Advanced Synthesis of 11-O-Mogrol Oxime Ethers: A Strategic Breakthrough for Functional Additive Manufacturing
Advanced Synthesis of 11-O-Mogrol Oxime Ethers: A Strategic Breakthrough for Functional Additive Manufacturing
The landscape of natural product-derived antioxidants is undergoing a significant transformation with the emergence of novel semi-synthetic derivatives designed to overcome the inherent limitations of their parent compounds. Patent CN111560044B introduces a groundbreaking class of 11-O-momordica grosvenori alcohol oxime ether derivatives, representing a sophisticated evolution in the modification of mogrol aglycones. This technology addresses critical challenges in bioavailability and stability that have long plagued the application of traditional mogrosides in high-value sectors such as functional foods and pharmaceuticals. By strategically modifying the C-11 position through oximation and subsequent etherification, researchers have unlocked a new tier of biochemical efficacy, characterized by markedly improved water solubility and potent free radical scavenging capabilities. For industry leaders seeking a reliable pharma intermediates supplier, this patent offers a validated pathway to producing next-generation active ingredients that bridge the gap between natural origin and synthetic performance.
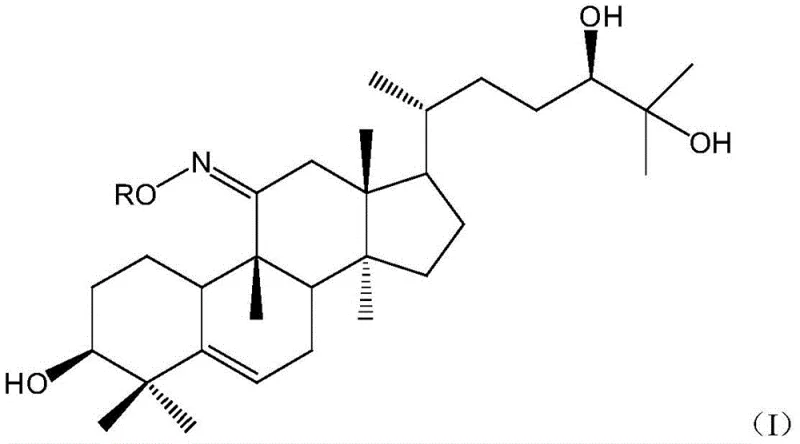
The core innovation lies in the structural versatility of the general formula (I), where the R group can be tailored to optimize physicochemical properties without compromising the triterpenoid scaffold's biological integrity. This adaptability allows manufacturers to fine-tune the lipophilicity and metabolic stability of the final product, catering to specific formulation requirements in oral delivery systems. The transition from simple hydroxylation to oxime ether functionality represents a paradigm shift in how we approach the valorization of Momordica grosvenori extracts, moving beyond mere extraction to precise molecular engineering.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the utilization of mogrol and its glycosides has been constrained by poor aqueous solubility and relatively rapid metabolic clearance, which limits their therapeutic window and commercial viability in liquid formulations. Conventional extraction methods yield mixtures that require extensive purification, often resulting in low overall yields and inconsistent potency profiles. Furthermore, the native hydroxyl groups on the mogrol skeleton, while biologically active, are prone to rapid conjugation and elimination in vivo, reducing the effective concentration at the target site. Traditional chemical modifications often involve harsh conditions that degrade the sensitive triterpenoid backbone or introduce toxic impurities that are difficult to remove, creating significant bottlenecks for cost reduction in pharmaceutical intermediates manufacturing. These legacy processes fail to deliver the consistent quality and enhanced performance metrics required by modern regulatory standards for food and drug additives.
The Novel Approach
In stark contrast, the methodology disclosed in CN111560044B employs a mild, two-step synthetic strategy that preserves the stereochemical integrity of the mogrol core while introducing robust functional groups. The process begins with a highly selective oximation at the C-11 carbonyl position, followed by a controlled etherification that installs diverse alkyl or aryl moieties. This approach not only enhances the lipophilic-hydrophilic balance but also sterically protects the reactive center from premature metabolic degradation. As illustrated in the synthetic pathway below, the reaction conditions are optimized for high conversion and ease of purification, utilizing standard solvent systems like DMF and petroleum ether.
