Advanced Copper-Catalyzed Synthesis of Polysubstituted Thienocyclopentanone Derivatives for Commercial Scale-up
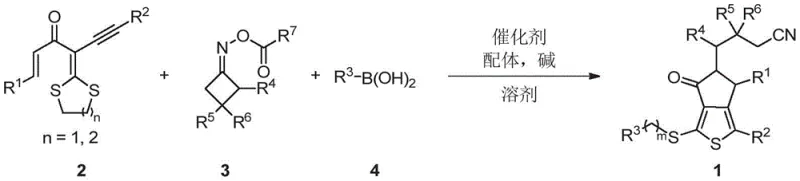
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to access complex heterocyclic scaffolds that serve as critical building blocks for drug discovery and organic electronic materials. Patent CN113549049B discloses a groundbreaking methodology for the synthesis of polysubstituted thienocyclopentanone derivatives, a structural motif prevalent in bioactive natural products and non-fullerene acceptors for solar cells. This innovation addresses the long-standing challenges in constructing fused thiophene-cyclopentanone systems by leveraging a visible-light-driven, copper-catalyzed intermolecular radical cascade. For R&D directors and procurement specialists, this technology represents a significant leap forward, offering a pathway to high-purity pharmaceutical intermediates with reduced environmental impact and simplified operational protocols compared to legacy synthetic strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted thienocyclopentanone derivatives has been fraught with synthetic inefficiencies and safety concerns. Traditional approaches typically rely on the multifunctionalization of pre-existing thiophene rings or the cyclization of sulfur-containing chain compounds, processes that often demand harsh reaction conditions such as elevated temperatures and strong acidic or basic environments. Furthermore, many established protocols necessitate the use of toxic organotin reagents or volatile thiols, which pose significant health hazards and create substantial waste disposal burdens for cost reduction in fine chemical manufacturing. These methods frequently suffer from poor functional group tolerance, limiting the structural diversity of the final products and requiring extensive protection-deprotection sequences that lower overall yields and increase production timelines.
The Novel Approach
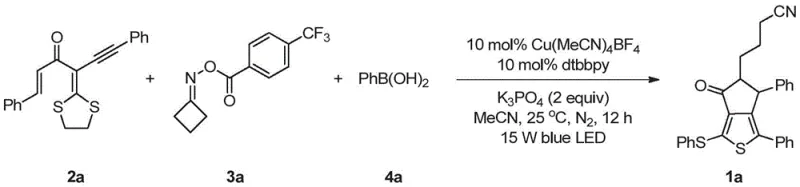
In stark contrast, the methodology outlined in CN113549049B introduces a streamlined, one-pot synthesis that elegantly bypasses these obstacles. By utilizing readily accessible alpha-enoyl-alpha-alkynyl dithioketals, cyclobutanone oxime esters, and boronic acids, the process achieves the simultaneous formation of two carbon-carbon bonds and two carbon-sulfur bonds through a radical cascade mechanism. This transformation is catalyzed by inexpensive copper salts under mild visible light irradiation, specifically using 15W blue LEDs at ambient temperature (25°C). This shift not only eliminates the need for toxic tin reagents but also dramatically enhances the operational simplicity, making it an ideal candidate for the commercial scale-up of complex heterocyclic scaffolds. The reaction exhibits remarkable efficiency, with representative examples achieving yields up to 76% under optimized conditions.
Mechanistic Insights into Copper-Catalyzed Radical Cascade Cyclization
The mechanistic elegance of this transformation lies in its ability to orchestrate multiple bond-forming events in a single operational step. The reaction initiates with the generation of a cyanoalkyl radical from the cyclobutanone oxime ester, facilitated by the copper catalyst and visible light excitation. This radical species undergoes a selective addition to the alkene moiety of the alpha-enoyl-alpha-alkynyl dithioketal substrate. Following this initial addition, an intramolecular radical cyclization occurs, closing the five-membered ring system. Crucially, the subsequent alkenyl radical intermediate promotes the homolytic cleavage of a C(sp3)-S bond within the dithioketal group, releasing a sulfur-centered radical that eventually recombines or participates in further transformations. The cycle concludes with a cross-coupling event involving the boronic acid, effectively installing the final substituent and regenerating the catalytic species. This intricate dance of radicals ensures high atom economy and precise control over the regioselectivity of the final product.
From an impurity control perspective, the use of specific ligands such as 4,4'-di-tert-butyl-2,2'-bipyridine (dtbbpy) plays a pivotal role in stabilizing the copper intermediates and suppressing off-cycle radical pathways that could lead to byproduct formation. The choice of base, optimally potassium phosphate (K3PO4), ensures the smooth transmetallation step with the boronic acid while maintaining a neutral to slightly basic environment that prevents the decomposition of sensitive functional groups. This level of mechanistic control translates directly to cleaner reaction profiles, reducing the burden on downstream purification processes and ensuring that the resulting high-purity organic functional materials meet the stringent specifications required for downstream applications in medicinal chemistry and material science.
