Advanced Synthesis of Halogen-Free Quaternary Ammonium Salts for High-Performance Supercapacitor Electrolytes
Advanced Synthesis of Halogen-Free Quaternary Ammonium Salts for High-Performance Supercapacitor Electrolytes
The rapid evolution of energy storage technologies has placed immense pressure on the supply chain for high-performance electronic chemicals, particularly those serving the supercapacitor sector. Patent CN102432474B introduces a transformative methodology for the preparation of high-purity alkylammonium tetrafluoroborate, a critical component in organic electrolytes. This technical breakthrough addresses the longstanding challenges of halogen contamination and equipment corrosion that have historically plagued the manufacturing of quaternary ammonium salts. By leveraging a one-step reaction between tertiary amines and alkoxonium tetrafluoroborate salts, this process delivers a product profile that meets the rigorous demands of modern high-energy density capacitors. For R&D Directors and Procurement Managers seeking a reliable supercapacitor electrolyte supplier, understanding the nuances of this halogen-free synthesis is paramount for securing a competitive advantage in the energy storage market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tetrafluoroborate quaternary ammonium salts has relied heavily on routes involving hazardous and corrosive reagents such as anhydrous hydrogen fluoride or boron trifluoride. These traditional methodologies, while chemically viable, introduce significant operational risks and hidden costs into the manufacturing workflow. The aggressive nature of fluoroboric acid and hydrogen fluoride necessitates the use of specialized corrosion-resistant equipment, which drastically increases capital expenditure and maintenance overheads. Furthermore, these routes often struggle to completely eliminate halogen ions from the final product matrix, leading to electrolytes that can cause increased internal resistance and gas swelling in supercapacitor cells. The presence of trace chloride or other halide impurities compromises the electrochemical stability and safety profile of the energy storage device, making these conventional methods increasingly untenable for high-end applications.
The Novel Approach
In stark contrast to the hazardous legacy processes, the novel approach detailed in the patent utilizes a clean, one-step alkylation strategy that fundamentally reshapes the production landscape. By reacting tertiary amines directly with alkoxonium tetrafluoroborate salts, such as triethoxonium tetrafluoroborate, the process bypasses the need for free fluoroboric acid or hydrogen fluoride entirely. This shift not only mitigates the safety risks associated with handling corrosive gases but also simplifies the downstream purification requirements significantly. The reaction proceeds with high selectivity and conversion rates under moderate temperature conditions, typically ranging from 0°C to 150°C, allowing for precise control over the reaction kinetics. This methodological pivot represents a significant leap forward in cost reduction in electronic chemical manufacturing, as it reduces the complexity of waste treatment and extends the lifespan of standard reactor vessels.
Mechanistic Insights into Alkoxonium-Mediated Quaternization
The core of this technological advancement lies in the nucleophilic substitution mechanism where the tertiary amine acts as a nucleophile attacking the alkyl group of the alkoxonium salt. This reaction pathway is highly efficient because the alkoxonium species serves as an excellent leaving group, facilitating the formation of the quaternary ammonium cation without generating acidic byproducts. The absence of acid generation is critical, as it prevents the degradation of sensitive functional groups that might be present on more complex amine substrates. The reaction stoichiometry is flexible, with molar ratios of tertiary amine to alkoxonium salt ranging from 1:1 to 1:4, allowing process engineers to optimize for yield or raw material consumption based on specific economic constraints. This mechanistic clarity provides a robust foundation for scaling the process from laboratory benchtop to commercial production volumes without encountering the unpredictable side reactions common in acid-catalyzed routes.
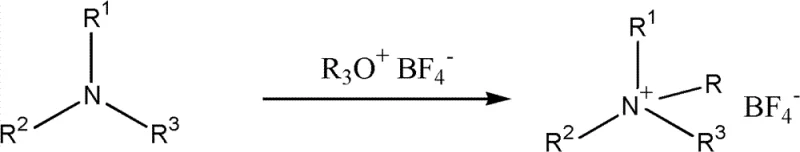
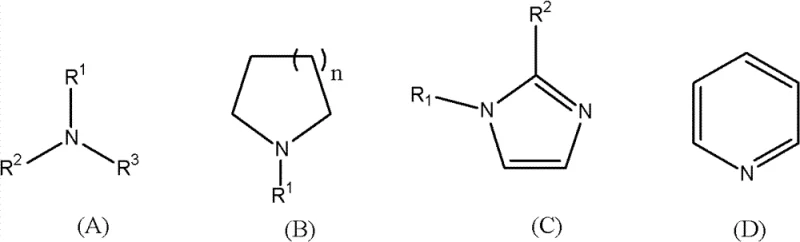
Beyond the primary reaction mechanism, the versatility of this synthesis is demonstrated by its compatibility with a wide array of amine structures, including chain, cyclic, and heterocyclic variants. The ability to accommodate diverse substrates means that manufacturers can produce a broad portfolio of electrolytes tailored to specific voltage and conductivity requirements. Impurity control is inherently built into the chemistry, as the byproducts of the reaction are typically neutral alcohols or ethers which are easily separated from the ionic product. This inherent selectivity ensures that the final electrolyte salt possesses the ultra-low metal content required for high-performance applications, with ion chromatography data confirming the absence of detectable halogen ions. Such purity levels are essential for minimizing self-discharge rates and maximizing the cycle life of the supercapacitor modules in which they are deployed.
