Advanced Synthesis of 6-Alpha-Fluoro Steroids: A Safer, Scalable Route for Pharmaceutical Manufacturing
Introduction to Patent CN102040649A
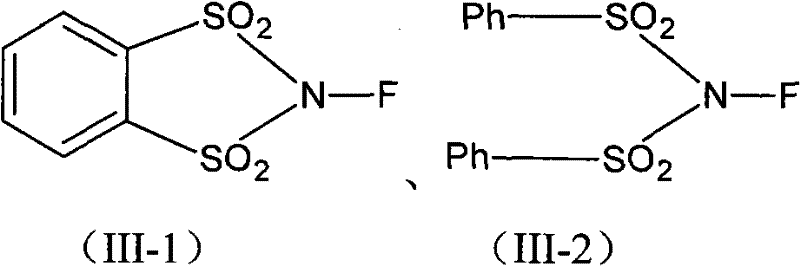
The pharmaceutical industry constantly seeks robust methodologies for introducing fluorine atoms into complex molecular scaffolds, particularly within the corticosteroid class where 6-alpha-fluorination dramatically enhances anti-inflammatory potency. Patent CN102040649A presents a groundbreaking synthesis process for preparing 6-alpha-fluoro steroids utilizing novel fluorinating agents, specifically N-fluoro-o-benzenedisulfonimide (NFOBS) and N-fluorobenzenesulfonimide (NFSI). This technology addresses critical limitations associated with traditional fluorination methods, offering a pathway that balances high chemical yield with exceptional operational safety. For R&D directors and process chemists, this patent represents a pivotal shift away from hazardous gaseous reagents toward manageable solid-state alternatives that simplify purification protocols. The ability to effectively introduce the fluorine atom at the 6-alpha position without compromising sensitive functional groups elsewhere on the steroid nucleus is a significant technical achievement. Furthermore, the versatility of the substrate scope, accommodating various substituents at positions 1, 2, 9, 11, and 16, underscores the broad applicability of this method across a wide range of valuable API intermediates. As a reliable steroid intermediate supplier, understanding these mechanistic nuances is essential for optimizing production lines and ensuring consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of fluorine at the 6-position of steroids has been fraught with significant technical and safety challenges that hinder efficient manufacturing. Traditional methods often relied heavily on perchloryl fluoride, a reagent notorious for its explosive nature and extreme corrosivity, which necessitates specialized, expensive reactor materials and rigorous safety protocols that drive up capital expenditure. Moreover, perchloryl fluoride acts as a strong oxidizer, frequently leading to unwanted side reactions such as the oxidation of the C6 position to hydroxyl or keto compounds, thereby complicating the impurity profile and reducing overall yield. Alternative modern reagents like Selectfluor, while safer than perchloryl fluoride, suffer from prohibitively high manufacturing costs due to their complex synthetic preparation, making them economically unviable for large-scale industrial applications. These conventional approaches often require harsh reaction conditions, including extreme temperatures or strongly acidic environments, which can degrade sensitive ester or epoxy groups present in advanced steroid intermediates. Consequently, the industry has long suffered from a lack of cost-effective, safe, and scalable solutions for producing high-purity 6-alpha-fluoro steroids, creating a bottleneck in the supply chain for critical anti-inflammatory medications.
The Novel Approach
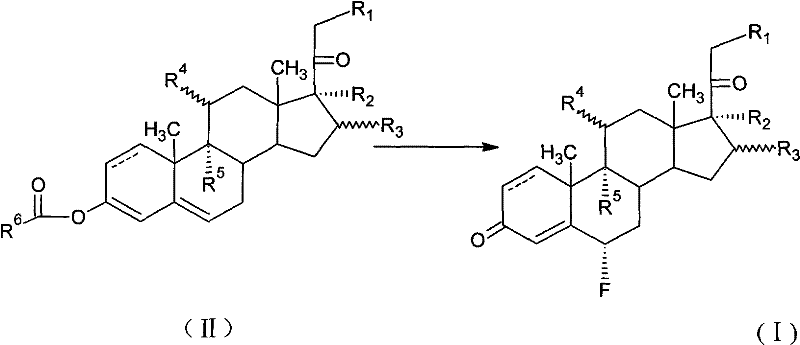
The methodology disclosed in CN102040649A revolutionizes this landscape by employing NFOBS and NFSI as superior electrophilic fluorinating agents that operate under remarkably mild and controllable conditions. Unlike the hazardous gases of the past, these reagents are stable solids that can be handled with standard industrial equipment, drastically reducing safety risks and infrastructure requirements. The process involves reacting a compound of Formula (II) with the fluorinating agent in common organic solvents such as acetonitrile, DMF, or chloroform-methanol mixtures, allowing for precise temperature control between -20°C and 60°C. This flexibility enables manufacturers to optimize reaction kinetics without fearing thermal runaway or decomposition of the valuable steroid substrate. Crucially, this novel approach eliminates the need for complex transposition steps often required in older routes, directly yielding the desired 6-alpha configuration with high selectivity. By removing the reliance on expensive reagents like Selectfluor and dangerous gases like perchloryl fluoride, this technology offers a clear path toward substantial cost reduction in pharmaceutical manufacturing while maintaining rigorous purity standards.
