Revolutionizing Alpha-Hydroxylation: A Green Photocatalytic Route for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Photocatalytic Oxidation Technologies
The landscape of organic synthesis is undergoing a paradigm shift towards sustainability, driven by the urgent need to replace hazardous stoichiometric oxidants with greener alternatives. Patent CN113121338A introduces a groundbreaking synthetic method for preparing alpha-hydroxy-beta-keto acid ester compounds, which serve as critical scaffolds in the construction of biologically active molecules. This technology leverages visible light as a driving force and molecular oxygen from air as the terminal oxidant, marking a significant departure from traditional methods that rely on toxic or explosive reagents. The ability to synthesize these valuable intermediates under such mild and environmentally benign conditions represents a major leap forward for the fine chemical industry, particularly for manufacturers seeking reliable pharmaceutical intermediate supplier partnerships that prioritize green chemistry principles without compromising on yield or purity.
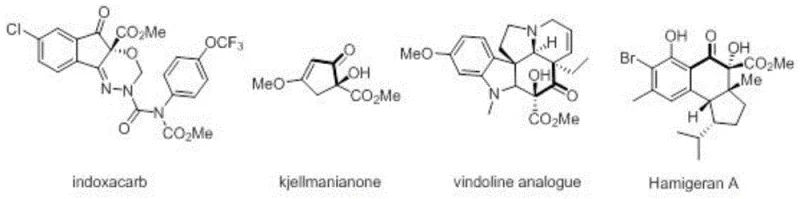
The significance of this innovation extends beyond mere academic interest; it addresses real-world production challenges faced by global supply chains. Alpha-hydroxy-beta-keto esters are pivotal precursors for a wide array of high-value compounds, including prominent agrochemicals and pharmaceuticals. As illustrated in the structural diversity of bioactive molecules below, the utility of this scaffold is vast, underpinning the efficacy of products like Indoxacarb and various natural product analogues. By mastering this photocatalytic approach, producers can secure a competitive edge in cost reduction in API manufacturing while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alpha-hydroxylation of beta-keto esters has been achieved using stoichiometric amounts of strong oxidizing agents such as organic peroxides, Davis reagents (sulfonyloxaziridines), or nitrosobenzenes. While these methods can deliver the desired transformation, they are fraught with significant drawbacks that hinder their application in large-scale commercial settings. The use of organic peroxides, for instance, introduces severe safety hazards due to their potential instability and explosiveness, necessitating specialized handling equipment and rigorous safety protocols that drive up operational costs. Furthermore, these traditional oxidants often suffer from poor atom economy, generating substantial quantities of chemical waste that require complex and expensive disposal procedures. The reliance on such reagents also limits the functional group tolerance of the reaction, often leading to side reactions that compromise the purity of the final product and complicate downstream purification processes.
The Novel Approach
In stark contrast, the method disclosed in patent CN113121338A utilizes a sophisticated catalytic system that harnesses the power of visible light and ambient air. This approach replaces dangerous stoichiometric oxidants with molecular oxygen, which is abundant, inexpensive, and produces water as the only byproduct, thereby aligning perfectly with the principles of green chemistry. The core of this innovation lies in the synergistic combination of a copper-amine complex catalyst and a photosensitizer. This dual-catalyst system enables the activation of molecular oxygen under mild irradiation, facilitating the selective insertion of an oxygen atom at the alpha-position of the beta-keto ester. The result is a process that operates at near-room temperature with exceptional efficiency, eliminating the thermal stress on sensitive substrates and drastically reducing the energy footprint of the synthesis. This novel pathway not only enhances safety but also streamlines the workflow, making it an ideal candidate for the commercial scale-up of complex polymer additives and fine chemicals.
Mechanistic Insights into Copper-Amine Photocatalytic Alpha-Hydroxylation
The mechanistic elegance of this transformation relies on the intricate interplay between the copper-amine complex and the photosensitizer under visible light irradiation. The copper salt, coordinated with specific amine ligands such as ethylenediamine or proline derivatives, forms a reactive species capable of activating the beta-keto ester substrate. Upon exposure to visible light, the photosensitizer (e.g., tetraphenylporphyrin or TPP) absorbs photons to reach an excited state, which then interacts with the copper complex or directly with molecular oxygen to generate reactive oxygen species, likely singlet oxygen or superoxide radicals. These activated oxygen species then engage with the enol or enolate form of the beta-keto ester, which is stabilized by the copper center, to effect the alpha-hydroxylation. The general reaction scheme below depicts this transformation, highlighting the conversion of the starting material (Formula I) to the valuable alpha-hydroxy product (Formula II).

