Optimizing Palladium Catalyst Production for High-Efficiency Pharmaceutical Intermediates
The landscape of homogeneous catalysis for pharmaceutical and fine chemical synthesis is constantly evolving, driven by the need for higher purity intermediates and more cost-effective manufacturing processes. A pivotal advancement in this domain is detailed in patent CN102977151A, which discloses a novel method for preparing bis(tricyclohexylphosphine) palladium dichloride, a critical catalyst widely used in cross-coupling reactions such as Suzuki-Miyaura couplings. This patent addresses long-standing inefficiencies in catalyst production by introducing a room-temperature protocol that utilizes palladium dichloride and dilute hydrochloric acid, bypassing the need for expensive and problematic sodium salts. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards cleaner reaction profiles and significantly reduced raw material costs. The technology not only simplifies the operational workflow but also ensures that the resulting catalyst possesses superior structural integrity, free from the metallic impurities that often plague traditional synthesis routes. By leveraging this intellectual property, manufacturers can achieve a competitive edge in the production of complex API intermediates and electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of bis(tricyclohexylphosphine) palladium dichloride has relied heavily on the reaction between tricyclohexylphosphine and sodium tetrachloropalladate in methanol solutions. While this conventional method offers a degree of stability, it suffers from inherent chemical drawbacks that limit its efficacy in high-performance applications. The primary issue lies in the presence of sodium ions within the reaction matrix; these cations tend to occupy coordination space around the palladium center, effectively competing with the bulky tricyclohexylphosphine ligands for binding sites. This steric hindrance impedes the formation of the desired 'active compound,' leading to lower conversion rates and compromised product quality. Furthermore, the reliance on sodium salts introduces additional metallic impurities that are difficult to remove completely, often necessitating rigorous and costly purification steps post-synthesis. The result is a catalyst with variable purity, typically hovering around 98.5%, which can introduce unpredictable variables in downstream pharmaceutical synthesis, affecting yield and reproducibility.
The Novel Approach
In stark contrast, the methodology outlined in patent CN102977151A revolutionizes this synthesis by eliminating sodium ions entirely from the reaction pathway. By directly utilizing palladium dichloride dissolved in a dilute hydrochloric acid solution, the process ensures that the palladium centers are fully available for coordination with the phosphine ligands without cationic interference. This approach allows for a much more efficient assembly of the catalyst structure, where the palladium ion can readily absorb the tricyclohexylphosphine molecules to form the stable, active complex. The reaction is conducted at room temperature under anaerobic conditions, which not only preserves the integrity of the air-sensitive phosphine ligands but also drastically reduces energy consumption compared to reflux-based methods. The simplicity of the workup—mere filtration and washing—further underscores the operational superiority of this technique, yielding a bright yellow solid powder with exceptional purity levels exceeding 99%.
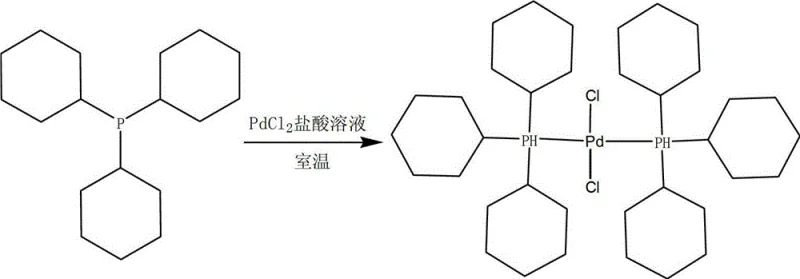
Mechanistic Insights into PdCl2-Mediated Coordination
The mechanistic advantage of this novel synthesis lies in the direct interaction between the palladium dichloride species and the tricyclohexylphosphine ligand in an acidic aqueous-organic interface. In the absence of competing alkali metal cations, the electrophilic palladium center is more accessible, facilitating a smoother ligand exchange process. The dilute hydrochloric acid serves a dual purpose: it solubilizes the palladium dichloride effectively while maintaining an acidic environment that prevents the premature hydrolysis or oxidation of the phosphine. As the tricyclohexylphosphine, dissolved in a water-miscible organic solvent like acetone or ethanol, encounters the acidic palladium solution, the coordination bonds form rapidly at ambient thermal energy. This kinetic favorability is crucial for industrial scalability, as it removes the need for prolonged heating or cryogenic cooling, thereby minimizing thermal degradation of sensitive functional groups. The resulting complex is thermodynamically stable, precipitating out of the solution as a high-purity solid that is ready for immediate use in catalytic cycles.
