Scalable Palladium-Free Synthesis of 3-Methylbenzoxazolone Boronic Ester for Pharmaceutical Applications
Introduction to Advanced Benzoxazolone Intermediate Manufacturing
The pharmaceutical industry continuously demands more efficient and cost-effective pathways for synthesizing complex heterocyclic intermediates, particularly those serving as key building blocks for next-generation therapeutics. Patent CN116396316A introduces a groundbreaking preparation method for 3-methylbenzo[d]oxazol-2(3H)-one-5-boronic acid pinacol ester (CAS: 1220696-32-1), a critical organic砌块 used in the synthesis of dipeptidyl peptidase 1 (DPP-1) inhibitors for treating obstructive airway diseases. This technology represents a significant paradigm shift by abandoning traditional palladium-catalyzed cross-coupling reactions in favor of a streamlined, metal-free nucleophilic substitution and cyclization strategy. By leveraging readily available starting materials such as 4-hydroxyphenylboronic acid pinacol ester, the process achieves a total yield exceeding 81% and a final purity greater than 99%, addressing both economic and quality constraints faced by modern API manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of boron-functionalized benzoxazolones has relied heavily on transition metal-catalyzed borylation reactions, specifically the Suzuki-Miyaura coupling or direct C-H borylation. As illustrated in prior art such as WO2018/93569A1, the conventional approach typically involves reacting a halogenated benzoxazolone precursor with bis(pinacolato)diboron using expensive catalysts like PdCl2(dppf)2·CH2Cl2. 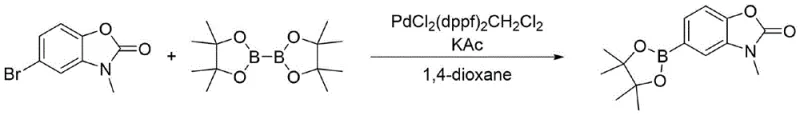 These legacy methods suffer from inherent economic inefficiencies due to the high cost of palladium complexes and specialized phosphine ligands. Furthermore, the presence of residual heavy metals in the final product poses a severe regulatory hurdle, necessitating additional purification steps such as scavenger resin treatment or repeated chromatography, which drastically reduce overall throughput and increase waste generation. Alternative routes, such as those described in US2015/210655A1, utilize Pd(OAc)2 and X-Phos ligands, which, while offering moderate yields around 90%, often struggle with consistency in purity and content, requiring further downstream processing that complicates the supply chain.
These legacy methods suffer from inherent economic inefficiencies due to the high cost of palladium complexes and specialized phosphine ligands. Furthermore, the presence of residual heavy metals in the final product poses a severe regulatory hurdle, necessitating additional purification steps such as scavenger resin treatment or repeated chromatography, which drastically reduce overall throughput and increase waste generation. Alternative routes, such as those described in US2015/210655A1, utilize Pd(OAc)2 and X-Phos ligands, which, while offering moderate yields around 90%, often struggle with consistency in purity and content, requiring further downstream processing that complicates the supply chain.
The Novel Approach
In stark contrast, the methodology disclosed in CN116396316A circumvents these bottlenecks by constructing the benzoxazolone core after establishing the boronic ester functionality, rather than installing the boron group onto a pre-formed heterocycle. This reverse synthetic logic utilizes 4-hydroxyphenylboronic acid pinacol ester as a stable and commercially accessible starting scaffold. The process involves a strategic sequence of Boc-protection, regioselective halogenation, and finally, a high-pressure aminolysis-cyclization cascade. This approach completely eliminates the need for precious metal catalysts, thereby removing the associated cost burden and metal contamination risks. The operational simplicity is enhanced by the use of common organic solvents and straightforward workup procedures, such as filtration and slurry washing, which are highly amenable to large-scale industrial implementation. This structural innovation not only improves the economic profile but also ensures a cleaner impurity profile, delivering a high-purity intermediate ready for subsequent coupling reactions in drug discovery pipelines.