How to Synthesize High-Purity Quaternary Ammonium Salts Efficiently
Implementing this synthesis route requires careful attention to solvent selection and atmospheric control to maintain the integrity of the reactants. The process is typically conducted under a nitrogen atmosphere to prevent moisture ingress, which could hydrolyze the alkoxonium salt and reduce overall yield. Solvents such as dichloromethane, chloroform, or ethyl acetate are preferred due to their ability to dissolve both the amine and the salt while facilitating easy removal during the workup phase. Following the reaction, a purification step involving activated carbon and molecular sieves is employed to adsorb any trace colored impurities or residual moisture. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols.
- Prepare the reaction vessel under a nitrogen atmosphere and charge with a suitable organic solvent such as dichloromethane or chloroform.
- Add the tertiary amine substrate and the alkoxonium tetrafluoroborate salt in a molar ratio ranging from 1: 1 to 1:4.
- Maintain the reaction temperature between 0°C and 150°C, followed by purification using activated carbon and molecular sieves to ensure ultra-low metal content.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this halogen-free synthesis route offers tangible strategic benefits that extend beyond simple chemical performance. The elimination of corrosive reagents like hydrogen fluoride removes a major bottleneck in the supply chain, as it reduces reliance on specialized hazardous material transport and storage infrastructure. This simplification translates directly into enhanced supply chain reliability, as the production process becomes less susceptible to regulatory delays or safety incidents that often plague facilities handling extreme hazards. Furthermore, the use of readily available tertiary amines and alkoxonium salts ensures a stable raw material base, reducing the risk of supply disruptions that can occur with more exotic fluorinating agents. This stability is crucial for maintaining continuous production schedules in the fast-paced electronic materials sector.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the reduction in capital and operational expenditures associated with equipment maintenance. By avoiding the use of highly corrosive acids, manufacturers can utilize standard glass-lined or stainless-steel reactors rather than expensive Hastelloy or Teflon-lined vessels, leading to substantial cost savings over the lifecycle of the plant. Additionally, the simplified purification process reduces the consumption of energy and auxiliary materials, further driving down the unit cost of the final electrolyte salt. The high yield and selectivity of the reaction also minimize raw material waste, ensuring that a greater proportion of input costs are converted into saleable product. These factors combine to create a highly competitive cost structure that allows for aggressive pricing strategies without compromising margin.
- Enhanced Supply Chain Reliability: From a logistics perspective, the safer nature of the reactants simplifies the regulatory compliance burden associated with transporting hazardous chemicals. This ease of transport allows for a more flexible sourcing strategy, enabling procurement teams to qualify multiple suppliers for key raw materials without being constrained by strict hazardous material handling certifications. The robustness of the reaction conditions also means that production can be scaled up or down rapidly in response to market demand fluctuations, providing a buffer against supply shocks. Reducing lead time for high-purity electrolytes becomes feasible when the manufacturing process is not bottlenecked by complex safety protocols or lengthy purification cycles. This agility is a critical asset in a market where time-to-market for new energy storage devices is increasingly compressed.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns well with modern sustainability goals, as it generates less hazardous waste compared to traditional fluorination methods. The absence of acidic off-gases simplifies the exhaust treatment requirements, reducing the load on scrubber systems and lowering the overall environmental footprint of the facility. This compliance advantage is increasingly important as global regulations on industrial emissions become more stringent, protecting the manufacturer from future regulatory risks. The commercial scale-up of complex organic salts via this route is straightforward, as the reaction exotherm is manageable and the workup procedures are compatible with standard industrial filtration and drying equipment. This scalability ensures that the technology can meet the growing global demand for supercapacitor electrolytes driven by the electrification of transport and renewable energy integration.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this material into their supply chain or product development roadmap. The answers reflect the consensus on best practices for handling and utilizing high-purity quaternary ammonium tetrafluoroborates in industrial settings.
Q: Why is the absence of halogen ions critical for supercapacitor electrolytes?
A: Halogen ions, such as chloride, can significantly increase the internal resistance of supercapacitors and cause gas swelling during operation. The patented method ensures the final product is free from halogen ions, thereby enhancing electrochemical performance and safety reliability.
Q: How does this method improve upon conventional synthesis routes?
A: Conventional methods often utilize corrosive reagents like anhydrous hydrogen fluoride or boron trifluoride, which complicate equipment requirements and introduce impurities. This novel approach uses alkoxonium salts in a one-step reaction, eliminating corrosion risks and simplifying the purification process.
Q: What is the typical purity profile achievable with this synthesis?
A: The process yields products with extremely low metal impurity levels, typically in the low ppm range for sodium, potassium, and iron, as verified by ion chromatography. This high purity is essential for meeting the stringent specifications of high-performance energy storage devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quaternary Ammonium Tetrafluoroborate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced electrolyte chemistries requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot trials to full-scale supply. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs that verify every batch against the highest industry standards for metal content and halogen levels. Our commitment to quality ensures that the electrolytes you receive will perform consistently in your supercapacitor applications, minimizing the risk of field failures.
We invite you to engage with our technical procurement team to discuss how this halogen-free synthesis route can be optimized for your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits this technology offers compared to your current supply base. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecular structures. Let us help you secure a stable, high-quality supply of critical electronic chemicals that will power the next generation of energy storage solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
