Scalable Synthesis of C5 Acetal Sulfone for High-Purity Vitamin D3 Intermediates
Scalable Synthesis of C5 Acetal Sulfone for High-Purity Vitamin D3 Intermediates
The pharmaceutical industry constantly seeks robust and efficient pathways for synthesizing critical intermediates, particularly for high-value nutrients like Vitamin D3. Patent CN113979901A introduces a groundbreaking preparation method for C5 acetal sulfone, chemically known as 2-methyl-2-(1-ethoxyethoxy)-4-(phenylsulfonyl)butane. This compound serves as a pivotal building block in the synthesis of 25-hydroxy vitamin D3, a metabolite with significantly higher biological activity and absorption rates compared to standard Vitamin D3. The disclosed technology addresses long-standing challenges in the field by offering a streamlined, two-step synthetic route that bypasses the need for hazardous organometallic reagents. By leveraging mild reaction conditions and readily available industrial raw materials, this innovation promises to enhance the reliability of the supply chain for pharmaceutical intermediates while adhering to strict environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the C5 fragment required for Vitamin D3 analogues has relied heavily on the use of Grignard reagents, organic zinc reagents, or phosphorus ylides. These traditional methodologies are fraught with significant operational drawbacks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Grignard reactions, for instance, demand strictly anhydrous conditions and often require cryogenic temperatures to control exothermicity and prevent side reactions, leading to high energy consumption and safety risks. Furthermore, the handling of pyrophoric materials necessitates specialized equipment and rigorous safety protocols, which drastically increase capital expenditure and operational complexity. The resulting processes often suffer from moderate yields and generate substantial amounts of hazardous waste, complicating disposal and increasing the environmental footprint of the manufacturing facility.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a strategic sequence of hydroxyl acetalization followed by nucleophilic substitution. This method replaces unstable organometallic species with stable, easy-to-handle reagents like vinyl ethyl ether and sodium benzene sulfinate. The reaction conditions are remarkably mild, proceeding effectively at temperatures ranging from 0°C to 80°C, which significantly reduces energy requirements and thermal stress on the equipment. By avoiding the use of toxic heavy metals and eliminating the generation of hazardous byproducts, this route aligns perfectly with modern green chemistry initiatives. The simplicity of the workup procedure, which eschews column chromatography in favor of crystallization or filtration, further underscores the practical advantages of this technology for cost reduction in API manufacturing.
Mechanistic Insights into Acid-Catalyzed Acetalization and Substitution
The first stage of the synthesis involves the acid-catalyzed protection of the hydroxyl group in 2-methyl-2-butanol-4-iodine. Under the influence of a catalyst such as p-toluenesulfonic acid monohydrate, the hydroxyl group reacts with vinyl ethyl ether to form a mixed acetal linkage. This transformation is crucial as it stabilizes the molecule against unwanted side reactions in subsequent steps while introducing the necessary ethoxyethoxy functionality. The reaction is conducted in non-polar solvents like cyclohexane or dichloromethane at low temperatures (0-10°C) to ensure high selectivity and minimize polymerization of the vinyl ether. The precise control of moisture content in the solvent is vital, as water can compete with the alcohol for the vinyl ether, leading to hydrolysis and reduced yields. This step typically achieves conversion rates where the starting material is reduced to less than 0.5%, demonstrating the high efficiency of the catalytic system.
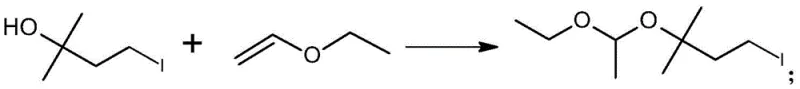
Following the acetalization, the intermediate 1-iodo-3-(1-ethoxyethoxy)-3-methylbutane undergoes a nucleophilic substitution reaction with sodium benzene sulfinate. This step is facilitated by a phase transfer catalyst, such as tetrabutylammonium bromide, which enhances the reactivity of the sulfinate anion in the organic phase. The reaction takes place in polar aprotic solvents like DMF or DMSO, often containing a controlled amount of water to solubilize the inorganic salt. The presence of an inorganic base, such as sodium bicarbonate or sodium carbonate, helps to neutralize any acidic byproducts and drive the equilibrium towards the formation of the sulfone. The mechanism proceeds via an SN2 pathway where the sulfinate ion displaces the iodide leaving group, forming the stable carbon-sulfur bond characteristic of the sulfone functional group. This robust transformation allows for the direct isolation of the product with high purity, often exceeding 85% without the need for further chromatographic purification.
