Advanced Synthetic Route For 6-Methyl-Norpregnane-Diketone Ensuring Commercial Scalability
Advanced Synthetic Route For 6-Methyl-Norpregnane-Diketone Ensuring Commercial Scalability
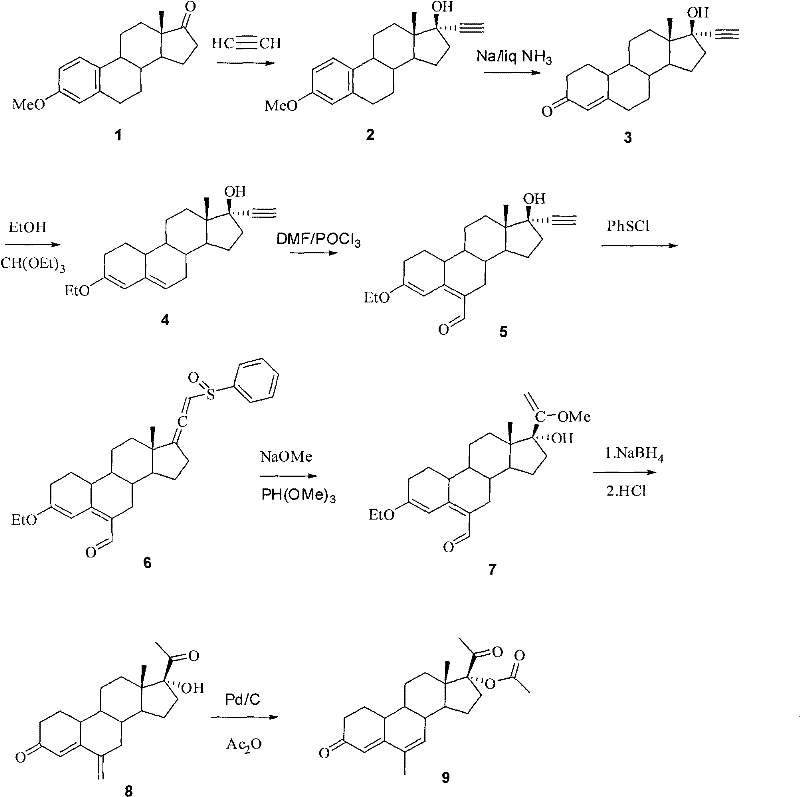
The pharmaceutical industry constantly seeks robust synthetic pathways for critical hormonal intermediates, and the methodology described in patent CN102134265A represents a significant leap forward in the production of 6-methyl-17alpha-acetoxyl-19-norpregnane-4,6-diene-3,20-diketone. This compound serves as a pivotal precursor for fourth-generation oral contraceptives and therapeutic agents for conditions such as uterine displacement and breast cancer resistance. The disclosed innovation fundamentally shifts the paradigm from relying on scarce, plant-extracted steroid precursors to utilizing abundant estrone derivatives, thereby securing the supply chain against agricultural volatility. By streamlining the synthesis into eight highly efficient steps, the patent addresses long-standing bottlenecks related to raw material availability and cumulative yield losses that have historically plagued the manufacturing of this high-value active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-methyl-17alpha-acetoxyl-19-norpregnane-4,6-diene-3,20-diketone has been hindered by a heavy dependence on 17-alpha-pregnane steroids derived from diosgenin, a saponin extracted primarily from wild yam or potato sources. This reliance creates a fragile supply chain susceptible to crop failures, seasonal variations, and geopolitical instability in sourcing regions. Furthermore, the traditional routes often necessitate upwards of fifteen distinct reaction steps to transform these bulky natural precursors into the desired 19-nor configuration, resulting in catastrophic cumulative yield losses and excessive solvent consumption. The conventional chemistry also frequently employs hazardous reagents like lithium methylide in ether for converting 17-carbonyls to 17-alpha-hydroxy-17-alpha-cyano groups, a step notorious for generating nearly fifty percent waste in the form of unwanted isomers, drastically inflating production costs and environmental disposal burdens.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages estrone-3-methyl ether, a commercially abundant and cost-effective starting material, to construct the steroid skeleton with superior efficiency. This route strategically employs a Birch reduction to simultaneously reduce the aromatic A-ring and establish the necessary 19-demethyl framework, effectively condensing multiple structural modifications into a single, high-yielding transformation. The subsequent introduction of the critical 6-methyl group is achieved through a sophisticated sequence involving Vilsmeier-Haack formylation followed by a unique elimination-rearrangement cascade, which avoids the harsh conditions and low selectivity of older methylation techniques. By reducing the total step count to just eight operations and eliminating the need for scarce plant extracts, this methodology offers a direct pathway to substantial cost reduction in pharmaceutical intermediates manufacturing while ensuring consistent batch-to-batch quality.
Mechanistic Insights into Vilsmeier-Haack Formylation and Pd-C Catalyzed Isomerization
The core chemical ingenuity of this process lies in the precise functionalization of the steroid backbone, particularly during the Vilsmeier-Haack formylation and the final catalytic isomerization steps. In the formylation stage, the enol ether intermediate reacts with the Vilsmeier reagent (generated in situ from DMF and POCl3) to selectively introduce a formyl group at the C6 position. This electrophilic substitution is critical because it sets the stage for the subsequent carbon-carbon bond formation that establishes the 6-methyl substituent, a key pharmacophore for the biological activity of the final drug. The reaction conditions are meticulously controlled between 0°C and 30°C to prevent poly-formylation or degradation of the sensitive enol ether moiety, ensuring that the regioselectivity remains high and the impurity profile is manageable for downstream processing.