Mechanistic Insights into the Metal-Free Cyclization Strategy
The core of this technological advancement lies in the clever manipulation of protecting groups and nucleophilic reactivity to forge the oxazolone ring without metal assistance. The synthesis initiates with the protection of the phenolic hydroxyl group of the starting boronic ester using di-tert-butyl dicarbonate (Boc2O) in the presence of DMAP. This step is crucial as it masks the reactive phenol, preventing unwanted side reactions during the subsequent halogenation phase. Following protection, the aromatic ring undergoes electrophilic substitution using N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS). The electron-donating nature of the Boc-oxy group directs the halogen specifically to the ortho-position (3-position relative to the oxygen), setting the stage for ring closure. 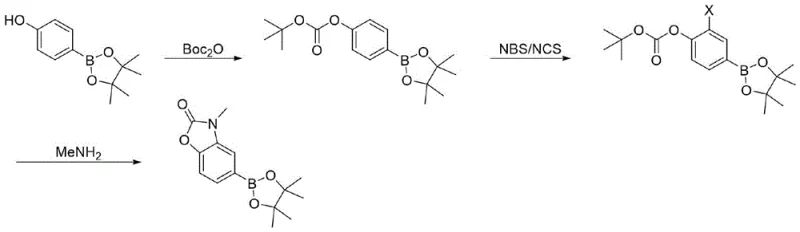 The final transformation is a remarkable intramolecular cyclization where the protected carbamate serves as the carbonyl source. Under high-pressure conditions (13-15 atm) with aqueous methylamine, the Boc group is cleaved, and the resulting amine attacks the carbonyl carbon, displacing the tert-butyl group and closing the five-membered oxazolone ring. This tandem deprotection-cyclization mechanism is highly efficient, driving the reaction to completion with minimal byproduct formation.
The final transformation is a remarkable intramolecular cyclization where the protected carbamate serves as the carbonyl source. Under high-pressure conditions (13-15 atm) with aqueous methylamine, the Boc group is cleaved, and the resulting amine attacks the carbonyl carbon, displacing the tert-butyl group and closing the five-membered oxazolone ring. This tandem deprotection-cyclization mechanism is highly efficient, driving the reaction to completion with minimal byproduct formation.
From an impurity control perspective, this route offers distinct advantages over metal-catalyzed alternatives. The absence of palladium eliminates the risk of metal-ligand complexes persisting in the final API, a common failure mode in traditional borylation. The primary impurities generated are typically succinimide derivatives from the halogenation step or unreacted starting materials, both of which are polar and easily removed via aqueous washes or crystallization. The use of 40% aqueous methylamine in the final step ensures that any acidic byproducts are neutralized, and the high pressure facilitates the solubility of the gaseous amine in the organic phase, promoting uniform reaction kinetics. This mechanistic robustness translates directly to the reported HPLC purity of >99%, ensuring that the intermediate meets the stringent specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize 3-Methylbenzoxazolone Boronic Ester Efficiently
The execution of this synthesis requires precise control over reaction stoichiometry and pressure conditions to maximize yield and safety. The process is divided into three distinct operational stages, each optimized for scalability. The initial protection step is exothermic and requires controlled addition of Boc2O to maintain temperature stability, followed by a thermal workup to crystallize the intermediate. The halogenation step demands careful batch-wise addition of the halogenating agent to manage heat release and ensure mono-substitution selectivity. Finally, the cyclization step necessitates the use of a rated pressure vessel capable of withstanding 15 atm at elevated temperatures (110-130°C). Detailed standardized operating procedures for each stage, including specific solvent swaps and quenching protocols, are essential for reproducible commercial production.
- Protect 4-hydroxyphenylboronic acid pinacol ester with di-tert-butyl dicarbonate (Boc2O) using DMAP catalyst in dichloromethane to form the Boc-protected intermediate.
- Perform electrophilic aromatic substitution using N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS) in acetonitrile to introduce the halogen at the 3-position.
