Scalable Production of Aromatic Bisimides: Overcoming Purification Bottlenecks in Pharmaceutical Intermediate Manufacturing
Scalable Production of Aromatic Bisimides: Overcoming Purification Bottlenecks in Pharmaceutical Intermediate Manufacturing
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, scalable, and cost-effective synthetic routes. A pivotal advancement in this domain is documented in Chinese Patent CN1124265C, which introduces a robust method for producing aromatic bisimides. This technology addresses a critical pain point in the synthesis of complex heterocyclic compounds: the reliance on labor-intensive and expensive purification techniques like column chromatography. By leveraging a tertiary amine-mediated reaction system, specifically utilizing N-methylmorpholine, this process transforms the production of valuable intermediates used in pharmaceuticals and functional materials. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential for optimizing supply chains and reducing the cost of goods sold (COGS) for high-value aromatic imide derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclic imides, particularly aromatic variants like bis-naphthalimides, has been plagued by significant downstream processing challenges. Traditional methodologies often involve reacting cyclic carboxylic anhydrides with amines in solvents such as DMF, dioxane, or ethanol, frequently requiring high temperatures up to 200°C or the use of strong bases like sodium ethoxide. While these methods can achieve the desired chemical transformation, they often result in crude products laden with impurities that are difficult to remove. As noted in prior art such as WO 94/02466, the resulting bis-naphthalimides typically require purification via column chromatography. In an industrial setting, column chromatography is a major bottleneck; it is slow, consumes vast amounts of silica and solvents, and is notoriously difficult to scale beyond kilogram quantities. This reliance on chromatographic purification renders many otherwise viable synthetic routes economically unfeasible for commercial-scale production, creating a barrier to entry for reliable aromatic bisimide suppliers.
The Novel Approach
The methodology outlined in CN1124265C offers a transformative solution by fundamentally altering the reaction environment and workup procedure. Instead of relying on harsh conditions or difficult purifications, this novel approach utilizes a dicarboxylic acid anhydride reacted with a diamine in the presence of a tertiary amine. The genius of this system lies in the dual role of the tertiary amine, particularly N-methylmorpholine, which serves as both the solvent and the catalyst. This simplifies the reaction matrix significantly. Furthermore, the patent describes a workup procedure that bypasses chromatography entirely. By partially concentrating the reaction mixture and inducing precipitation at low temperatures (0-5°C), the product crystallizes out with high purity. This shift from chromatographic separation to crystallization-based isolation is a game-changer for cost reduction in pharmaceutical intermediates manufacturing, enabling the transition from bench-scale discovery to multi-ton commercial production.
Mechanistic Insights into Tertiary Amine-Catalyzed Imidization
To fully appreciate the technical superiority of this process, one must delve into the mechanistic underpinnings of the tertiary amine-catalyzed imidization. The reaction begins with the nucleophilic attack of the primary amine group from the diamine onto the carbonyl carbon of the dicarboxylic acid anhydride. In conventional solvents, this step can be sluggish or require excessive heat, leading to side reactions. However, in the presence of a tertiary amine like N-methylmorpholine, the reaction kinetics are favorably altered. The tertiary amine likely facilitates the opening of the anhydride ring and stabilizes the intermediate amic acid species, promoting the subsequent cyclodehydration to form the imide ring. The patent specifies that the reaction is typically initiated at mild temperatures between 15-30°C and then slowly raised to reflux. This controlled thermal profile minimizes the formation of thermal degradation byproducts, ensuring a cleaner reaction profile compared to the 200°C methods of the past.
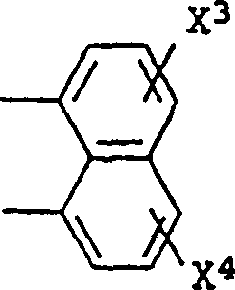
Impurity control is another critical aspect where this mechanism excels. In traditional high-temperature syntheses, the risk of polymerization or over-reaction is significant. By maintaining the reaction in a homogeneous solution of N-methylmorpholine and controlling the temperature ramp, the formation of oligomeric impurities is suppressed. The subsequent precipitation step acts as a powerful purification mechanism. As the solubility of the target bisimide decreases sharply upon cooling to 0-5°C, the pure product selectively crystallizes, leaving soluble impurities in the mother liquor. This phenomenon allows for the production of high-purity aromatic bisimides suitable for sensitive applications, such as fluorescence quenchers or potential anticancer agents, without the need for further extensive purification steps. The structural versatility shown in the patent, allowing for various substituents (X1-X4) like halogens, nitro groups, or alkyl chains, further demonstrates the robustness of this catalytic system across a wide range of substrates.