Mechanistic Insights into Electrophilic Fluorination with N-Sulfonyl Imides
The core of this technological advancement lies in the unique electrophilic character of the nitrogen-fluorine bond within the N-sulfonyl imide structure of NFOBS and NFSI. These reagents function by delivering a positively charged fluorine species (F+) to the electron-rich enol or enolate form of the steroid substrate, specifically targeting the C6 position adjacent to the carbonyl group. The sulfonimide moiety acts as an excellent leaving group, stabilizing the transition state and facilitating the formation of the carbon-fluorine bond with high regioselectivity. The presence of electron-withdrawing sulfonyl groups enhances the electrophilicity of the fluorine atom, making the reagent reactive enough to fluorinate the steroid nucleus yet stable enough to prevent non-specific oxidation or degradation of other functional groups. This mechanistic precision is vital for preserving the integrity of sensitive moieties such as 9-alpha-fluoro, 11-beta-hydroxy, or 16-methyl groups that are often present in potent corticosteroids. Furthermore, the reaction mechanism minimizes the formation of 6-beta isomers, which are difficult to separate and represent a significant loss of material value. Understanding this electronic interplay allows process chemists to fine-tune solvent polarity and temperature to maximize the ratio of the desired 6-alpha product, ensuring a cleaner crude profile that simplifies downstream purification.
Impurity control in this process is inherently superior due to the mild nature of the reaction conditions and the specific reactivity of the N-sulfonyl imides. Traditional oxidizers often generate chlorinated byproducts or over-oxidized ketones that require extensive chromatographic purification, driving up costs and waste. In contrast, the byproducts of the NFOBS/NFSI reaction are typically sulfonimide salts which are water-soluble and easily removed during the aqueous workup phase described in the patent examples. The use of buffered systems or the addition of mild bases like triethylamine or ammonia during the quenching step further ensures that any acidic byproducts are neutralized, preventing acid-catalyzed degradation of the steroid backbone. This clean reaction profile translates directly to higher isolated yields and reduced solvent consumption during recrystallization, key metrics for green chemistry and sustainable manufacturing. For supply chain managers, this means fewer batch failures and more predictable production schedules, as the process is less susceptible to the variability often seen with harsher fluorination chemistries.
How to Synthesize 6-Alpha-Fluoro Steroids Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometry to achieve optimal results as demonstrated in the patent examples. The process generally begins with dissolving the steroid precursor in a polar aprotic solvent like acetonitrile or DMF, or a mixed solvent system such as chloroform and methanol, under an inert nitrogen or argon atmosphere to prevent moisture interference. The reaction temperature is a critical parameter, with the patent indicating a broad operable range from -20°C to 60°C, though lower temperatures around 0°C to 10°C often provide the best balance between reaction rate and selectivity. The fluorinating agent is added in a slight molar excess, typically ranging from 1.1 to 1.4 equivalents relative to the substrate, to ensure complete conversion without excessive waste. Following the reaction period, which can last from 1.5 to 5 hours depending on the specific substrate and temperature, the mixture is quenched into ice water and neutralized with a base to precipitate or extract the product. Detailed standardized synthesis steps see the guide below.
- Dissolve the steroid precursor (Formula II) in a suitable organic solvent such as acetonitrile, DMF, or a chloroform/methanol mixture under an inert atmosphere.