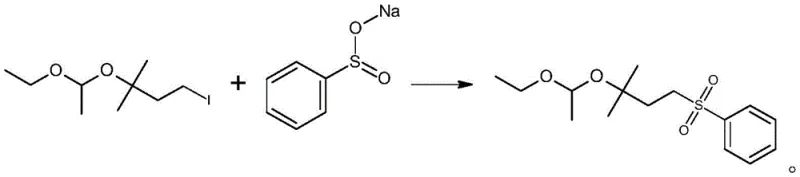
How to Synthesize C5 Acetal Sulfone Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity and scalability, making it an ideal candidate for industrial adoption. The process begins with the careful preparation of the reaction vessel under an inert atmosphere to exclude moisture and oxygen, which could degrade the sensitive reagents. The acetalization step requires precise temperature control and staged addition of vinyl ethyl ether to manage the reaction exotherm and maximize conversion. Once the acetal iodide is formed, it is directly subjected to the substitution conditions without extensive purification, streamlining the workflow. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined below to guide process engineers in replicating this efficient route.
- Perform hydroxyl acetalization of 2-methyl-2-butanol-4-iodine with vinyl ethyl ether using p-toluenesulfonic acid catalyst at 0-10°C.
- Conduct substitution reaction of the resulting acetal iodide with sodium benzene sulfinate using a phase transfer catalyst in aqueous DMF/DMSO at 60-80°C.
- Isolate the final product via solvent recovery and filtration without the need for column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this novel synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. The reliance on commodity chemicals such as vinyl ethyl ether and sodium benzene sulfinate ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or hazardous reagents. The elimination of complex purification steps like column chromatography translates directly into reduced processing times and lower solvent consumption, which are major cost drivers in fine chemical manufacturing. Furthermore, the mild reaction conditions reduce the wear and tear on reactor vessels and lower the energy burden on the facility, contributing to a more sustainable and cost-effective operation. These factors collectively enhance the overall economic viability of producing high-purity pharmaceutical intermediates at a commercial scale.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the substitution of expensive and hazardous organometallic reagents with inexpensive, bulk-available industrial chemicals. By removing the need for transition metal catalysts and the associated removal steps, the process significantly lowers the cost of raw materials and waste treatment. Additionally, the high yield and purity achieved without chromatographic purification result in substantial savings in solvent usage and labor hours. The ability to recover and recycle solvents like DMF and cyclohexane further amplifies these cost efficiencies, ensuring a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for global supply chains, and this method addresses it by utilizing raw materials that are produced on a massive industrial scale. Unlike specialized reagents that may face supply bottlenecks or long lead times, vinyl ethyl ether and sodium benzene sulfinate are readily accessible from multiple suppliers worldwide. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the simplified process flow reduces the dependency on highly skilled operators for complex manipulations, thereby minimizing human error and ensuring consistent batch-to-batch quality and delivery schedules.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a clear path to sustainable manufacturing. The absence of toxic heavy metals and the generation of minimal waste streams simplify compliance with increasingly stringent environmental regulations. The mild operating conditions reduce the risk of thermal runaway incidents, enhancing plant safety and lowering insurance premiums. As production scales from pilot batches to multi-ton quantities, the robustness of the chemistry ensures that yield and purity remain consistent, facilitating a smooth transition to full commercial production without the need for extensive re-optimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or for initiating new projects focused on Vitamin D3 derivatives.
Q: What are the primary advantages of this C5 acetal sulfone synthesis route over traditional Grignard methods?
A: Unlike traditional methods utilizing Grignard reagents or organic zinc which require harsh anhydrous conditions and cryogenic temperatures, this patented process operates under mild conditions (0-10°C and 60-80°C) using stable raw materials. It eliminates the safety hazards associated with pyrophoric reagents and avoids the generation of toxic waste, aligning with green chemistry principles while achieving yields exceeding 85% without complex purification.
Q: Is column chromatography required for purifying the final C5 acetal sulfone product?
A: No, column chromatography is not required. The process is designed such that the final product can be isolated through solvent recovery, dispersion in a nonpolar solvent like cyclohexane, and simple filtration. This significantly reduces processing time, solvent consumption, and operational costs, making it highly suitable for large-scale commercial production.
Q: What represents the key cost-saving factor in this manufacturing process?
A: The key cost-saving factors include the use of inexpensive, industrially available raw materials such as vinyl ethyl ether and sodium benzene sulfinate, combined with the elimination of expensive transition metal catalysts and complex purification steps. Furthermore, the ability to recover and reuse solvents like DMF and cyclohexane contributes substantially to reducing the overall cost of goods sold (COGS).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C5 Acetal Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value pharmaceutical intermediates like C5 acetal sulfone. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing the quality required for downstream synthesis of active pharmaceutical ingredients. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions outlined in this patent, allowing us to offer a seamless transition from development to commercial supply.
We invite you to collaborate with us to leverage this advanced synthetic technology for your Vitamin D3 projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
