Advanced Silver-Mediated Synthesis of Tolvaptan Intermediates for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of efficient pathways for vasopressin receptor antagonists has brought significant attention to the synthesis of Tolvaptan intermediates, specifically 2-methyl-4-N-(2-methylbenzoyl)benzoic acid. Patent CN102093247B, published in March 2014, introduces a transformative preparation method that fundamentally shifts the synthetic paradigm from hazardous high-pressure carbonylation to a mild, silver-mediated N-acylation strategy. This technological breakthrough addresses critical bottlenecks in the manufacturing of diuretic agents used for treating hyponatremia, offering a route that is not only chemically robust but also inherently safer for large-scale operations. By utilizing soluble silver salts in aprotic polar solvents, the inventors have successfully eliminated chloride ions from the reaction system, driving the acylation equilibrium forward with exceptional efficiency. The resulting process delivers the target compound with remarkable purity levels exceeding 99%, effectively bypassing the complex purification steps often associated with traditional transition metal catalysis. For global supply chain stakeholders, this patent represents a viable pathway to secure reliable supplies of high-value pharmaceutical building blocks while mitigating the operational risks inherent in older methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 2-methyl-4-N-(2-toluyl)benzoic acid was heavily reliant on palladium-catalyzed carbonylation techniques, as documented in literature such as Bioorganic & Medicinal Chemistry Letters (2007). These conventional routes necessitate the use of carbon monoxide gas under elevated pressures and temperatures ranging from 90 to 120°C, creating substantial safety hazards due to the flammability and toxicity of CO. Furthermore, the reliance on expensive palladium catalysts like Pd(OAc)2 alongside phosphine ligands and cesium salts drives up the raw material costs significantly, impacting the overall economic feasibility of the process. The requirement for strict anhydrous conditions and specialized high-pressure equipment further complicates the manufacturing landscape, limiting the number of capable suppliers and increasing lead times. Additionally, the presence of inflammable and explosive substances in the reaction mixture poses severe potential safety hazards that require rigorous engineering controls and monitoring systems. As illustrated in the legacy synthetic pathway below, the complexity of managing gas-liquid-solid multiphase reactions often leads to inconsistent yields and challenging scale-up scenarios.
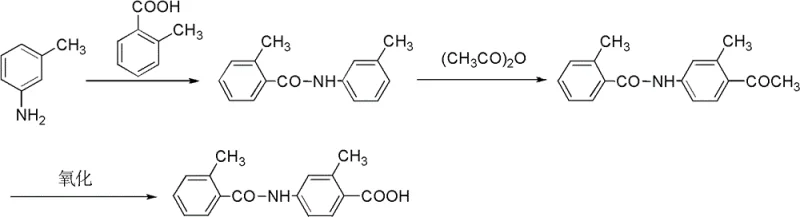
The Novel Approach
In stark contrast to the harsh conditions of carbonylation, the novel approach detailed in CN102093247B utilizes a silver-promoted acylation between methyl 2-methyl-4-aminobenzoate and 2-methylbenzoyl chloride. This method operates under mild thermal conditions, typically between -20°C and 40°C, drastically reducing energy consumption and eliminating the need for pressurized reactors. The core innovation lies in the use of silver salts, such as silver nitrate or silver trifluoromethanesulfonate, which act as chloride scavengers to drive the formation of the amide bond without generating corrosive hydrogen chloride byproducts. The reaction proceeds rapidly in common haloalkane solvents like dichloromethane or chloroform, allowing for straightforward workup procedures involving simple filtration and washing. Following the acylation, a subsequent hydrolysis step under basic conditions converts the methyl ester to the final carboxylic acid with yields reaching up to 96%. This two-step sequence, depicted in the reaction scheme below, simplifies the entire production workflow, removing the need for column chromatography and enabling the direct use of crude intermediates in downstream processing.
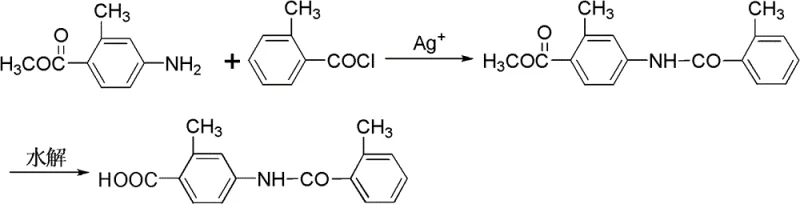
Mechanistic Insights into Silver-Salt Catalyzed N-Acylation
The mechanistic elegance of this process centers on the role of the silver cation (Ag+) as a potent Lewis acid and halide abstractor. When 2-methylbenzoyl chloride reacts with the amine substrate, the silver ion coordinates with the chloride leaving group, precipitating it as insoluble silver chloride or stabilizing it in solution, thereby preventing the reverse reaction and suppressing the formation of hydrochloride salts of the amine. This irreversible removal of chloride ions shifts the chemical equilibrium decisively towards the formation of the N-(2-methylbenzoyl) amide bond, ensuring high conversion rates even at ambient or sub-ambient temperatures. The choice of solvent is critical; aprotic polar solvents like acetonitrile or tetrahydrofuran are used to dissolve the silver salt, while haloalkanes serve as the bulk reaction medium to maintain solubility of the organic substrates. The molar ratio of the silver catalyst is carefully optimized, typically ranging from 0.9 to 3 equivalents relative to the amine, to ensure complete scavenging of chloride without excessive reagent waste. This precise stoichiometric control minimizes the formation of side products such as N,N-diacylated impurities, which are common pitfalls in uncatalyzed acylation reactions.
