Scalable Production of Fenprodone: A Technical Breakthrough in Green Agrochemical Manufacturing
Scalable Production of Fenprodone: A Technical Breakthrough in Green Agrochemical Manufacturing
The global demand for high-efficacy fungicides continues to drive innovation in agrochemical intermediate synthesis, particularly for complex benzophenone derivatives like Fenprodone. Patent CN111943823A introduces a transformative preparation method that addresses critical bottlenecks in the traditional manufacturing of 3′-bromo-2,3,4,6′-tetramethoxy-2′,6-dimethylbenzophenone. This technical disclosure outlines a convergent synthetic strategy that replaces hazardous reagents with greener catalytic systems, ensuring both economic viability and environmental compliance. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the nuances of this eleven-step sequence is paramount. The patent details a robust pathway starting from readily available raw materials such as 2,3-dimethylaniline and p-cresol, culminating in a high-purity final product through optimized Friedel-Crafts acylation. By integrating advanced oxidation techniques and efficient solvent recovery loops, this methodology sets a new benchmark for cost reduction in fungicide manufacturing while maintaining stringent quality standards required for global registration.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fenprodone has been plagued by inefficient protocols that rely heavily on stoichiometric amounts of corrosive and environmentally damaging reagents. Prior art, such as the methods disclosed in EP 0897904 A1 and US 5922905 A, typically employs phosphorus pentoxide or large excesses of aluminum trichloride as catalysts for the key acylation step. These traditional approaches suffer from severe drawbacks, including prolonged reaction times extending up to sixty hours and notoriously low overall yields, often hovering around thirty percent. Furthermore, the generation of inorganic phosphorus waste poses a significant threat of water eutrophication, creating substantial disposal costs and regulatory hurdles for manufacturers. The reliance on harsh acidic conditions also complicates downstream processing, requiring extensive neutralization and purification steps that erode profit margins. For supply chain heads, these inefficiencies translate into unpredictable lead times and volatile pricing structures, making the secure sourcing of high-purity Fenprodone a persistent challenge in the competitive agrochemical market.
The Novel Approach
In stark contrast, the methodology presented in CN111943823A offers a streamlined, convergent synthesis that dramatically improves process economics and operational safety. This novel approach bifurcates the synthesis into two parallel streams: the preparation of 5-bromo-2-methoxy-6-methylbenzoyl chloride and the synthesis of 3,4,5-trimethoxytoluene, which are subsequently coupled. A standout feature of this route is the replacement of toxic phosphorus-based catalysts with anhydrous ferric chloride in the final coupling step, coupled with a highly selective copper-catalyzed oxidation system earlier in the sequence. The process incorporates sophisticated solvent recovery mechanisms, allowing for the recycling of chlorobenzene and methanol, which significantly lowers raw material consumption. By optimizing reaction temperatures and utilizing phase-transfer catalysts during hydrolysis, the method achieves superior conversion rates and simplifies isolation procedures. This strategic redesign not only enhances the commercial scale-up of complex benzophenones but also aligns with modern green chemistry principles, offering a sustainable alternative for industrial production.
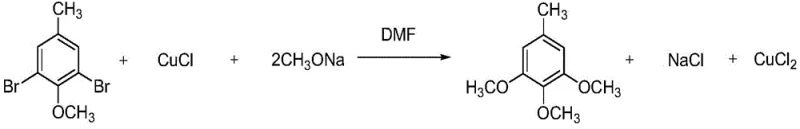
Mechanistic Insights into Copper-Catalyzed Selective Oxidation and Acylation
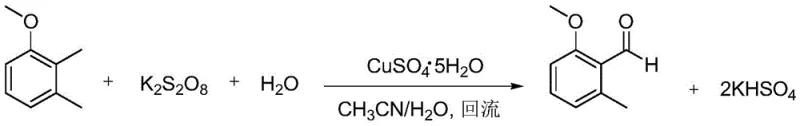
A critical innovation within this patent is the implementation of a copper-catalyzed selective oxidation system to convert 2,3-dimethylanisole into 2-methoxy-6-methylbenzaldehyde. Unlike traditional oxidants that often lack selectivity and produce tarry byproducts, this system utilizes a mixture of copper sulfate pentahydrate and potassium persulfate in an aqueous acetonitrile medium. The mechanism involves the generation of reactive radical species under reflux conditions, which selectively target the benzylic methyl group without compromising the integrity of the methoxy substituents. The visual progression of the reaction, marked by a distinct color change from blue to yellow-brown, provides a reliable in-process control indicator for operators. This precise control over oxidation states is essential for minimizing impurity profiles, ensuring that the subsequent conversion to the carboxylic acid proceeds with high fidelity. The use of sulfamic acid as a buffering agent during the subsequent positioning oxidation with sodium chlorite further exemplifies the patent's focus on controlling reaction kinetics to prevent over-oxidation or ring degradation.
The final assembly of the Fenprodone molecule relies on a classic yet optimized Friedel-Crafts acylation mechanism mediated by anhydrous ferric chloride. In this step, the electrophilic acyl chloride intermediate attacks the electron-rich aromatic ring of 3,4,5-trimethoxytoluene. The patent specifies rigorous moisture control and the use of chlorobenzene as a high-boiling solvent to drive the equilibrium towards product formation at elevated temperatures around 145°C. Continuous nitrogen sparging is employed to remove evolved hydrogen chloride gas, shifting the reaction equilibrium and preventing acid-catalyzed side reactions. This mechanistic understanding allows for the fine-tuning of catalyst loading, reducing the requirement for stoichiometric Lewis acids that are difficult to remove. The result is a crude product with a significantly cleaner impurity profile, facilitating the final recrystallization step and ensuring the delivery of high-purity Fenprodone suitable for formulation into agricultural fungicides.