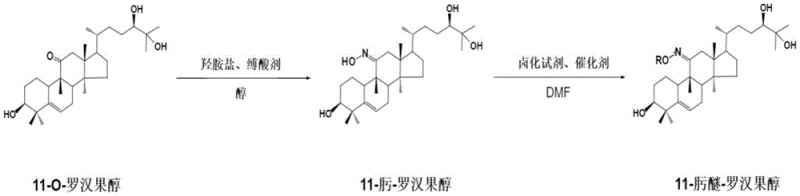
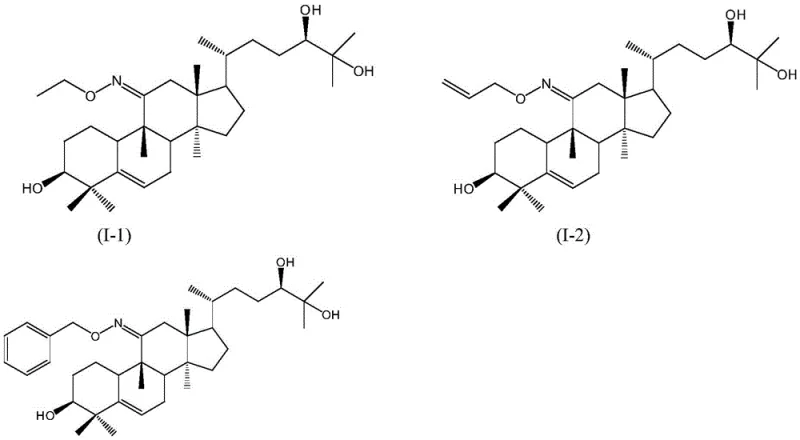
The novelty of this route is further evidenced by its ability to generate a library of derivatives with tunable properties, as seen in specific examples where R varies from simple ethyl groups to complex benzyl structures. This modularity ensures that manufacturers can adapt the synthesis to available raw materials while maintaining a consistent quality output. The result is a product with obviously enhanced antioxidant activity and a significantly prolonged in-vivo half-life, directly addressing the pharmacokinetic deficiencies of the parent compound.
Mechanistic Insights into Oximation and Etherification Catalysis
The chemical transformation relies on a precise sequence of nucleophilic additions and substitutions that leverage the unique reactivity of the 11-keto group on the mogrol skeleton. In the first stage, hydroxylamine salts act as potent nucleophiles, attacking the electrophilic carbonyl carbon to form an intermediate hemiaminal, which subsequently dehydrates to yield the stable oxime. The presence of an organic base, such as pyridine or triethylamine, is critical in this step to scavenge the generated acid and drive the equilibrium towards product formation. This step is conducted under reflux in alcoholic solvents, ensuring complete conversion while minimizing side reactions that could affect the adjacent double bonds in the triterpenoid ring system.
Following isolation, the 11-oxime-mogrol undergoes O-alkylation via an SN2 mechanism in the second stage. Here, the oxime oxygen acts as a nucleophile, displacing a halide leaving group from the alkylating agent in the presence of a strong base catalyst like sodium methoxide or sodium hydride. The reaction is performed at low temperatures, typically between -10°C and 10°C, to suppress competing elimination reactions and ensure high regioselectivity for the O-ether over potential N-alkylation byproducts. The structural diversity achievable through this mechanism is highlighted by the specific compounds synthesized, ranging from ethyl ethers to allyl and benzyl derivatives.
This mechanistic control is paramount for achieving the high purity levels (>98%) reported in the patent examples, as it minimizes the formation of difficult-to-separate isomers. The use of phase transfer catalysts or specific alkoxide bases further enhances the reaction kinetics, allowing for shorter reaction times and reduced energy consumption. Understanding these mechanistic nuances is essential for scaling the process, as slight deviations in temperature or base strength can impact the ratio of O-alkylation to N-alkylation, thereby affecting the final product's biological profile.
