ensures high stereoselectivity while maintaining operational simplicity, making it an attractive option for industrial applications.
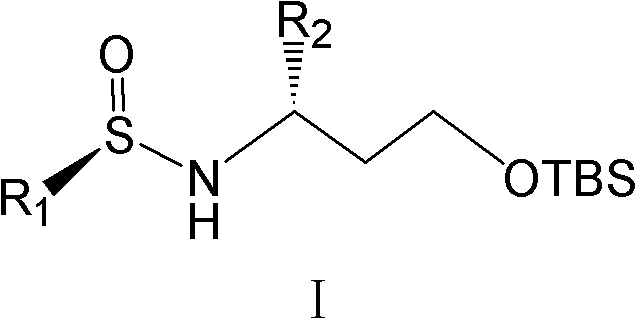
For R&D directors and process chemists, the structural integrity and purity of intermediates are paramount. The compounds described in this patent, represented generally by Formula I, feature a specific stereochemical configuration that is essential for the biological activity of the final drug product. The ability to synthesize these molecules with an enantiomeric excess (ee) exceeding 99% eliminates the need for costly and wasteful chiral separation steps later in the process. This level of precision not only streamlines the synthetic route but also ensures consistent quality across batches, a critical factor for regulatory compliance in pharmaceutical manufacturing. The versatility of the substituents allowed on the aromatic ring further expands the utility of this platform, enabling the rapid development of diverse analogue libraries for structure-activity relationship (SAR) studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure 1,3-amino alcohols like (S)-3-amino-3-phenylpropanol has relied heavily on the resolution of racemic mixtures. Traditional methods often employ chiral resolving agents such as L-(+)-tartaric acid or D-(+)-di-p-toluoyltartaric acid to separate enantiomers. While effective in a laboratory setting, these resolution processes are inherently inefficient for large-scale manufacturing because they theoretically discard 50% of the synthesized material—the unwanted enantiomer. Furthermore, achieving high optical purity through resolution frequently necessitates multiple recrystallization steps, which drastically reduces overall yield and increases solvent consumption. Other approaches involving enzymatic kinetic resolution, while greener, often suffer from substrate specificity issues and lower catalytic efficiency, leading to inconsistent reaction rates and difficulties in scaling up to multi-ton production levels.
The Novel Approach
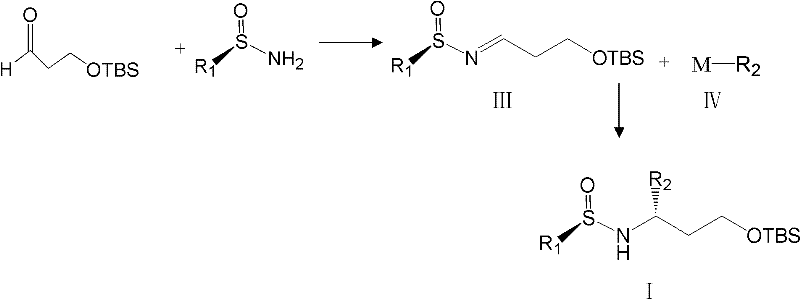
The methodology outlined in patent CN101875666B represents a paradigm shift from separation to direct asymmetric synthesis. Instead of creating a racemate and trying to fix it, this route constructs the chiral center directly with high fidelity. The core innovation lies in the use of chiral sulfinimines as electrophiles. By condensing a protected hydroxy-aldehyde with a chiral sulfinamide, a stable intermediate is formed that directs the subsequent addition of nucleophiles with exceptional stereocontrol. This approach bypasses the 50% yield ceiling inherent in resolution strategies. Moreover, the sulfinamide group serves a dual purpose: it acts as a chiral director during the bond-forming event and subsequently as a protecting group for the amine, simplifying the overall synthetic sequence. This consolidation of steps translates directly into reduced processing time and lower operational expenditures for manufacturing facilities.
Mechanistic Insights into Chiral Sulfinamide-Mediated Asymmetric Addition
The success of this synthetic strategy hinges on the unique properties of the tert-butylsulfinamide auxiliary. In the first stage of the reaction, 3-(tert-butyldimethylsilyloxy)propanal reacts with the chiral sulfinamide in the presence of a dehydrating agent, such as anhydrous copper(II) sulfate or titanium tetraisopropoxide. This condensation yields the N-sulfinyl imine (Compound III), which possesses a rigid geometry due to the coordination of the sulfur oxygen lone pair with the imine nitrogen. This rigidity is crucial for stereoinduction. When an organometallic reagent, such as a Grignard reagent (M-R2 where M is MgX) or an organolithium species, approaches the imine carbon, the bulky tert-butyl group on the sulfur effectively blocks one face of the molecule. Consequently, the nucleophile attacks exclusively from the less hindered face, establishing the new stereocenter with high diastereoselectivity. The reaction is typically conducted at low temperatures, ranging from -78°C to -25°C, to further suppress any non-selective background reactions and maximize the optical purity of the product.
