Advanced Aqueous Synthesis of Oxaspiro Quaternary Ammonium Salts for High-Performance Supercapacitor Electrolytes
The rapid evolution of energy storage technologies has placed immense pressure on the chemical industry to develop electrolytes that offer superior stability and conductivity for next-generation supercapacitors. Patent CN102070651B introduces a groundbreaking methodology for preparing organic electrolyte oxaspiro quaternary ammonium salts, addressing critical purity and scalability challenges faced by traditional synthesis routes. This innovation utilizes water as the primary reaction medium, a strategic shift that fundamentally alters the economic and environmental footprint of producing these high-value electronic chemicals. By leveraging a condensation reaction between cyclic diamines and oxadihaloalkanes or oxadiol disulfonic acid esters, the process achieves exceptional selectivity and yield without relying on volatile organic compounds. For R&D directors and procurement specialists seeking a reliable supercapacitor electrolyte supplier, this technology represents a pivotal advancement in material science. The ability to control impurity profiles down to less than 5ppm ensures that the resulting electrolytes meet the stringent electrochemical stability requirements of modern high-energy density devices. This report analyzes the technical merits and commercial implications of adopting this aqueous-based synthesis for the mass production of battery & energy storage materials.
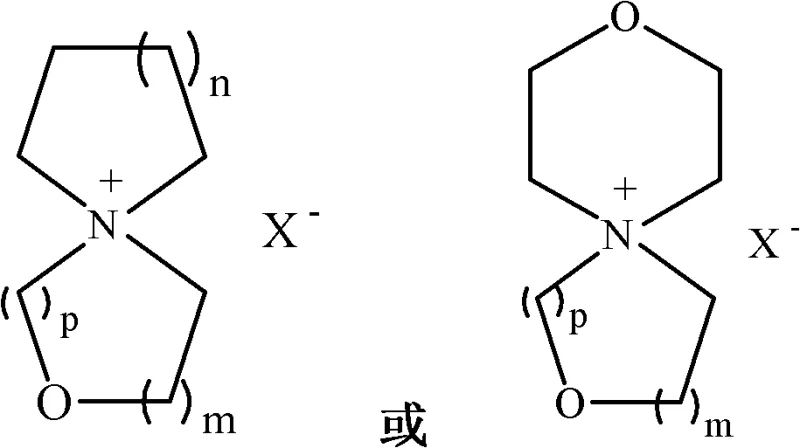
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spirocyclic quaternary ammonium salts has been plagued by significant inefficiencies that hinder large-scale adoption in the electronics sector. Traditional protocols, such as those described in prior art like US 2007-49750 and JP2005-294332, typically rely on nucleophilic substitution reactions conducted in substantial volumes of organic solvents. These conventional methods often suffer from low yields and poor selectivity, necessitating complex and costly downstream purification processes to remove residual halide ions. The presence of halide ions is particularly detrimental in electrochemical applications, as they can corrode current collectors and degrade the overall lifespan of the supercapacitor. Furthermore, the reliance on organic solvents introduces severe safety hazards, including flammability and toxicity, which escalate operational costs related to ventilation, waste disposal, and regulatory compliance. For supply chain heads, these factors translate into unpredictable lead times and higher logistical burdens associated with hazardous material handling. The inability to consistently achieve ultra-low impurity levels using these legacy methods creates a bottleneck for manufacturers aiming to produce high-performance energy storage components that require absolute chemical integrity.
The Novel Approach
In stark contrast to the solvent-intensive legacy processes, the novel approach detailed in the patent data utilizes water as the sole reaction medium, marking a paradigm shift towards greener and more efficient manufacturing. This aqueous-based condensation reaction between cyclic diamines and oxadihaloalkanes proceeds with remarkable efficiency, driven by the use of inorganic bases such as potassium carbonate or sodium hydroxide. The elimination of organic solvents during the primary synthesis step drastically simplifies the isolation of the intermediate sulfonic acid oxaspiro quaternary ammonium salt. Following the initial condensation, a targeted ion exchange step converts the intermediate into the desired tetrafluoroborate or hexafluorophosphate salts, which are critical for high-voltage electrolyte formulations. This method not only enhances the safety profile of the manufacturing facility but also significantly reduces the cost reduction in battery & energy storage materials manufacturing by minimizing solvent recovery and waste treatment expenses. The process is designed to be robust and scalable, allowing for the commercial scale-up of complex electronic chemicals without the technical barriers associated with anhydrous conditions. By integrating a final purification stage involving recrystallization with activated carbon and molecular sieves, the method guarantees a product quality that surpasses industry standards for electrochemical stability.