Following the construction of the exocyclic methylene group via sodium borohydride reduction and acid dehydration, the final transformation involves a delicate isomerization catalyzed by Palladium on Carbon (Pd-C) in the presence of cyclohexene. This step is mechanistically fascinating as it facilitates the migration of the double bond from the exocyclic 6-methylene position into the ring system to form the conjugated 4,6-diene structure required for the target molecule. The use of cyclohexene acts as a hydrogen donor, preventing the complete saturation of the double bonds which would render the product inactive. This catalytic system operates under mild reflux conditions, demonstrating exceptional chemoselectivity by preserving the 17-alpha-acetoxy group and the 3,20-diketone functionality, which are prone to hydrolysis or reduction under less optimized conditions.
How to Synthesize 6-Methyl-Norpregnane-Diketone Efficiently
Implementing this synthetic route requires strict adherence to the reaction parameters outlined in the patent to maximize yield and purity, particularly regarding temperature control during the Birch reduction and the stoichiometry of the Pd-C catalyst. The process begins with the ethynylation of estrone-3-methyl ether using potassium tert-butoxide and acetylene gas, a step that must be conducted under anhydrous conditions to prevent the quenching of the strong base. Subsequent steps involve careful handling of liquid ammonia for the reduction phase and precise pH control during the acid-catalyzed dehydration to ensure the formation of the correct olefinic geometry. While the general workflow is straightforward, the success of the synthesis hinges on the rigorous exclusion of moisture and oxygen during the organometallic and reduction phases to maintain the integrity of the reactive intermediates.
- React acetylene with estrone-3-methyl ether to obtain 17alpha-ethynyl estrone-3-methyl ether.
- Perform Birch reduction at low temperature (-60 to -70°C) to obtain the 19-demethyl androstane derivative.
- Execute enol etherification followed by Vilsmeier reaction to introduce the formyl group at the 6-position.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this estrone-based synthesis route offers profound strategic benefits that extend far beyond simple unit price negotiations. By decoupling production from the volatile agricultural markets that dictate the price of diosgenin, manufacturers can achieve a level of cost stability and predictability that is essential for long-term contract planning. The simplification of the process from fifteen steps down to eight not only reduces the direct cost of goods sold by minimizing labor, energy, and solvent usage but also drastically shortens the manufacturing cycle time. This acceleration in production throughput allows for more responsive inventory management, enabling suppliers to react swiftly to sudden spikes in demand for contraceptive or oncology medications without the lead times associated with complex, multi-step semi-synthetic routes.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of expensive and scarce starting materials, replacing them with commodity chemicals like estrone-3-methyl ether which are produced at massive scales globally. Furthermore, the high yield reported in each step, often exceeding seventy percent, means that less raw material is wasted as byproduct, directly improving the mass balance and reducing the cost per kilogram of the final API intermediate. The removal of transition metal catalysts in earlier stages, relying instead on more benign reagents like sodium and liquid ammonia, also lowers the cost burden associated with heavy metal scavenging and waste treatment compliance.
- Enhanced Supply Chain Reliability: Sourcing estrone derivatives is significantly more reliable than sourcing specialized steroid precursors from limited botanical supplies, as the former is a standard item in the global fine chemical catalog. This abundance ensures that production schedules are not disrupted by raw material shortages, providing a secure continuity of supply for downstream pharmaceutical customers. Additionally, the robustness of the chemical steps, such as the crystallization-based purifications described in the examples, reduces the risk of batch failures due to chromatographic inconsistencies, further stabilizing the supply pipeline.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations like reflux, filtration, and crystallization that are easily transferred from pilot plants to multi-ton reactors without significant re-engineering. From an environmental perspective, the reduction in total solvent volume and the avoidance of toxic heavy metal waste streams align with modern green chemistry principles, simplifying the regulatory approval process for new drug master files. This environmental efficiency translates to lower operational overheads related to waste disposal and emissions control, making the facility more sustainable and compliant with increasingly stringent international environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology, drawing directly from the experimental data and claims within the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing specification limits for the new process. The answers provided reflect the specific advantages of the estrone-based route over legacy methods, focusing on yield optimization, impurity profiles, and operational safety.
Q: What are the primary advantages of this synthesis route over traditional methods?
A: The primary advantage is the use of readily available estrone-3-methyl ether as a starting material, bypassing the reliance on scarce potato-derived steroid precursors. Additionally, the process reduces the total number of reaction steps significantly, leading to higher overall yields and simplified operational complexity.
Q: How does the new method address impurity control during the isomerization step?
A: The method utilizes a specific Pd-C/cyclohexene system for the isomerization of the exocyclic methylene group. This catalytic system allows for precise control over the double bond migration to the 4,6-diene position while minimizing over-reduction or side reactions, ensuring high purity specifications for the final API intermediate.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. Key steps such as the Birch reduction and Vilsmeier formylation are well-established industrial reactions. The elimination of complex chromatographic purifications in favor of crystallization steps further enhances its viability for multi-kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methyl-Norpregnane-Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of hormonal intermediates for the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of 6-methyl-17alpha-acetoxyl-19-norpregnane-4,6-diene-3,20-diketone meets the highest international standards. Our infrastructure is designed to handle the specific safety requirements of reactions involving liquid ammonia and acetylene, guaranteeing a safe and compliant manufacturing environment.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic route can be integrated into your supply chain to drive value and efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements and regional logistics. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the competitiveness and resilience of your pharmaceutical product portfolio.