A critical aspect of this mechanism is the remarkable efficiency of the catalytic system, which allows for the use of the photosensitizer in extremely low concentrations, ranging from 0.005 mol% to 0.1 mol%. This is made possible by the excellent induction effect of the copper-amine complex, which lowers the activation energy for the oxidation step and prevents the rapid degradation of the photosensitizer often seen in other photocatalytic systems. The choice of ligand plays a pivotal role in tuning the redox potential of the copper center, ensuring high selectivity for the alpha-position while minimizing over-oxidation or decomposition of the sensitive keto-ester framework. This precise control over the reaction pathway is essential for maintaining high-purity OLED material standards or pharmaceutical grade specifications, where even trace impurities can be detrimental. The robustness of this catalytic cycle ensures consistent performance across a broad range of substrates, including those with electron-withdrawing or electron-donating substituents on the aromatic ring.
How to Synthesize Alpha-Hydroxy-Beta-Keto Esters Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to the preparation of the catalyst system and the control of reaction parameters. The process begins with the in-situ formation of the active copper-amine complex by mixing the copper salt and the chosen ligand in a suitable organic solvent such as tetrahydrofuran (THF) or toluene. Once the catalyst is formed, the beta-keto ester substrate and the trace amount of photosensitizer are introduced. The reaction vessel is then exposed to visible light, typically using green LEDs (around 525 nm) which match the absorption profile of common porphyrin-based photosensitizers, while maintaining an atmosphere of air to supply the necessary oxygen. Detailed standardized synthesis steps see the guide below.
- Prepare the catalyst system by mixing a copper salt (e.g., copper acetate) with an amine ligand (such as ethylenediamine) in an organic solvent like THF or toluene.
- Add the beta-keto ester substrate and a trace amount of photosensitizer (e.g., TPP) to the reaction mixture under an air atmosphere.
- Irradiate the mixture with visible light (e.g., green LED) at mild temperatures (room temperature to 50°C) until conversion is complete, followed by standard extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers compelling strategic advantages that extend far beyond the laboratory bench. The shift from hazardous stoichiometric oxidants to air as the primary reagent fundamentally alters the cost structure and risk profile of the manufacturing process. By eliminating the need for expensive and regulated oxidizing agents, companies can achieve substantial cost savings in raw material procurement. Furthermore, the simplified workup procedure, which typically involves standard extraction and chromatography without the need for quenching dangerous residual oxidants, reduces the consumption of auxiliary chemicals and solvents. This streamlined process flow translates directly into reduced lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this method are driven by the drastic reduction in reagent costs and waste disposal fees. Traditional methods utilizing Davis reagents or peroxides incur high costs not only for the reagents themselves but also for the safe handling and disposal of the resulting byproducts. In contrast, the use of air as an oxidant is virtually cost-free, and the ultra-low loading of the photosensitizer minimizes the expense associated with this specialized additive. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, contributing to a lower overall carbon footprint and operational expenditure. The elimination of heavy metal scavengers, often required in other transition-metal catalyzed processes, further simplifies the purification train and reduces material costs.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available and stable reagents. Organic peroxides and specialized oxidants often face supply volatility due to transportation restrictions and regulatory hurdles. By relying on copper salts, simple amine ligands, and atmospheric oxygen, manufacturers can secure a more stable and predictable supply of raw materials. The robustness of the reaction across various substrates means that a single catalytic platform can be used to produce a diverse portfolio of intermediates, reducing the need for multiple specialized production lines. This flexibility ensures continuity of supply even when specific precursor availability fluctuates, safeguarding against production bottlenecks.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but advancements in reactor design have made this increasingly feasible. The mild nature of this reaction, operating at ambient temperature and pressure, simplifies the engineering requirements for scale-up compared to high-pressure oxidation processes. From an environmental compliance perspective, the generation of minimal waste and the avoidance of toxic oxidants make this process highly attractive for meeting stringent global environmental standards. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand value of the end products in markets that prioritize sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light driven alpha-hydroxylation technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the primary advantages of using air over traditional oxidants in this synthesis?
A: Using air as the oxidant eliminates the need for hazardous and expensive reagents like organic peroxides or Davis reagents, significantly improving atom economy and reducing safety risks associated with explosive oxidants.
Q: How does the copper-amine complex improve catalytic efficiency?
A: The copper-amine complex acts as a highly efficient co-catalyst that synergizes with the photosensitizer, allowing for ultra-low loading of the photosensitizer (down to 0.005 mol%) while maintaining high yields.
Q: Is this method scalable for industrial production of agrochemical intermediates?
A: Yes, the mild reaction conditions (room temperature, atmospheric pressure) and the use of inexpensive, abundant reagents make this protocol highly suitable for commercial scale-up in the production of complex agrochemical and pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Beta-Keto Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology in the synthesis of high-value chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated at the gram scale are faithfully translated to industrial volumes. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications required by the global pharmaceutical and agrochemical industries. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the highest quality standards, leveraging our expertise in catalytic process development to optimize yield and minimize impurities.
We invite potential partners to engage with our technical procurement team to explore how this innovative synthetic route can be tailored to your specific project needs. By collaborating with us, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener methodology for your specific product portfolio. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both scientific excellence and commercial success in your supply chain operations.