Following the asymmetric addition, the resulting sulfinamide adduct (Compound I) contains both the desired chiral amine functionality and a protected alcohol. The removal of the protecting groups is straightforward and efficient. Treatment with acidic conditions, such as hydrochloric acid in methanol or trifluoroacetic acid in dichloromethane, cleaves both the N-sulfinyl group and the O-silyl ether simultaneously. This one-pot deprotection reveals the free 1,3-amino alcohol (Compound VII) without requiring intermediate isolation steps. The ability to remove both protecting groups under similar conditions is a significant process advantage, as it minimizes unit operations and solvent swaps. This streamlined workflow reduces the potential for impurity generation and ensures that the final intermediate retains the high optical purity established in the initial addition step, consistently delivering products with ee values greater than 99%.
How to Synthesize Optically Pure 1,3-Alkamine Efficiently
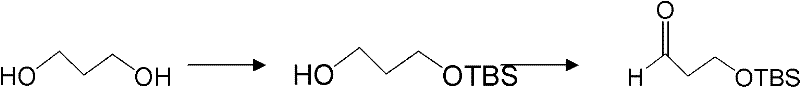
The synthesis of these valuable chiral intermediates follows a logical and scalable progression that begins with commodity chemicals. The process initiates with the protection of 1,3-propanediol to generate the mono-protected aldehyde precursor, followed by condensation with the chiral auxiliary and subsequent nucleophilic addition. This sequence is designed to be robust, tolerating a variety of aromatic and heteroaromatic substituents, which allows for the synthesis of a wide range of Dapoxetine analogues. The detailed standardized synthesis steps for implementing this route in a pilot or production plant are provided in the guide below.
- Condense 3-(tert-butyldimethylsilyloxy)propanal with a chiral sulfinamide (e.g., (R)-tert-butylsulfinamide) using a dehydrating agent like anhydrous copper sulfate to form the chiral sulfinimine intermediate.
- React the resulting chiral sulfinimine with an organometallic reagent (such as a Grignard or organolithium reagent) at low temperatures (-78°C to -25°C) to achieve stereoselective addition.
- Remove the protecting groups (sulfinyl and silyl) under acidic conditions to yield the optically pure 1,3-amino alcohol, which can be further derivatized into Dapoxetine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from resolution-based methods to this asymmetric synthesis offers tangible economic and logistical benefits. The primary advantage lies in the drastic improvement of atom economy. By eliminating the discard of the unwanted enantiomer, the effective yield of the process is nearly doubled compared to traditional resolution, significantly lowering the cost of goods sold (COGS). Furthermore, the starting materials required for this synthesis, such as 1,3-propanediol and various aryl Grignard reagents, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of raw material shortages and price volatility, ensuring a more stable and predictable procurement environment for long-term production contracts.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes the need for expensive resolving agents and the associated solvent-intensive recrystallization processes. Additionally, the high stereoselectivity reduces the burden on analytical quality control, as there is less need to monitor and separate closely related enantiomeric impurities. The consolidation of protection and deprotection steps further reduces labor and utility costs, contributing to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Because the synthetic route relies on robust chemical transformations rather than sensitive biological catalysts, it is less susceptible to variations in raw material quality or environmental conditions. The use of standard organometallic chemistry means that the process can be easily transferred between different manufacturing sites without the need for specialized enzymatic equipment. This flexibility enhances supply chain resilience, allowing for rapid scale-up or technology transfer to meet fluctuating market demands for Dapoxetine.
- Scalability and Environmental Compliance: The process generates significantly less chemical waste compared to resolution methods, aligning with modern green chemistry principles and reducing the costs associated with waste disposal and environmental compliance. The solvents used, such as dichloromethane, toluene, and tetrahydrofuran, are standard industrial solvents with well-established recovery and recycling protocols. This makes the process highly scalable from kilogram to multi-ton production levels while maintaining a favorable environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What is the optical purity achievable with this synthesis method?
A: According to the patent data, this asymmetric synthesis route utilizing chiral sulfinamide auxiliaries can achieve an enantiomeric excess (ee) value greater than 99%, significantly reducing the need for downstream purification compared to traditional resolution methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes readily available starting materials like 3-hydroxypropanal derivatives and standard organometallic reagents, avoiding complex enzymatic resolutions that often face bottlenecks during commercial scale-up.
Q: How does this method compare to traditional chiral resolution?
A: Traditional resolution methods typically waste up to 50% of the racemic material and require multiple recrystallizations. This asymmetric addition approach builds chirality directly, maximizing atom economy and reducing raw material costs and environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Alkamine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the successful commercialization of pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR instrumentation. Our expertise in chiral sulfinamide chemistry allows us to optimize this specific route for maximum efficiency and cost-effectiveness, providing our clients with a competitive edge in the global marketplace.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By leveraging our optimized synthesis of optically pure 1,3-alkamine compounds, you can secure a reliable supply of key Dapoxetine precursors while significantly reducing your manufacturing overhead. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and quotations for custom synthesis projects. Let us partner with you to drive innovation and efficiency in your pharmaceutical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