How to Synthesize Polysubstituted Thienocyclopentanone Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, strict adherence to the optimized protocol is essential. The process begins with the preparation of the catalytic system under an inert atmosphere to prevent oxygen quenching of the radical intermediates. A typical procedure involves dissolving the copper catalyst and ligand in dry acetonitrile, followed by the sequential addition of the three key coupling partners and the inorganic base. The reaction vessel is then subjected to irradiation from a 15W blue LED source while maintaining a constant temperature of 25°C. Upon completion, typically within 12 hours, the crude mixture is purified via standard silica gel column chromatography. The detailed standardized synthesis steps for producing the model compound 1a are provided below.
- Prepare the catalytic system by mixing copper salt (e.g., Cu(MeCN)4BF4) and ligand (e.g., dtbbpy) in acetonitrile under inert atmosphere.
- Add the substrates including the dithioketal precursor, oxime ester, boronic acid, and base (e.g., K3PO4) to the reaction mixture.
- Irradiate the mixture with 15W blue LED light at 25°C for 12 hours, then isolate the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed protocol offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike legacy methods that rely on specialized or hazardous reagents like organotin compounds, this process utilizes commodity chemicals such as boronic acids and simple ketone derivatives, which are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages and price volatility, ensuring a more resilient production pipeline. Furthermore, the elimination of toxic heavy metals like tin simplifies waste treatment protocols, aligning with increasingly rigorous environmental regulations and reducing the overhead costs associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium, which are subject to significant market price fluctuations. Additionally, the reaction proceeds efficiently at room temperature, eliminating the energy costs associated with heating or cooling large-scale reactors. The one-pot nature of the synthesis reduces the number of unit operations, solvent exchanges, and isolation steps required, leading to substantial savings in labor, time, and solvent consumption. These factors collectively contribute to a significantly lower cost of goods sold (COGS) for the final thienocyclopentanone intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply chain reliability. Because the process tolerates a wide range of functional groups and operates under mild conditions, it is less prone to batch failures caused by minor variations in temperature or reagent quality. The use of stable starting materials that do not require cryogenic storage further simplifies logistics and inventory management. This stability allows for more predictable production schedules and shorter lead times, enabling manufacturers to respond more agilely to market demands for reliable pharmaceutical intermediate supplier capabilities without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the use of flow chemistry-compatible conditions, such as visible light irradiation and homogeneous catalysis. The absence of explosive or highly toxic reagents lowers the safety barrier for scale-up, allowing for larger batch sizes in standard glass-lined or stainless steel reactors. Moreover, the greener profile of the synthesis, characterized by lower E-factors and the avoidance of persistent organic pollutants, supports corporate sustainability goals. This environmental compliance is increasingly becoming a prerequisite for doing business with major multinational corporations in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial applications.
Q: What are the key advantages of this copper-catalyzed method over traditional thienocyclopentanone synthesis?
A: Unlike conventional methods that often require harsh conditions, toxic tin reagents, or multi-step functionalization of existing thiophene rings, this novel approach utilizes a mild, one-pot radical cascade. It operates at room temperature (25°C) under visible light, offers excellent functional group tolerance, and constructs two C-C and two C-S bonds simultaneously, significantly simplifying the synthetic route.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly amenable to scale-up. It employs inexpensive and commercially available copper catalysts rather than precious metals, uses common solvents like acetonitrile, and avoids hazardous reagents. The mild reaction conditions (25°C, ambient pressure) reduce energy consumption and safety risks associated with high-temperature or high-pressure reactors, facilitating reliable commercial scale-up of complex heterocyclic scaffolds.
Q: What is the substrate scope and functional group compatibility?
A: The method demonstrates broad substrate scope. The alpha-enoyl-alpha-alkynyl dithioketals can bear various aryl groups (phenyl, naphthyl, furyl, thienyl) and alkyl chains. The boronic acid component allows for the introduction of diverse aryl and alkenyl substituents. Furthermore, the resulting thienocyclopentanone core contains reactive handles like carbonyl and cyano groups, enabling further derivatization into high-purity organic functional materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Thienocyclopentanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed radical cascade methodology disclosed in CN113549049B for the production of advanced heterocyclic intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polysubstituted thienocyclopentanone derivative meets the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next drug discovery or material science project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you accelerate your development timeline while optimizing your manufacturing costs through our advanced synthetic capabilities.