Furthermore, the quality of the final catalyst is intrinsically linked to the purity of the tricyclohexylphosphine precursor, which the patent also addresses through an optimized Grignard-based synthesis. By converting cyclohexyl halides into Grignard reagents and reacting them with phosphorus trihalides, followed by a unique salt formation and decomposition step using tetrafluoroboric acid and triethylamine, the process yields ligand precursors with purity levels above 99.3%. This rigorous control over the ligand quality ensures that the final palladium complex is free from phosphine oxides or other phosphorus-containing impurities that could poison the catalyst during downstream reactions. For R&D teams, this means that the impurity profile of the catalyst is tightly controlled, leading to more predictable reaction outcomes in the synthesis of complex drug molecules and agrochemical intermediates.
How to Synthesize Bis(Tricyclohexylphosphine) Palladium Dichloride Efficiently
The synthesis protocol described in the patent offers a robust framework for producing this valuable catalyst with minimal equipment requirements and maximum safety. The process begins with the careful dissolution of tricyclohexylphosphine in a suitable organic solvent under an inert atmosphere, ensuring that oxygen sensitivity is managed from the outset. Subsequently, the dropwise addition of the acidic palladium solution allows for precise control over the exotherm and precipitation rate, preventing the formation of amorphous aggregates. This streamlined approach not only enhances operator safety by avoiding high-pressure or high-temperature conditions but also facilitates easier validation and regulatory compliance for GMP manufacturing environments.
- Dissolve tricyclohexylphosphine in an organic solvent such as acetone or ethanol under strict anaerobic conditions to prevent oxidation.
- Dropwise add a diluted hydrochloric acid solution of palladium dichloride at room temperature while maintaining a molar ratio of 1: 2 to 1:3.
- Stir the reaction mixture for 2 to 6 hours at room temperature, then filter, wash the resulting yellow solid cake, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method presents compelling advantages for procurement managers and supply chain leaders looking to optimize their cost structures and reliability metrics. The elimination of sodium tetrachloropalladate in favor of the more abundant and less expensive palladium dichloride results in a substantial reduction in raw material expenditures. Additionally, the simplified processing steps—specifically the removal of energy-intensive reflux and complex purification stages—translate directly into lower utility costs and reduced labor hours per batch. These efficiencies compound to offer a significantly lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing without sacrificing margin. For supply chain heads, the robustness of the room-temperature reaction implies a lower risk of batch failure due to equipment malfunction or thermal runaway, ensuring a more consistent and reliable supply of this critical catalyst.
- Cost Reduction in Manufacturing: The strategic substitution of raw materials drives a drastic decrease in production costs, as palladium dichloride is generally more economical and readily available than its sodium salt counterparts. By removing the need for expensive purification technologies to strip away sodium residues, the overall manufacturing overhead is significantly lowered, creating a leaner production model that is highly resilient to market fluctuations in precious metal prices.
- Enhanced Supply Chain Reliability: The operational simplicity of this method, characterized by ambient temperature conditions and straightforward filtration, minimizes the dependency on specialized high-energy infrastructure. This accessibility means that production can be scaled up or adjusted rapidly in response to demand spikes, reducing lead times for high-purity catalysts and ensuring that downstream pharmaceutical manufacturing schedules are met without interruption.
- Scalability and Environmental Compliance: The process generates fewer waste streams associated with salt byproducts and solvent recovery, aligning well with modern green chemistry principles and environmental regulations. The ability to scale this reaction from gram to kilogram quantities without altering the fundamental parameters makes it an ideal candidate for commercial scale-up of complex catalytic intermediates, supporting long-term sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of bis(tricyclohexylphosphine) palladium dichloride, based on the specific innovations detailed in the patent literature. Understanding these nuances is essential for stakeholders evaluating the integration of this technology into their existing supply chains. These insights provide a foundational understanding of why this method is becoming the preferred standard for high-performance catalytic applications.
Q: Why does this method avoid sodium ions compared to traditional synthesis?
A: Traditional methods often use sodium tetrachloropalladate, where sodium ions can occupy coordination space and hinder the binding of bulky tricyclohexylphosphine ligands. This novel approach uses palladium dichloride directly, eliminating sodium interference and promoting the formation of the active catalytic species.
Q: What are the purity specifications achievable with this protocol?
A: By utilizing high-purity tricyclohexylphosphine prepared via a specialized Grignard-salt decomposition route and avoiding metallic impurities, this method consistently achieves product purities exceeding 99%, significantly higher than the 98.5% typical of prior art.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process operates at room temperature without the need for reflux or extreme cooling, utilizing simple filtration for isolation. This simplicity drastically reduces energy consumption and operational complexity, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(Tricyclohexylphosphine) Palladium Dichloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality catalysts play in the successful synthesis of advanced pharmaceutical intermediates and fine chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102977151A are fully realized in practical, large-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst meets the exacting standards required for GMP-compliant drug substance production. Our commitment to quality assurance means that clients can rely on us for consistent lot-to-lot reproducibility, minimizing the risk of downstream process deviations.
We invite global partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our optimized catalyst grades. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
