Advanced Diboron Derivative Coupling Technology for Commercial Scale-Up of Complex Biaryl Compounds
Advanced Diboron Derivative Coupling Technology for Commercial Scale-Up of Complex Biaryl Compounds
The landscape of organic synthesis for fine chemicals and active pharmaceutical ingredients (APIs) is constantly evolving, driven by the need for more efficient, scalable, and environmentally benign processes. Patent CN1255123A introduces a groundbreaking methodology for covalently coupling organic compounds utilizing diboron derivatives, representing a significant leap forward in the construction of biaryl and aryl-alkenyl frameworks. This technology addresses long-standing challenges in cross-coupling reactions by enabling the formation of carbon-carbon bonds under remarkably mild conditions, often in protic solvents like ethanol or water. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this diboron-mediated coupling is essential, as it offers a robust pathway to high-purity aryl boron intermediates without the harsh constraints of traditional methods. The core innovation lies in the use of stable diboron derivatives which react with aryl halides in the presence of Group VIII metal catalysts to form aryl boron intermediates in situ, which can then be coupled further or isolated.
This patent data provides a comprehensive framework for synthesizing substituted di- and tri-aryl compounds that are of immense value to the pharmaceutical and agrochemical industries. Unlike conventional approaches that often require stringent anhydrous conditions and expensive reagents, this method demonstrates exceptional functional group tolerance. It allows for the direct coupling of substrates containing active hydrogen atoms, such as hydroxyl and amino groups, thereby streamlining synthetic routes by removing the need for protection and deprotection steps. For procurement managers focused on cost reduction in API manufacturing, this reduction in synthetic steps translates directly to lower material costs and reduced waste generation. Furthermore, the ability to perform these reactions in one pot, or with minimal isolation of intermediates, enhances the overall throughput and operational efficiency of the manufacturing process, making it an attractive option for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for forming covalent bonds between aromatic rings, such as the Grignard reaction or early iterations of palladium-catalyzed couplings, have historically been plagued by significant operational limitations that hinder their industrial applicability. Conventional Grignard reactions, for instance, require strictly anhydrous conditions and cryogenic temperatures to manage the high reactivity of the organomagnesium species, which poses safety risks and increases energy consumption. Moreover, these harsh conditions are incompatible with a wide range of functional groups; substituents containing active hydrogen atoms, such as alcohols, amines, or carboxylic acids, must be protected prior to the reaction to prevent unwanted side reactions or reagent decomposition. This necessity for protection and subsequent deprotection adds multiple steps to the synthetic route, drastically increasing the cost of goods sold (COGS) and extending the production timeline. Additionally, the solvents typically employed in these classical methods are often toxic, difficult to remove, and environmentally hazardous, creating substantial burdens for waste treatment and regulatory compliance.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN1255123A utilizes diboron derivatives to facilitate coupling under significantly milder and more forgiving conditions. This method employs stable diboron reagents, such as bis(pinacolato)diboron or bis(neopentyl glycolato)diboron, which react with aryl halides in the presence of a Group VIII metal catalyst and a suitable base. 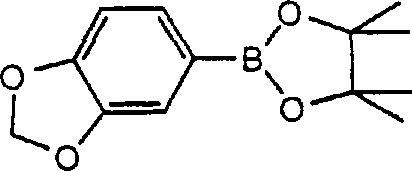 The reaction can be conducted in protic solvents like ethanol, methanol, or even water, which are inexpensive, safe, and easy to handle on a commercial scale. A key advantage of this methodology is its remarkable tolerance for functional groups; it allows for the direct coupling of aryl rings bearing active hydrogen substituents without the need for prior protection. This capability simplifies the synthetic strategy, reduces the number of unit operations, and minimizes the generation of chemical waste. Furthermore, the process allows for the control of reaction products by selecting specific diboron derivatives, enabling the selective formation of mono-borylated intermediates or symmetric biaryl products depending on the stoichiometry and reaction conditions employed.