Mechanistic Insights into Aqueous Condensation and Ion Exchange
The core of this technological breakthrough lies in the precise control of the nucleophilic substitution mechanism within an aqueous environment, which facilitates the formation of the spirocyclic structure with high fidelity. The reaction initiates when the lone pair electrons on the nitrogen atom of the cyclic diamine attack the electrophilic carbon centers of the oxadihaloalkane or oxadiol disulfonic acid ester. The presence of water as a solvent plays a crucial role in stabilizing the transition states and solvating the inorganic base, which acts as an acid scavenger to drive the equilibrium towards product formation. Unlike organic media where solubility issues often limit reaction kinetics, the aqueous system ensures homogeneous mixing of the inorganic reagents, leading to uniform reaction rates and consistent batch-to-batch reproducibility. The subsequent ring-closing step forms the stable oxaspiro quaternary ammonium cation, which is inherently resistant to electrochemical decomposition at high potentials. This mechanistic pathway is critical for R&D teams focusing on the structural feasibility of new electrolyte candidates, as it provides a clear route to modifying the ring sizes (n, m, p) to tune the physical properties of the final salt. Understanding this mechanism allows chemists to optimize reaction parameters such as temperature and molar ratios to maximize yield while suppressing side reactions that could generate electrochemically active impurities.
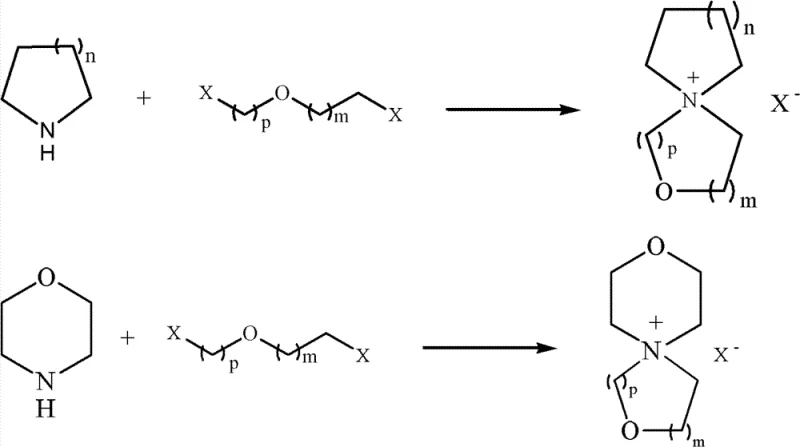
Impurity control is another vital aspect of this mechanism, particularly regarding the removal of halide ions and metal cations that can compromise electrolyte performance. The patent specifies a rigorous purification protocol where the crude product is subjected to recrystallization in alcohols or ketones in the presence of activated carbon and molecular sieves. This step is engineered to adsorb trace organic byproducts and sequester metal ions such as sodium, potassium, iron, and magnesium, which are detected at levels as low as 2.0ppm to 4.5ppm in the final product. The ion exchange process itself is highly selective, utilizing specific salts like sodium tetrafluoroborate or lithium hexafluorophosphate to replace the initial sulfonate or halide anions with electrochemically stable counterparts. This dual strategy of selective synthesis followed by aggressive purification ensures that the impurity content in the electrolyte remains below the critical threshold of 5ppm. For quality assurance managers, this level of control is essential for meeting the stringent specifications required by automotive and grid-scale energy storage applications, where even trace contaminants can lead to catastrophic failure modes over extended cycling.
How to Synthesize Oxaspiro Quaternary Ammonium Salt Efficiently
Implementing this synthesis route requires a systematic approach to reaction engineering, focusing on the optimization of reagent stoichiometry and thermal profiles to ensure maximum efficiency. The process begins with the preparation of an aqueous solution containing the cyclic diamine and the appropriate diester or dihalide, followed by the controlled addition of the inorganic base to initiate the condensation. Maintaining the reaction temperature within the range of 25°C to reflux is critical for balancing reaction rate and selectivity, with typical reaction times spanning from 2 to 16 hours depending on the specific substrates used. After the initial condensation, the mixture is concentrated, and the residue is treated with ethanol to isolate the intermediate salt before proceeding to the ion exchange step. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and purification techniques necessary to replicate the high yields reported in the patent data. Adhering to these protocols allows manufacturers to consistently produce high-purity oxaspiro quaternary ammonium salt suitable for demanding electronic applications.
- Conduct condensation reaction between cyclic diamine and oxadihaloalkane or oxadiol disulfonic acid ester in water with alkali.
- Perform ion exchange on the resulting sulfonic acid oxaspiro quaternary ammonium salt using fluoroboric or hexafluorophosphoric acid salts.