- Cool the reaction mixture to a temperature between -20°C and 40°C, preferably around 0°C to 10°C, to control exothermicity and selectivity.
- Add the fluorinating agent (NFOBS or NFSI) slowly, maintain stirring for 1.5 to 5 hours, then quench with aqueous base and extract the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NFOBS/NFSI-based fluorination process offers transformative benefits that extend far beyond simple chemical yield improvements. The shift away from hazardous gaseous reagents eliminates the need for specialized storage facilities and complex gas handling systems, resulting in significant capital expenditure savings and reduced regulatory compliance burdens. The stability of the solid fluorinating agents simplifies logistics and inventory management, allowing for just-in-time purchasing strategies that reduce working capital tied up in dangerous raw materials. Furthermore, the compatibility of this method with common, low-cost industrial solvents like acetonitrile and methanol ensures that raw material sourcing remains resilient against market volatility. The simplified workup procedure, which avoids complex chromatographic separations, drastically reduces solvent usage and waste disposal costs, contributing to a leaner and more environmentally compliant manufacturing operation. These factors collectively enhance the overall reliability of the supply chain for critical steroid intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive reagents like Selectfluor with cost-effective NFOBS and NFSI directly lowers the bill of materials for every kilogram of product produced. By eliminating the need for expensive corrosion-resistant reactors required for perchloryl fluoride, manufacturers can utilize standard stainless steel equipment, further reducing depreciation and maintenance costs. The high selectivity of the reaction minimizes the loss of valuable starting materials to side products, effectively increasing the throughput of existing production assets without additional investment. Additionally, the simplified purification process reduces the consumption of high-grade solvents and silica gel, leading to substantial operational expense savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The use of stable, solid reagents mitigates the risk of supply disruptions associated with the transport and storage of hazardous gases or temperature-sensitive liquids. Since NFOBS and NFSI have longer shelf lives and are easier to ship globally, manufacturers can secure larger inventories with less risk, ensuring continuous production even during logistical bottlenecks. The robustness of the reaction conditions means that batch-to-batch variability is minimized, reducing the likelihood of failed batches that could delay shipments to downstream API manufacturers. This reliability is crucial for maintaining long-term contracts with major pharmaceutical clients who demand consistent quality and on-time delivery.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently scalable from pilot plant to multi-ton commercial production without significant engineering hurdles. The reduction in hazardous waste generation, particularly the avoidance of perchlorate salts and chlorinated byproducts, simplifies wastewater treatment and aligns with increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability credentials of the manufacturer, a growing requirement for inclusion in global pharmaceutical supply chains. The ability to scale safely and cleanly ensures that production capacity can be rapidly expanded to meet surging market demand for anti-inflammatory therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology, derived directly from the patent specifications and comparative data. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The answers reflect the specific advantages of using N-sulfonyl imide reagents over traditional methods, focusing on safety, selectivity, and operational efficiency. Stakeholders are encouraged to review these points when assessing the potential for technology transfer or licensing agreements.
Q: Why are NFOBS and NFSI preferred over perchloryl fluoride for steroid fluorination?
A: Perchloryl fluoride is highly explosive and corrosive, posing severe safety risks and requiring specialized equipment. NFOBS and NFSI are stable, solid reagents that operate under mild conditions, significantly enhancing operational safety and reducing infrastructure costs.
Q: What is the regioselectivity of this fluorination process?
A: The process specifically targets the 6-alpha position of the steroid nucleus. The electrophilic nature of the fluorine source combined with the specific steric environment of the steroid substrate ensures high selectivity for the 6-alpha isomer, minimizing difficult-to-separate impurities.
Q: Can this method be scaled for industrial production?
A: Yes, the use of common organic solvents like acetonitrile and DMF, along with the stability of the fluorinating agents, makes this process highly amenable to scale-up. The mild temperature requirements (-20°C to 60°C) further facilitate large-scale manufacturing without extreme cooling or heating demands.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Alpha-Fluoro Steroid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies like the NFOBS/NFSI fluorination process to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-scale operations. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 6-alpha-fluoro steroid intermediate meets the highest international standards for potency and impurity profiles. Our commitment to continuous improvement allows us to offer clients a supply partner that is not only technically proficient but also deeply aligned with the evolving needs of the modern drug development landscape.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to deliver high-quality steroid intermediates reliably and cost-effectively.