The reaction can be conducted in protic solvents like ethanol, methanol, or even water, which are inexpensive, safe, and easy to handle on a commercial scale. A key advantage of this methodology is its remarkable tolerance for functional groups; it allows for the direct coupling of aryl rings bearing active hydrogen substituents without the need for prior protection. This capability simplifies the synthetic strategy, reduces the number of unit operations, and minimizes the generation of chemical waste. Furthermore, the process allows for the control of reaction products by selecting specific diboron derivatives, enabling the selective formation of mono-borylated intermediates or symmetric biaryl products depending on the stoichiometry and reaction conditions employed.
Mechanistic Insights into Palladium-Catalyzed Diboron Coupling
The mechanistic pathway of this coupling reaction involves a sophisticated interplay between the Group VIII metal catalyst, the diboron derivative, and the base, facilitating the transmetallation and reductive elimination steps essential for carbon-carbon bond formation. Initially, the palladium catalyst undergoes oxidative addition with the aryl halide substrate to form an aryl-palladium-halide complex. Simultaneously, the base activates the diboron derivative, generating a nucleophilic boron species that can participate in transmetallation. The aryl group from the palladium complex is then transferred to the boron atom, yielding the aryl boron intermediate and regenerating the palladium catalyst. This intermediate is stable enough to be isolated if desired, but can also react in situ with a second electrophile to form the final coupled product. The choice of ligand on the palladium catalyst plays a critical role in modulating the electronic and steric environment of the metal center, thereby influencing the reaction rate and selectivity. Ligands such as dppf or triphenylphosphine are commonly used to stabilize the active catalytic species and promote efficient turnover.
Impurity control is a paramount concern in the synthesis of high-purity pharmaceutical intermediates, and this method offers distinct advantages in managing byproduct formation. One potential issue in diboron coupling is the formation of symmetric biaryl byproducts due to the homocoupling of the aryl halide or the aryl boron intermediate. However, the patent data indicates that by carefully controlling the stoichiometry of the diboron derivative and the timing of reagent addition, the formation of these symmetric impurities can be minimized. For instance, adding water or a weak oxidizing agent after the formation of the aryl boron intermediate can decompose any unreacted diboron derivative, preventing it from interfering with the subsequent coupling step. 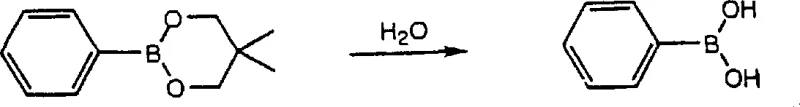 This strategic quenching step ensures that the aryl boron intermediate reacts selectively with the intended coupling partner, thereby enhancing the purity of the final product. Additionally, the use of mild reaction temperatures, typically ranging from 0°C to 120°C, helps to suppress thermal degradation pathways and side reactions that are common in high-temperature processes.
This strategic quenching step ensures that the aryl boron intermediate reacts selectively with the intended coupling partner, thereby enhancing the purity of the final product. Additionally, the use of mild reaction temperatures, typically ranging from 0°C to 120°C, helps to suppress thermal degradation pathways and side reactions that are common in high-temperature processes.
How to Synthesize Aryl Boron Intermediates Efficiently
The synthesis of aryl boron intermediates using this diboron methodology is a streamlined process that can be adapted for both laboratory-scale discovery and commercial-scale manufacturing. The general procedure involves charging a reaction vessel with the aryl halide substrate, the diboron derivative, a palladium catalyst, and a base in a suitable solvent system. The mixture is then heated to the desired temperature, typically between 15°C and 80°C, and stirred until the conversion is complete as monitored by GC or HPLC. Following the reaction, the workup procedure is straightforward, often involving extraction with organic solvents and purification by crystallization or chromatography. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below, providing a clear roadmap for technical teams to implement this technology.
- React an aromatic ring compound having a halogen substituent with a diboron derivative in the presence of a Group VIII metal catalyst and a suitable base.