- Purify the final spirocyclic quaternary ammonium salt via recrystallization in alcohol or ketone solvents with activated carbon.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this aqueous synthesis method offers profound advantages that directly address the pain points of procurement managers and supply chain directors in the specialty chemical sector. The primary value driver is the elimination of expensive and hazardous organic solvents from the main reaction phase, which translates into substantial cost savings in terms of raw material procurement and waste management. Traditional methods often require vast quantities of acetonitrile or other polar aprotic solvents, the recovery and disposal of which constitute a significant portion of the operating budget. By switching to water, manufacturers can drastically simplify their infrastructure requirements, reducing the need for explosion-proof equipment and complex solvent recovery distillation columns. This shift not only lowers capital expenditure but also enhances the overall safety culture of the production facility, mitigating the risk of costly shutdowns due to safety incidents. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove byproducts, streamlining the purification workflow and increasing the overall throughput of the manufacturing plant.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental simplification of the chemical workflow, removing the need for costly anhydrous conditions and solvent-intensive workups. By utilizing water as the medium, the process avoids the volatility and price fluctuations associated with petrochemical-derived solvents, providing a more stable cost structure for long-term production planning. The high yield reported in the examples, often exceeding 85% to 90%, ensures that raw material utilization is maximized, reducing the cost per kilogram of the final active ingredient. Additionally, the use of common inorganic bases like potassium carbonate instead of specialized organic bases further drives down the input costs. These factors combine to create a highly competitive cost position for suppliers who adopt this technology, allowing them to offer better pricing to downstream battery manufacturers without compromising on margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the reliance on readily available and non-restricted raw materials such as cyclic diamines and simple diols. Unlike processes that depend on exotic catalysts or regulated solvents, this method utilizes commodity chemicals that are accessible from multiple global sources, reducing the risk of supply disruptions. The robustness of the aqueous reaction conditions means that production is less sensitive to minor variations in environmental controls, ensuring consistent output even in diverse manufacturing locations. This reliability is crucial for reducing lead time for high-purity electrolytes, as it minimizes the need for re-processing batches that fail to meet quality standards. For procurement teams, this translates into more predictable delivery schedules and the ability to secure long-term contracts with confidence, knowing that the supply source is not vulnerable to niche material shortages.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the absence of complex unit operations like cryogenic cooling or high-vacuum distillation. The use of water facilitates heat transfer and mixing in large reactors, making it easier to manage exothermic reactions safely at the 100 MT scale. From an environmental standpoint, the process aligns perfectly with increasingly strict global regulations regarding VOC emissions and hazardous waste generation. The aqueous waste streams generated are easier to treat compared to mixed organic waste, lowering the environmental compliance burden and associated fees. This sustainability profile is becoming a key differentiator in the market, as end-users in the automotive and renewable energy sectors demand greener supply chains. Adopting this technology positions suppliers as leaders in sustainable manufacturing, enhancing their brand value and appeal to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific data points and claims found within the patent documentation. These answers are designed to provide clarity on the feasibility, purity, and scalability of the oxaspiro quaternary ammonium salt production method. Understanding these details is essential for stakeholders evaluating the potential integration of this material into their existing electrolyte formulations or supply networks. The responses reflect the objective capabilities of the process as demonstrated in the experimental examples, ensuring that expectations are aligned with the verified performance metrics.
Q: What is the primary advantage of using water as a reaction medium in this synthesis?
A: Using water eliminates the need for large volumes of hazardous organic solvents, significantly reducing environmental impact and safety risks while simplifying the workup process.
Q: How does this method ensure the low impurity levels required for supercapacitors?
A: The process achieves high selectivity and includes a rigorous purification step involving activated carbon and molecular sieves, ensuring impurity content remains below 5ppm.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the method avoids complex anhydrous conditions and uses common inorganic bases, making it highly suitable for large-scale commercial manufacturing with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaspiro Quaternary Ammonium Salt Supplier
As the demand for high-performance supercapacitors continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. We understand the critical nature of stringent purity specifications in energy storage applications and operate rigorous QC labs to verify that every batch meets the <5ppm impurity threshold. Our commitment to technical excellence allows us to adapt complex aqueous condensation routes to fit your specific volume requirements while maintaining the highest standards of quality and safety. By leveraging our infrastructure, you can accelerate your time-to-market for next-generation electrolyte solutions without the capital risk of building new production lines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce overall manufacturing costs. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits applicable to your operation. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project goals. Whether you require pilot-scale quantities for R&D validation or full commercial tonnage for mass production, NINGBO INNO PHARMCHEM is equipped to support your growth in the evolving energy storage market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