![Specific example structure of N,N'-bis[2-(1,8-naphthalimido)ethyl]-1,3-diaminopropane synthesized in Example 1](/insights/img/aromatic-bisimide-synthesis-nmm-catalysis-pharma-supplier-20260309220605-02.webp)
How to Synthesize N,N'-bis[2-(1,8-naphthalimido)ethyl]-1,3-diaminopropane Efficiently
Implementing this synthesis requires precise adherence to the stoichiometric and thermal parameters defined in the patent to ensure optimal yield and purity. The process is designed to be operationally simple, making it highly attractive for contract development and manufacturing organizations (CDMOs). The key lies in the molar ratio of the anhydride to the amine, which is maintained at approximately 2:1 to drive the formation of the bis-imide rather than the mono-imide. The use of N-methylmorpholine not only drives the reaction but also simplifies the solvent recovery process, as it can be distilled off and potentially recycled. The following guide outlines the standardized operational protocol derived from the patent examples, providing a clear roadmap for process chemists looking to replicate this efficiency.
- Mix dicarboxylic acid anhydride and diamine in a tertiary amine solvent such as N-methylmorpholine at room temperature.
- Heat the reaction mixture gradually to reflux temperature and maintain for 1 to 2 hours to ensure complete conversion.
- Partially concentrate the mixture, filter hot if necessary, and cool to 0-5°C to precipitate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical advantages of CN1124265C translate directly into tangible business value. The elimination of column chromatography is perhaps the most significant factor, as it removes a major variable cost and time sink from the manufacturing process. Chromatography scales poorly; what works on a gram scale often becomes a logistical nightmare at the kilogram or ton scale. By replacing this with a precipitation and filtration workflow, the process becomes inherently more scalable and predictable. This predictability is crucial for securing long-term supply contracts and ensuring consistent delivery schedules for downstream pharmaceutical clients. Furthermore, the use of N-methylmorpholine, a commercially available and relatively inexpensive reagent, helps keep raw material costs stable and manageable.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the drastic simplification of the purification stage. By avoiding silica gel chromatography, manufacturers save significantly on consumables (silica, large volumes of elution solvents) and labor hours associated with column packing and running. Additionally, the reaction time of approximately 2-4 hours is relatively short for this class of compounds, improving reactor turnover rates. The ability to isolate the product via simple filtration after cooling means that equipment utilization is maximized, and the overall energy consumption per kilogram of product is optimized compared to high-temperature fusion methods or complex distillation setups required for solvent removal in other systems.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The process tolerates a wide range of tertiary amine concentrations and uses common starting materials like naphthalic anhydride and various diamines, which are readily available from global chemical suppliers. This reduces the risk of supply disruptions caused by niche reagent shortages. Moreover, the high purity achieved directly from precipitation reduces the likelihood of batch failures or the need for re-processing, which can severely impact lead times. A reliable aromatic bisimide supplier can leverage this consistency to offer shorter lead times and more dependable inventory levels, crucial for just-in-time manufacturing environments in the pharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method offers distinct advantages. The avoidance of chlorinated solvents often used in chromatography (like dichloromethane) aligns better with modern green chemistry principles and regulatory standards. N-methylmorpholine, while requiring careful handling, is a single solvent system that simplifies waste stream management. The process generates less hazardous waste compared to chromatography-heavy routes, lowering disposal costs and environmental footprint. The scalability is proven by the patent examples which describe reactor sizes up to 50 liters, indicating a clear path to commercial scale-up of complex pharmaceutical intermediates without the engineering hurdles associated with scaling chromatographic separations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aromatic bisimide synthesis technology. These insights are derived directly from the experimental data and claims within patent CN1124265C, providing clarity for technical teams evaluating this route for potential adoption. Understanding these details is vital for assessing the feasibility of integrating this process into existing manufacturing workflows.
Q: How does this process eliminate the need for column chromatography?
A: The process utilizes a specific workup involving partial concentration and low-temperature precipitation (0-5°C), which yields products of sufficiently high purity without the need for complex chromatographic separation.
Q: What is the role of N-methylmorpholine in this reaction?
A: N-methylmorpholine acts as both the reaction solvent and a tertiary amine catalyst, facilitating the imidization reaction between the anhydride and the amine while simplifying the solvent system.
Q: Is this method suitable for industrial scale-up?
A: Yes, the patent explicitly states that the method is designed for industrial implementation, avoiding the bottlenecks of chromatography and utilizing standard heating and filtration equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Bisimide Supplier
The technological breakthroughs detailed in CN1124265C represent a significant opportunity for optimizing the production of high-value chemical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this efficient synthesis route are realized at the volume your business requires. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aromatic bisimide meets the exacting standards of the global pharmaceutical and fine chemical industries.
We invite you to explore how this advanced synthesis method can enhance your supply chain efficiency and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us be your partner in delivering high-quality, cost-effective chemical solutions.