How to Synthesize Fenprodone Efficiently
The synthesis of Fenprodone described in this patent represents a sophisticated multi-step organic transformation that requires precise adherence to reaction parameters to ensure safety and yield. The process is divided into the preparation of two key fragments followed by their convergence, necessitating careful management of exothermic events and solvent handling. Operators must maintain strict temperature controls during the diazotization and bromination steps to prevent runaway reactions, while the oxidation phases require monitoring of color changes to determine endpoints accurately. The detailed standardized synthesis steps provided below outline the specific reagent ratios, temperature profiles, and work-up procedures necessary to replicate this high-efficiency route in a pilot or production setting. Adhering to these protocols ensures the consistent production of intermediates that meet the rigorous specifications demanded by the agrochemical industry.
- Prepare 5-bromo-2-methoxy-6-methylbenzoyl chloride via diazotization of 2,3-dimethylaniline, followed by hydrolysis, etherification, selective copper-catalyzed oxidation, and bromination.
- Synthesize 3,4,5-trimethoxytoluene starting from p-cresol through bromination, methylation, and copper-mediated methoxylation.
- Execute the final Friedel-Crafts acylation between the acid chloride and trimethoxytoluene using anhydrous FeCl3, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond simple yield improvements. The elimination of phosphorus pentoxide and the reduction of aluminum waste directly correlate to a significant decrease in hazardous waste disposal costs, which are a major component of the total cost of ownership in fine chemical manufacturing. Furthermore, the ability to recycle solvents like chlorobenzene and methanol creates a closed-loop system that insulates the production process from volatility in raw material pricing. This enhanced resource efficiency translates into substantial cost savings in agrochemical intermediate manufacturing, allowing suppliers to offer more competitive pricing without sacrificing margin. Additionally, the simplified work-up procedures reduce the burden on utility infrastructure, such as wastewater treatment plants, thereby enhancing the overall sustainability rating of the supply chain.
- Cost Reduction in Manufacturing: The transition away from stoichiometric Lewis acids and hazardous phosphorus reagents fundamentally alters the cost structure of Fenprodone production. By utilizing catalytic amounts of ferric chloride and recyclable copper systems, the process drastically reduces the consumption of expensive reagents. The qualitative improvement in atom economy means that less raw material is wasted as byproduct, leading to a more efficient utilization of feedstock. Moreover, the reduced complexity of the purification stages lowers energy consumption associated with distillation and drying, contributing to a leaner manufacturing overhead. These cumulative efficiencies allow for a more robust pricing model that can withstand market fluctuations in commodity chemicals.
- Enhanced Supply Chain Reliability: The reliance on commodity-grade starting materials such as 2,3-dimethylaniline and p-cresol ensures a stable and diversified supply base, mitigating the risk of shortages associated with specialized precursors. The robustness of the reaction conditions, which tolerate minor variations in temperature and addition rates without catastrophic failure, enhances operational reliability. This resilience is crucial for maintaining consistent delivery schedules to downstream formulators who depend on just-in-time inventory models. By minimizing the number of sensitive unit operations, the process reduces the likelihood of batch failures, thereby securing the continuity of supply for critical crop protection products.
- Scalability and Environmental Compliance: The design of this synthesis pathway inherently supports commercial scale-up, with built-in safety features such as controlled gas evolution and manageable exotherms. The explicit focus on solvent recovery and waste minimization aligns perfectly with increasingly stringent global environmental regulations, reducing the regulatory burden on manufacturing sites. This proactive approach to environmental compliance future-proofs the supply chain against potential legislative changes regarding chemical discharge. Consequently, partners adopting this technology can confidently expand production capacity to meet growing global demand for fungicides without facing prohibitive environmental permitting obstacles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Fenprodone synthesis technology. These insights are derived directly from the experimental data and process descriptions found within the patent documentation, providing clarity on the operational realities of this method. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or capacity expansion. The answers reflect a commitment to transparency regarding the capabilities and limitations of the described chemical processes.
Q: How does this new synthesis route address the environmental issues of traditional Fenprodone production?
A: Traditional methods often rely on phosphorus pentoxide or excessive aluminum trichloride, generating significant inorganic waste and eutrophication risks. This patented route utilizes recyclable solvents like chlorobenzene and avoids heavy phosphorus waste, significantly reducing the ecological footprint.
Q: What are the yield advantages of the copper-catalyzed oxidation step compared to conventional oxidants?
A: The patent demonstrates that the selective oxidation using copper sulfate and potassium persulfate achieves high conversion rates with excellent purity, avoiding the over-oxidation and side reactions common with harsher oxidizing agents, thereby improving overall process efficiency.
Q: Is this process suitable for large-scale industrial manufacturing of agrochemical intermediates?
A: Yes, the process is designed for industrial scalability. It features robust temperature controls, solvent recovery systems for chlorobenzene and methanol, and simplified work-up procedures that facilitate safe and continuous commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fenprodone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemicals depends on the seamless integration of innovative chemistry with robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN111943823A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence ensures that the Fenprodone intermediates we supply meet the exacting standards required for global regulatory registration and field performance.
We invite forward-thinking agrochemical companies to collaborate with us to leverage this green synthesis technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your product portfolio. Together, we can drive the next generation of sustainable crop protection solutions to market with speed and confidence.
