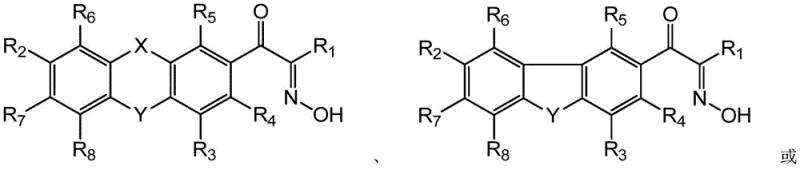
Scalable Production of High-Purity Alpha-Oxime Acetophenone Derivatives for Advanced Photoinitiator Applications
The chemical industry is constantly seeking more efficient and sustainable pathways for producing critical intermediates, and the technology disclosed in patent CN111233719B represents a significant leap forward in the synthesis of alpha-oxime acetophenone derivatives. These compounds serve as vital precursors in the manufacture of high-performance photoinitiators, which are essential for UV-curing applications in coatings, inks, and electronic materials. The traditional methods for preparing these isonitroso-containing intermediates have long been plagued by low conversion rates, typically hovering around 85 percent, and significant challenges in solvent management. By introducing a novel approach that utilizes hydrogen chloride gas saturation combined with batched solid sodium nitrite addition, this patent outlines a process that not only boosts yields to over 90 percent but also ensures the target product purity exceeds 98 percent. This technological advancement addresses the critical pain points of solvent recycling and reaction safety, offering a robust solution for manufacturers aiming to optimize their production lines for fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of alpha-hydroxyanilinone derivatives and related oxime structures has relied heavily on oximation reactions that suffer from inherent inefficiencies and environmental drawbacks. A primary issue with conventional protocols is the reliance on ethyl acetate as a solvent in the presence of aqueous hydrochloric acid, where water solubility in the organic phase creates a complex separation scenario. Because water has a certain solubility in ethyl acetate at normal temperatures, the subsequent dehydration treatment process becomes exceedingly complex and energy-intensive, often requiring secondary distillation steps to render the solvent reusable. Furthermore, ethyl acetate is prone to decomposition under prolonged acidic conditions, leading to a reduction in solvent content and increased waste generation. Another significant safety concern involves the isonitronizing reagents; while nitrite esters are effective, they are unstable when exposed to air and decompose upon heating to generate toxic nitrogen oxide gases, posing severe risks for industrial safety production. Alternatively, using solid nitrites dissolved in water leads to lower reaction conversion rates due to phase transfer limitations, creating a dilemma between safety, efficiency, and cost.
The Novel Approach
The innovative methodology presented in the patent data fundamentally alters the reaction environment to overcome these longstanding barriers by employing a hydrogen chloride gas introduction scheme. Instead of relying on aqueous acid solutions that introduce moisture, the process involves dissolving the acetophenone derivative in ethyl acetate with concentrated hydrochloric acid and then saturating the mixture with hydrogen chloride gas. This creates a highly acidic yet effectively anhydrous environment that drives the equilibrium towards the desired oxime product without compromising the integrity of the solvent. By adding solid sodium nitrite in controlled batches at temperatures between 0 and 50 degrees Celsius, the reaction avoids the thermal spikes associated with rapid exothermic events and eliminates the need for unstable nitrite esters. This approach allows the recovered ethyl acetate to be directly recycled without the need for complex dehydration and distillation procedures, drastically simplifying the downstream processing workflow. The result is a greener, safer reaction profile that achieves yields exceeding 90 percent while maintaining raw material costs at a minimum through efficient solvent reuse.