How to Synthesize 11-O-Mogrol Oxime Ether Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process utilizes readily available starting materials and avoids the use of exotic or prohibitively expensive reagents, making it an attractive option for commercial adoption. Detailed operational parameters, such as solvent ratios, cooling rates, and crystallization times, are meticulously defined to ensure consistent crystal morphology and purity. For a comprehensive guide on executing this synthesis with optimal yield and purity, please refer to the standardized procedure below.
- Dissolve 11-O-mogrol in absolute alcohol, add hydroxylamine salt and an organic base, and reflux to form 11-oxime-mogrol.
- Dissolve the intermediate in DMF, add a halogenating reagent and catalyst, and react at low temperature (-10 to 10°C).
- Quench in ice water, extract with petroleum ether, and purify via silica gel column chromatography to isolate the final oxime ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits, primarily driven by the accessibility of raw materials and the simplicity of the downstream processing. The starting material, 11-O-mogrol, can be sourced reliably through the hydrolysis of abundant natural mogrosides, ensuring a stable feedstock supply that is not subject to the volatility of fully synthetic petrochemical precursors. This biological origin aligns perfectly with the growing market demand for "natural-identical" or bio-based ingredients in the food and supplement sectors, providing a compelling marketing narrative alongside technical performance.
- Cost Reduction in Manufacturing: The process eliminates the need for precious metal catalysts or complex enzymatic systems, relying instead on inexpensive inorganic bases and common organic solvents. This simplification of the catalytic system drastically reduces the cost of goods sold (COGS) by removing expensive catalyst recovery steps and minimizing heavy metal contamination risks. Furthermore, the high selectivity of the reaction reduces the burden on purification, lowering the consumption of silica gel and eluents, which translates to significant operational savings in large-scale production environments.
- Enhanced Supply Chain Reliability: By utilizing a semi-synthetic approach rooted in agricultural feedstocks, manufacturers can decouple their supply chains from the geopolitical instabilities often associated with specialized chemical imports. The reagents required, such as hydroxylamine hydrochloride and alkyl halides, are commodity chemicals produced globally in massive volumes, ensuring uninterrupted availability. This resilience is critical for maintaining continuous production schedules and meeting the just-in-time delivery expectations of major pharmaceutical and nutraceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are inherently safe, operating at near-ambient pressures and moderate temperatures, which simplifies reactor design and reduces energy infrastructure requirements. The waste stream is primarily composed of benign salts and recoverable solvents, facilitating easier compliance with increasingly stringent environmental regulations regarding hazardous waste disposal. The ability to scale this process from grams to tons without fundamental changes to the chemistry ensures a smooth transition from R&D to commercial manufacturing, mitigating the technical risks typically associated with process scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel derivatives. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing product pipelines.
Q: What are the primary advantages of 11-O-mogrol oxime ethers over native mogrol?
A: The oxime ether derivatives exhibit significantly enhanced water solubility and antioxidant activity compared to the parent 11-O-mogrol, along with a prolonged in-vivo half-life, making them superior for oral bioavailability.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patent describes a robust two-step route using commercially available reagents like hydroxylamine salts and alkyl halides, with purification achievable through standard silica gel chromatography, ensuring scalability.
Q: What is the toxicity profile of these derivatives?
A: Toxicity tests on mice and rats indicated no death or toxic side effects even at high doses (40mg/kg/d), confirming their safety for use in food, beverages, and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11-O-Mogrol Oxime Ether Supplier
As the global demand for high-performance natural antioxidants continues to surge, the ability to deliver complex molecules with consistent quality becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this patented technology to market. Our state-of-the-art facilities are equipped to handle the specific solvent systems and low-temperature requirements of this synthesis, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that the transition from patent to product requires more than just chemical capability; it demands a partner who can navigate the complexities of regulatory compliance and supply chain logistics.
We invite procurement leaders and R&D directors to collaborate with us to unlock the full potential of 11-O-mogrol oxime ethers for your next generation of health products. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