- Decompose any excess diboron derivative using water, a base, or a weak oxidizing agent to prevent interference in subsequent steps.
- Couple the resulting aryl boron intermediate with a second organic compound bearing a halogen substituent to form the final covalently linked product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this diboron coupling technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary driver for cost optimization lies in the simplification of the synthetic route; by eliminating the need for protecting groups and reducing the number of isolation steps, manufacturers can achieve substantial cost savings in raw materials and labor. The ability to use inexpensive and non-toxic solvents like ethanol and water further reduces the operational expenditure associated with solvent purchase, recovery, and disposal. Moreover, the mild reaction conditions reduce the energy load on the manufacturing facility, contributing to a lower carbon footprint and aligning with modern sustainability goals. These factors combined result in a more resilient and cost-effective supply chain for critical intermediates.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps significantly reduces the number of synthetic operations required, leading to lower consumption of reagents and solvents. By avoiding the use of expensive and hazardous reagents typical of Grignard or lithiation chemistry, the overall material cost is drastically simplified. The process utilizes stable, commercially available diboron derivatives which, while specialized, offer a net cost benefit when factoring in the reduced processing time and waste treatment costs. This efficiency translates directly into a more competitive pricing structure for the final active pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: The robustness of this coupling method against moisture and oxygen, compared to traditional organometallic reactions, enhances the reliability of the supply chain. The reagents involved, such as pinacol boronic esters, are generally stable and have a long shelf life, reducing the risk of batch failures due to reagent degradation. Furthermore, the compatibility with a wide range of functional groups allows for greater flexibility in sourcing starting materials, as less refined grades of substrates may be tolerated. This flexibility mitigates the risk of supply disruptions and ensures a consistent flow of high-quality intermediates to downstream production lines.
- Scalability and Environmental Compliance: Scaling this process from kilogram to tonnage levels is facilitated by the use of standard reactor equipment and the absence of extreme temperature or pressure requirements. The preference for protic solvents and the generation of benign byproducts simplify the waste management process, ensuring compliance with increasingly stringent environmental regulations. The ability to perform one-pot reactions reduces the volume of waste generated per unit of product, supporting green chemistry initiatives. This scalability ensures that the technology can meet the demands of commercial production without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this diboron coupling technology, based on the specific embodiments and data provided in the patent documentation. These insights are designed to clarify the operational parameters and potential applications for R&D and process development teams evaluating this methodology for their own pipelines. Understanding these details is crucial for assessing the feasibility of adapting this chemistry to specific target molecules.
Q: Does this coupling method tolerate active hydrogen substituents?
A: Yes, the method described in patent CN1255123A is particularly suitable for aromatic rings containing active hydrogen substituents such as hydroxy and amino groups, eliminating the need for protection steps.
Q: What catalysts are preferred for this diboron coupling reaction?
A: Group VIII metal catalysts are used, with palladium catalysts like PdCl2(dppf)·CH2Cl2 and Pd(PPh3)4 being highly preferred for their efficiency in activating carbon-halogen bonds.
Q: Can the reaction be performed in protic solvents?
A: Yes, the process works effectively in protic solvents such as water, ethanol, and methanol, which simplifies the workup and reduces the reliance on toxic organic solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Boron Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced coupling technologies like the diboron derivative method described in patent CN1255123A for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your innovative synthetic routes can be successfully translated into robust manufacturing processes. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous containment strategies for metal catalysts and advanced purification capabilities to meet stringent purity specifications. We are committed to delivering high-purity aryl boron intermediates that adhere to the highest quality standards, supported by our rigorous QC labs and comprehensive analytical testing protocols.
We invite you to collaborate with our technical procurement team to explore how this technology can optimize your supply chain and reduce your overall manufacturing costs. By leveraging our expertise in process development and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact us to request specific COA data for our catalog of boron intermediates or to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the delivery of life-saving medicines to the market through efficient and sustainable chemical manufacturing.