Mechanistic Insights into HCl-Mediated Oximation
The core of this technological breakthrough lies in the precise manipulation of the reaction medium's acidity and water content to favor the formation of the oxime functional group. In standard oximation, the presence of water can hydrolyze the intermediate or shift the equilibrium back towards the ketone, limiting conversion. By introducing hydrogen chloride gas, the system ensures a high concentration of protons necessary to activate the carbonyl group of the acetophenone derivative for nucleophilic attack by the nitrite species. The batched addition of solid sodium nitrite allows for a steady generation of nitrous acid in situ within the organic phase, facilitated by the HCl saturation, without overwhelming the system with water. This controlled generation prevents the localized accumulation of water that typically hampers solvent recovery in traditional aqueous methods. Furthermore, the specific temperature control between 10 and 30 degrees Celsius during the addition phase minimizes side reactions such as the decomposition of the nitrite or the hydrolysis of the ethyl acetate solvent. The mechanism effectively decouples the need for water-soluble reagents from the reaction efficiency, enabling a homogeneous or near-homogeneous reaction environment that maximizes contact between the organic substrate and the nitrosating agent.
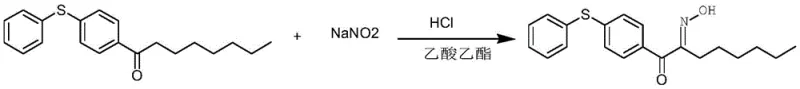
Impurity control in this process is achieved through the rigorous exclusion of water and the careful management of reaction kinetics via batch feeding. In comparative studies where recovered ethyl acetate was used without hydrogen chloride gas introduction, large amounts of impurities were generated, and the product conversion rate dropped below 50 percent, demonstrating the critical role of the acidic gas in suppressing side pathways. The absence of water prevents the hydrolysis of the newly formed oxime bond and reduces the formation of byproducts associated with solvent degradation. Additionally, the use of solid sodium nitrite rather than aqueous solutions prevents the dilution of the reaction mixture, which can lead to incomplete reactions and difficult separations. The washing and pH adjustment steps following the reaction are streamlined because the bulk of the solvent remains pure ethyl acetate, free from the azeotropic complications of water-ethanol or water-acetic acid mixtures that often arise from solvent decomposition. This mechanistic precision ensures that the final crystallization step yields a product with a content of more than 98 percent, suitable for sensitive applications in photoinitiator synthesis where trace impurities can detrimentally affect curing speeds and material properties.
How to Synthesize Alpha-Oxime Acetophenone Derivatives Efficiently
The synthesis protocol described in the patent offers a clear, scalable pathway for producing these valuable intermediates with high consistency and minimal waste. The process begins by charging a reaction kettle with the specific acetophenone derivative, fresh or recovered ethyl acetate, and concentrated hydrochloric acid, followed by stirring until complete dissolution is achieved. Once the substrate is fully solubilized, hydrogen chloride gas is introduced to saturate the solution, establishing the necessary acidic conditions for the subsequent oximation. Solid sodium nitrite is then added in small, controlled batches—typically every 3 to 10 minutes—to maintain the reaction temperature between 0 and 50 degrees Celsius, preventing thermal runaway. After the addition is complete, the mixture is held at temperature for 1 to 10 hours to ensure full conversion before undergoing a standard workup involving water washing, pH neutralization, and solvent distillation.
- Dissolve the acetophenone derivative in ethyl acetate with concentrated hydrochloric acid, then saturate the solution with hydrogen chloride gas to ensure anhydrous acidic conditions.
- Add solid sodium nitrite in controlled batches at 0-50°C, maintaining the temperature for 1-10 hours to complete the oximation reaction without generating toxic nitrogen oxides.
- Wash the reaction mixture to neutral pH, distill off the ethyl acetate for direct recycling, and crystallize the final product using cyclohexane to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible operational efficiencies and cost optimizations that extend beyond simple yield improvements. The ability to recycle ethyl acetate directly without energy-intensive dehydration steps represents a significant reduction in utility costs and solvent procurement volumes, directly impacting the bottom line of large-scale manufacturing operations. By eliminating the need for expensive and hazardous nitrite esters in favor of commodity-grade sodium nitrite, the raw material cost structure is substantially lowered while simultaneously improving workplace safety profiles. The robustness of the process against impurity formation means fewer batches are rejected due to quality failures, ensuring a more consistent and reliable flow of materials to downstream customers who depend on strict specifications for their own formulations. This reliability is crucial for maintaining just-in-time inventory levels and reducing the safety stock requirements that often burden supply chains dealing with inconsistent chemical suppliers.