- Execute high-pressure cyclization with 40% aqueous methylamine and a base catalyst (TEA or DBU) at 110-130°C to close the oxazolone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this palladium-free route offers transformative benefits regarding cost structure and supply reliability. The most immediate impact is the drastic reduction in raw material costs associated with the elimination of palladium catalysts and specialized ligands, which are subject to volatile market pricing and geopolitical supply risks. By switching to commodity chemicals like Boc2O, NBS, and methylamine, manufacturers can stabilize their Bill of Materials (BOM) and insulate themselves from precious metal shortages. Furthermore, the simplified purification process reduces the consumption of expensive scavenger resins and silica gel, lowering the overall cost of goods sold (COGS) and minimizing hazardous waste disposal fees. This economic efficiency makes the final API more competitive in price-sensitive markets.
- Cost Reduction in Manufacturing: The removal of palladium catalysts fundamentally alters the cost equation of producing this intermediate. Traditional methods incur significant expenses not only for the catalyst purchase but also for the analytical testing required to verify residual metal levels below ppm thresholds. By adopting this metal-free pathway, facilities can bypass these costly QC assays and metal removal unit operations. The high yield of each individual step (97.6% for protection, ~95% for halogenation, and ~87% for cyclization) compounds to a superior overall mass balance, meaning less raw material is wasted per kilogram of finished product. This efficiency gain directly translates to substantial cost savings that can be passed down the supply chain or retained as margin.
- Enhanced Supply Chain Reliability: Reliance on specialized catalysts often creates single points of failure in the supply chain; if a specific ligand supplier faces disruption, production halts. This new method utilizes globally available bulk chemicals with multiple qualified vendors, ensuring business continuity. The starting material, 4-hydroxyphenylboronic acid pinacol ester, is a stable solid that is easy to transport and store, unlike sensitive organometallic reagents. Additionally, the robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating demand from downstream pharmaceutical clients without long lead times for catalyst procurement.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is significantly greener. Eliminating heavy metals reduces the toxicity of the effluent stream, simplifying wastewater treatment and helping facilities meet increasingly strict environmental regulations. The solvents used, such as dichloromethane and acetonitrile, are standard industrial solvents with well-established recovery and recycling protocols. The high atom economy of the cyclization step, where the Boc group acts as an internal reagent, minimizes waste generation. This alignment with green chemistry principles enhances the sustainability profile of the supply chain, a key metric for modern ESG-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic value of this technology for potential licensees and manufacturing partners.
Q: Why is this palladium-free route superior to traditional Suzuki coupling methods?
A: Traditional methods rely on expensive palladium catalysts like PdCl2(dppf)2 or Pd(OAc)2/X-Phos, which increase raw material costs and require complex heavy metal removal steps to meet pharmaceutical purity standards. This novel route eliminates transition metals entirely, simplifying purification and significantly reducing production costs while achieving >99% purity.
Q: What is the overall yield and purity profile of this new manufacturing process?
A: The patented process demonstrates a robust total yield exceeding 81% across three steps, with individual step yields ranging from 86% to 97%. The final product consistently achieves HPLC purity greater than 99%, making it suitable for direct use in sensitive medicinal chemistry applications without extensive recrystallization.
Q: Is this process scalable for commercial kilogram-to-ton production?
A: Yes, the process utilizes common industrial solvents like dichloromethane and acetonitrile and avoids cryogenic conditions or highly sensitive reagents. The final cyclization step uses standard high-pressure autoclave technology (13-15 atm), which is readily scalable in modern cGMP facilities for commercial supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylbenzoxazolone Boronic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines. Our technical team has thoroughly analyzed the pathway described in CN116396316A and validated its potential for seamless technology transfer. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is smooth and compliant. Our state-of-the-art facilities are equipped with high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-methylbenzo[d]oxazol-2(3H)-one-5-boronic acid pinacol ester delivered meets the highest industry standards.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and robust synthetic route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can optimize your supply chain for DPP-1 inhibitor programs and other therapeutic applications.