Impurity control is further enhanced by the mild nature of the hydrolysis step, which follows the acylation without intermediate purification. By dissolving the crude methyl ester in methanol and treating it with aqueous bases like sodium hydroxide or potassium carbonate at temperatures between 0°C and 65°C, the ester moiety is selectively cleaved while the sensitive amide bond remains intact. The patent data indicates that controlling the pH during the final acidification step to a range of 4 to 7 is crucial for maximizing the precipitation of the target benzoic acid while keeping soluble impurities in the mother liquor. This selectivity ensures that the final product achieves purity specifications greater than 99% as measured by HPLC normalization methods. The absence of heavy metal residues like palladium, which often require complex scavenging resins to meet regulatory limits for API intermediates, simplifies the quality control profile and reduces the risk of metal contamination in the final drug substance.
How to Synthesize 2-Methyl-4-N-(2-Methylbenzoyl)Benzoic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this critical Tolvaptan intermediate with high reproducibility and minimal operational complexity. The process begins with the dissolution of the amine and acid chloride starting materials in dry dichloromethane, followed by the controlled addition of the silver salt solution to manage the exotherm and ensure uniform mixing. Detailed standardized operating procedures for temperature control, stirring rates, and filtration techniques are essential to replicate the high yields reported in the embodiments, which range from 83% to 89% for the acylation step alone. The subsequent hydrolysis is equally straightforward, requiring only common laboratory reagents and standard glassware, making it highly adaptable for both pilot plant and commercial manufacturing scales. For a comprehensive breakdown of the specific reagent quantities, reaction times, and workup parameters, please refer to the step-by-step guide below.
- Dissolve methyl 2-methyl-4-aminobenzoate and 2-methylbenzoyl chloride in a dry haloalkane solvent, then slowly add a solution of silver salt (e.g., AgNO3) in an aprotic polar solvent at -20 to 40°C.
- After stirring for 10 minutes to 5 hours, quench with alkali-metal carbonate solution, filter off silver salts, and isolate the intermediate ester without purification.
- Hydrolyze the crude ester in methanol using a basic catalyst (NaOH or KOH) at 0 to 65°C, then acidify to pH 4-7 to precipitate the final benzoic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this silver-mediated synthesis route offers compelling strategic advantages over traditional palladium-based methods. The elimination of carbon monoxide gas removes a major safety liability and the associated need for specialized high-pressure infrastructure, thereby lowering the barrier to entry for contract manufacturing organizations and expanding the pool of potential suppliers. This shift not only enhances supply chain resilience by diversifying the manufacturing base but also significantly reduces the capital expenditure required for facility upgrades and safety compliance monitoring. Furthermore, the replacement of expensive noble metal catalysts like palladium with more abundant silver salts, combined with the ability to operate at near-ambient temperatures, leads to substantial cost savings in raw material procurement and energy consumption. The simplified workup procedure, which avoids time-consuming column chromatography, accelerates the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost palladium catalysts and toxic gases with readily available silver salts and liquid reagents. By operating at lower temperatures and atmospheric pressure, the process drastically reduces energy utility costs associated with heating and pressurization systems. The high yield and purity achieved without extensive purification steps minimize material loss and solvent usage, contributing to a leaner and more cost-effective manufacturing profile. Additionally, the reduced need for specialized equipment for handling hazardous gases lowers maintenance and depreciation costs over the lifecycle of the production facility.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the raw materials, including methyl 2-methyl-4-aminobenzoate and 2-methylbenzoyl chloride, are commodity chemicals with established global supply chains. The removal of dependency on specialized catalysts that may face supply constraints ensures consistent production scheduling and reduces the risk of delays caused by raw material shortages. The robustness of the reaction conditions means that production is less susceptible to minor variations in environmental factors, leading to more predictable batch outcomes and reliable delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard batch reactor operations rather than complex continuous flow gas-liquid systems. The absence of volatile organic compounds generated from gas feeds and the reduction in heavy metal waste simplify wastewater treatment and废气 (exhaust gas) management protocols. This alignment with green chemistry principles facilitates easier regulatory approval and environmental permitting, ensuring long-term operational continuity without the threat of shutdowns due to compliance violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Tolvaptan intermediates. These insights are derived directly from the experimental data and comparative analysis provided in patent CN102093247B, offering clarity on reaction specifics and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing supplier capabilities.
Q: Why is the silver-mediated method preferred over palladium-catalyzed carbonylation for this intermediate?
A: The silver-mediated method eliminates the need for high-pressure carbon monoxide gas and expensive palladium catalysts, significantly reducing equipment requirements and safety hazards while maintaining high yields.
Q: What are the critical reaction conditions for the N-acylation step?
A: The reaction requires dry haloalkane solvents like dichloromethane and temperatures between -20°C and 40°C. The molar ratio of amine to acid chloride is typically controlled between 1:0.9 and 1:1.5 to ensure complete conversion.
Q: Does the intermediate require purification before hydrolysis?
A: No, the patent specifies that the intermediate methyl ester can be used directly in the next hydrolysis step without column chromatography, as the silver salt byproducts are removed by filtration during the workup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-4-N-(2-Methylbenzoyl)Benzoic Acid Supplier
As the global demand for Tolvaptan continues to rise, securing a stable supply of high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands ready to support your projects with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of silver-mediated chemistries, ensuring stringent purity specifications and rigorous QC labs verify every batch against the highest industry standards. We understand the critical nature of timeline adherence in drug development and are committed to delivering consistent quality that meets the exacting needs of regulatory filings.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this high-performance intermediate into your manufacturing pipeline.