- Cost Reduction in Manufacturing: The elimination of complex solvent dehydration processes significantly lowers energy consumption and operational time, leading to substantial cost savings in the overall production cycle. By utilizing inexpensive solid sodium nitrite instead of costly nitrite esters, the direct material costs are drastically reduced without compromising reaction efficiency or product quality. The high yield of over 90 percent ensures that raw material utilization is maximized, minimizing the waste disposal costs associated with unreacted starting materials and byproducts. Furthermore, the extended lifespan of the recycled ethyl acetate solvent reduces the frequency of fresh solvent purchases, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The simplified process flow, which avoids complex azeotropic distillations and hazardous reagent handling, reduces the risk of unplanned shutdowns or delays caused by equipment maintenance or safety incidents. The use of stable, solid reagents like sodium nitrite ensures that raw material availability is not subject to the volatility of specialized chemical markets, securing the supply chain against external disruptions. Consistent product quality with purity levels exceeding 98 percent reduces the need for extensive incoming quality control testing by customers, fostering stronger trust and longer-term contractual relationships. This stability allows for more accurate production planning and forecasting, enabling the supply chain team to respond more agilely to fluctuations in market demand.
- Scalability and Environmental Compliance: The batch addition method for sodium nitrite provides excellent control over exothermic reactions, making the process inherently safer and easier to scale from pilot plants to multi-ton commercial production facilities. The green nature of the reaction, characterized by the absence of toxic nitrogen oxide emissions and the recycling of solvents, aligns with increasingly stringent environmental regulations and corporate sustainability goals. Reduced solvent waste and lower energy requirements for distillation contribute to a smaller carbon footprint, enhancing the company's reputation as a responsible manufacturer in the global fine chemicals market. The process design facilitates easy integration into existing infrastructure, allowing for rapid capacity expansion to meet growing demand for high-purity photoinitiator intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced oximation technology, providing clarity on its operational benefits and compatibility with existing manufacturing setups. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring that the information is grounded in verified chemical principles and practical outcomes. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their specific product portfolios and for procurement specialists assessing the long-term value proposition.
Q: How does the HCl gas introduction method improve solvent recovery compared to traditional aqueous methods?
A: Traditional methods often introduce water via aqueous nitrite solutions, which complicates ethyl acetate recovery due to water solubility. The patented HCl gas saturation method maintains anhydrous conditions, allowing the ethyl acetate solvent to be distilled and reused directly without energy-intensive dehydration steps.
Q: What are the safety advantages of using solid sodium nitrite over nitrite esters in this process?
A: Nitrite esters are thermally unstable and can decompose to release toxic nitrogen oxide gases upon heating. Using solid sodium nitrite in batched additions eliminates the risk of sudden thermal decomposition and toxic gas release, significantly enhancing operational safety during large-scale manufacturing.
Q: What purity levels can be achieved with this optimized oximation protocol?
A: The process consistently achieves target product purities exceeding 98%, with specific examples demonstrating content levels greater than 99%. This high purity is critical for downstream applications in photoinitiator synthesis where impurities can affect curing performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Oxime Acetophenone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation photoinitiators and specialty chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch against the highest industry standards. By leveraging advanced synthetic methodologies like the HCl-mediated oximation process, we can offer our partners a reliable source of alpha-oxime acetophenone derivatives that drive performance in their final applications.
We invite you to collaborate with us to explore how these technical advancements can translate into competitive advantages for your business. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can support your supply chain goals and accelerate your time to market.
