Advanced Synthesis of Bistriphenanthrofuran Discotic Liquid Crystals for Optoelectronic Applications
Introduction to Next-Generation Discotic Liquid Crystal Technology
The rapid evolution of organic electronics demands materials that combine superior charge transport properties with processability, a challenge effectively addressed by the innovations detailed in patent CN114573532A. This intellectual property introduces a novel class of bistriphenanthrofuran discotic liquid crystal compounds, characterized by their ability to self-assemble into stable hexagonal columnar mesophases over a wide temperature range. These materials are not merely theoretical constructs but represent a tangible leap forward for the Display & Optoelectronic Materials sector, offering absolute quantum yields reaching up to 60% and exceptional gelation properties in various organic solvents. For R&D directors seeking high-performance organic semiconductors, the structural integrity and photophysical characteristics of these compounds provide a robust foundation for developing advanced photovoltaic and electroluminescent devices. The core innovation lies in the specific molecular architecture that balances rigid aromatic cores with flexible alkoxy chains, facilitating ordered stacking essential for efficient carrier mobility.
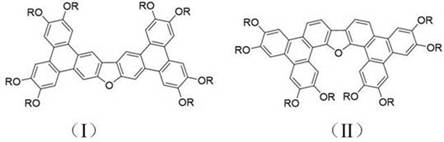
Furthermore, the patent elucidates a synthesis strategy that bypasses the traditional limitations associated with constructing complex polycyclic aromatic hydrocarbons. By leveraging a combination of Suzuki cross-coupling and mild oxidative cyclization, the methodology ensures that the final products maintain high structural fidelity with minimal defect formation. This is particularly crucial for a reliable OLED material supplier aiming to deliver consistent batch-to-batch performance. The ability of these compounds to form nano-microstructures through self-assembly directly translates to improved film-forming capabilities, a key requirement for solution-processable organic electronics. As the industry moves towards more sustainable and efficient manufacturing processes, the温和 (mild) conditions described in this patent position these materials as a prime candidate for next-generation commercial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of large polycyclic aromatic systems often relied on harsh Scholl reaction conditions or high-temperature pyrolysis, which frequently resulted in uncontrolled polymerization, skeletal rearrangements, and significant formation of regioisomeric impurities. These conventional pathways typically require aggressive Lewis acids and elevated temperatures that can degrade sensitive functional groups, leading to poor yields and complex purification challenges that inflate production costs. For procurement managers, such inefficiencies manifest as volatile pricing and extended lead times due to the difficulty in sourcing high-purity intermediates that survive these brutal reaction environments. Moreover, the lack of stereochemical control in traditional fusion methods often necessitates extensive chromatographic separation, generating substantial chemical waste and complicating the environmental compliance profile of the manufacturing process. The resulting materials often suffer from batch-to-batch variability in their liquid crystalline properties, undermining their reliability in precision electronic applications where consistent charge carrier mobility is non-negotiable.
The Novel Approach
In stark contrast, the methodology outlined in CN114573532A employs a modular synthetic strategy centered on palladium-catalyzed Suzuki coupling followed by a controlled iron(III) chloride-mediated oxidative cyclization. This approach allows for the precise construction of the bistriphenanthrofuran core under remarkably mild conditions, specifically at room temperature, which preserves the integrity of the peripheral alkoxy chains essential for solubility and mesophase formation. By decoupling the carbon-carbon bond formation from the ring-closing step, chemists can optimize each stage independently, ensuring high conversion rates and minimizing side reactions that plague one-pot fusion strategies. This strategic shift not only enhances the overall yield but also simplifies the downstream processing, making it an ideal candidate for cost reduction in electronic chemical manufacturing. The use of commercially available catalysts and solvents further democratizes access to these advanced materials, enabling a more resilient supply chain capable of meeting the growing demand for high-performance organic semiconductors without compromising on quality or sustainability.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The cornerstone of this synthesis is the final intramolecular oxidative cyclization step, where the pre-assembled biaryl precursors are fused into the rigid bistriphenanthrofuran framework using ferric chloride (FeCl3) as the oxidant. Mechanistically, this reaction proceeds via a single-electron transfer (SET) pathway, where FeCl3 generates radical cation intermediates on the electron-rich aromatic rings, prompting nucleophilic attack by adjacent aryl groups to form the new carbon-carbon bonds. The choice of a mixed solvent system comprising chloroform and nitromethane is critical, as it modulates the oxidation potential of the iron species, preventing over-oxidation while ensuring complete ring closure. This delicate balance is what allows the reaction to proceed efficiently at room temperature, avoiding the thermal stress that typically leads to decomposition in similar polycyclic syntheses. For technical teams, understanding this mechanism is vital for troubleshooting and optimizing reaction parameters, such as the stoichiometric ratio of FeCl3 to substrate, which the patent specifies at a precise 1:6 molar ratio to drive the reaction to completion.
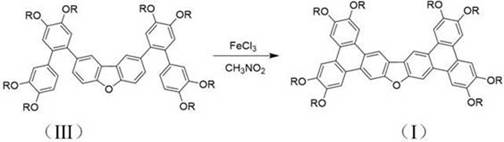
Impurity control in this stage is achieved through the inherent selectivity of the radical cation mechanism, which favors intramolecular cyclization over intermolecular coupling due to entropic factors and the specific geometry of the precursor. The resulting crude product, often a tan or white solid depending on the specific isomer, can be readily purified via standard silica gel chromatography and recrystallization techniques, yielding materials with the high purity required for electronic applications. The patent data indicates that this method consistently produces compounds with well-defined liquid crystalline phases, evidenced by characteristic DSC curves and polarized light textures, confirming the successful formation of the target columnar structures. This level of mechanistic control ensures that the final product exhibits the desired optoelectronic properties, such as high fluorescence quantum yield and stable charge transport, making it a superior choice for developers of organic field-effect transistors and light-emitting diodes.
How to Synthesize Bistriphenanthrofuran Efficiently
The synthesis of these high-value discotic liquid crystals follows a logical four-step sequence that begins with the functionalization of biphenyl precursors and culminates in the oxidative ring closure. The process starts with the selective bromination of tetraalkoxy-biphenyls, followed by conversion to boronic esters, which then undergo Suzuki coupling with dibromofuran derivatives to build the linear backbone. The final transformation involves the aforementioned FeCl3-mediated cyclization, which locks the structure into its final planar conformation. Detailed operational parameters, including specific molar ratios, solvent volumes, and reaction times, are critical for reproducing the high yields reported in the patent examples. For laboratory teams aiming to replicate this chemistry, adherence to the specified inert atmosphere conditions during the palladium-catalyzed steps is essential to prevent catalyst deactivation and ensure optimal coupling efficiency.
- Perform bromination of tetraalkoxy-biphenyl precursors using molecular bromine at room temperature to generate the aryl bromide intermediate.
- Execute a palladium-catalyzed borylation reaction to convert the aryl bromide into the corresponding pinacol boronate ester.
- Conduct Suzuki coupling with dibromofuran derivatives followed by FeCl3-mediated oxidative cyclization to form the final fused ring system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for supply chain stability and cost management, primarily driven by the simplicity and robustness of the chemical transformations involved. The reliance on commodity chemicals such as ferric chloride, potassium carbonate, and common organic solvents like THF and dichloromethane means that raw material sourcing is not constrained by geopolitical bottlenecks or specialized vendor dependencies. This accessibility translates directly into enhanced supply chain reliability, allowing manufacturers to maintain consistent production schedules even during periods of market volatility. Furthermore, the mild reaction conditions, particularly the room temperature cyclization, drastically reduce energy consumption compared to traditional high-temperature fusion methods, contributing to significant operational cost savings and a lower carbon footprint for the manufacturing facility. These factors collectively make the production of bistriphenanthrofuran derivatives a economically viable proposition for large-scale commercialization.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the final cyclization step, replacing them with inexpensive iron salts, removes the need for costly heavy metal scavenging processes that are often required to meet pharmaceutical or electronic grade purity standards. This simplification of the purification workflow reduces both the consumption of specialized resins and the time spent on downstream processing, leading to substantial cost savings per kilogram of finished product. Additionally, the high yields reported in the patent examples minimize raw material waste, ensuring that the input costs are efficiently converted into saleable inventory. By streamlining the synthetic route, manufacturers can achieve a more competitive pricing structure, making these advanced materials accessible for a broader range of optoelectronic applications without sacrificing margin.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis, which relies on well-established cross-coupling chemistry, allows for the decoupling of intermediate production from final assembly, providing flexibility in inventory management. If demand fluctuates, manufacturers can stockpile stable intermediates like the boronic ester or the linear furan precursor, which have longer shelf lives than the final reactive species, thereby buffering against supply shocks. The use of standard equipment such as round-bottom flasks and simple filtration setups means that production can be easily scaled across multiple facilities without requiring bespoke reactor designs or specialized infrastructure. This adaptability ensures that a reliable OLED material supplier can meet urgent customer requests and maintain continuity of supply even in the face of unexpected logistical challenges or equipment maintenance downtime.
- Scalability and Environmental Compliance: The aqueous workup procedures and the use of recyclable solvents align with modern green chemistry principles, simplifying waste treatment and reducing the environmental burden associated with chemical manufacturing. The absence of highly toxic reagents or extreme pressure conditions lowers the safety risk profile of the plant, potentially reducing insurance premiums and regulatory compliance costs. As the industry faces increasing pressure to adopt sustainable practices, this synthesis route offers a clear pathway to producing high-performance materials with a minimized ecological impact. The ability to scale this process from gram-scale laboratory synthesis to multi-ton commercial production is supported by the linear scalability of the batch reactions, ensuring that quality remains consistent regardless of the production volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of bistriphenanthrofuran discotic liquid crystals, based on the specific data provided in the patent literature. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating these materials into their product pipelines. The answers reflect the practical realities of scaling this chemistry while maintaining the high standards required for electronic grade materials.
Q: What are the key advantages of the FeCl3 cyclization method described in CN114573532A?
A: The method utilizes mild reaction conditions at room temperature, significantly reducing energy consumption and minimizing thermal degradation of sensitive intermediates compared to high-temperature fusion methods.
Q: What is the expected purity profile for these discotic liquid crystal compounds?
A: The process employs rigorous purification steps including silica gel column chromatography and recrystallization, ensuring high purity suitable for semiconductor applications where charge transport efficiency is critical.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the use of standard reagents like FeCl3 and common solvents such as chloroform and THF, combined with straightforward workup procedures, supports robust commercial scale-up capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bistriphenanthrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis technology described in CN114573532A and are fully equipped to bring these advanced discotic liquid crystals from the laboratory to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of major display and semiconductor manufacturers. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of bistriphenanthrofuran meets the exacting standards necessary for high-efficiency charge transport and luminescence. Our commitment to quality assurance means that clients receive materials with consistent particle size, purity profiles, and mesomorphic behavior, eliminating the variability that often plagues early-stage organic electronic materials.
We invite potential partners to engage with our technical procurement team to discuss how we can tailor our production capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall material costs while improving supply security. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments for your projects, allowing you to make informed decisions based on hard data rather than theoretical projections. Together, we can accelerate the commercialization of next-generation organic electronics by ensuring a stable, high-quality supply of these critical functional materials.
