Advanced Synthesis of Arylpyrrole Intermediates for Scalable Agrochemical Production
Advanced Synthesis of Arylpyrrole Intermediates for Scalable Agrochemical Production
The development of efficient synthetic routes for high-performance agrochemical intermediates remains a critical priority for the global crop protection industry. Patent CN1035669C discloses a robust methodology for the preparation of 4-halo-2-aryl-1-(alkoxymethyl)-5-(trifluoromethyl)pyrrole compounds, which serve as pivotal precursors for potent insecticides and acaricides. This technology addresses significant challenges in process chemistry by optimizing reaction conditions to enhance both yield and product purity while mitigating safety risks associated with traditional alkylation agents. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this patented approach is essential for securing a stable supply chain. The disclosed process leverages stepwise halogenation and nucleophilic substitution to construct the complex pyrrole scaffold with high precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for generating functionalized pyrrole derivatives often rely on direct alkylation strategies that utilize hazardous reagents such as chloromethyl ethyl ether. These conventional methods present substantial operational risks due to the toxicity and volatility of the alkylating agents, necessitating expensive containment infrastructure and rigorous waste management protocols. Furthermore, direct alkylation can lead to poor regioselectivity and the formation of difficult-to-remove impurities, which compromises the quality of the final active ingredient. The use of aggressive conditions in older processes frequently results in lower overall yields and increased production costs, making them less attractive for large-scale commercial manufacturing. Supply chain heads are increasingly concerned with the environmental and safety liabilities associated with these legacy chemistries, driving the demand for safer alternatives.
The Novel Approach
The innovative process described in the patent overcomes these limitations by employing a sequential halogenation and etherification strategy that eliminates the need for dangerous chloromethyl ethers. By first installing a halomethyl group through controlled radical halogenation and subsequently displacing it with an alkoxide, the method achieves superior control over the reaction trajectory. This stepwise approach not only enhances the safety profile of the manufacturing process but also significantly improves the purity of the resulting intermediate by minimizing side reactions. The ability to conduct these transformations in common industrial solvents like tetrahydrofuran or chlorobenzene facilitates easier scale-up and reduces solvent recovery costs. This novel route represents a significant advancement in cost reduction in agrochemical manufacturing by streamlining the synthesis of complex heterocyclic structures.
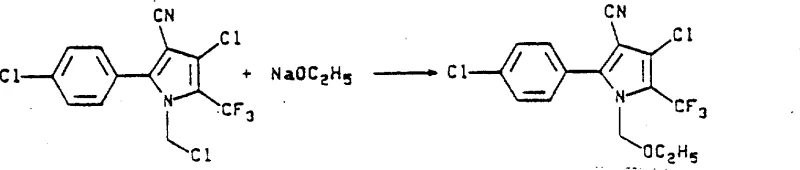
Mechanistic Insights into Halogenation and Cyclization
The core of this synthetic technology lies in the precise construction of the pyrrole ring and the subsequent functionalization of the nitrogen substituent. The cyclization step typically involves the reaction of an N-trifluoroacetyl glycine derivative with an activated alkene, such as acrylonitrile, in the presence of an acid anhydride and an organic base. This condensation reaction proceeds through a concerted mechanism that efficiently forms the five-membered heterocyclic ring while incorporating the crucial trifluoromethyl group at the 5-position. The electron-withdrawing nature of the trifluoromethyl group stabilizes the pyrrole nucleus and enhances the biological activity of the downstream insecticide. Understanding this mechanistic detail is vital for R&D teams aiming to replicate or optimize the process for high-purity agrochemical intermediate production.
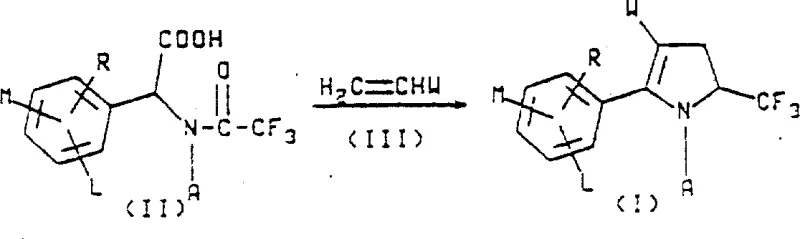
Following ring formation, the introduction of halogen atoms at the 4-position and the N-methyl group is achieved through a carefully controlled halogenation sequence. The process utilizes elemental halogens like chlorine or bromine, often with radical initiators such as benzoyl peroxide, to ensure selective substitution without degrading the sensitive pyrrole core. The incremental addition of the halogen reagent is a critical parameter that prevents over-halogenation and maintains the integrity of the nitrile functionality. Subsequent nucleophilic displacement of the halomethyl group by alkali metal alkoxides proceeds smoothly under mild basic conditions, yielding the final alkoxymethyl ether. This mechanistic robustness ensures consistent quality and supports the commercial scale-up of complex agrochemical intermediates.
How to Synthesize 4-Chloro-2-(p-Chlorophenyl)-1-(Ethoxymethyl)-5-(Trifluoromethyl)Pyrrole-3-Carbonitrile Efficiently
The synthesis of this specific high-value intermediate requires strict adherence to the patented reaction parameters to ensure optimal yield and safety. The process begins with the preparation of the N-trifluoroacetyl sarcosine precursor, followed by cyclization with acrylonitrile in acetonitrile solvent under reflux conditions. Once the pyrroline core is established, the material undergoes a multi-stage chlorination process involving thermal activation and radical initiation to install chlorine atoms at the ring and side-chain positions. The final transformation involves treating the chloromethyl intermediate with sodium ethoxide to generate the desired ethoxymethyl ether functionality. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-efficiency route.
- Prepare N-trifluoroacetyl glycine derivatives via trifluoroacetylation of arylglycine precursors using ethyl trifluoroacetate.
- Execute cyclization with activated alkenes like acrylonitrile in the presence of acid anhydrides and organic bases to form the pyrroline ring.
- Perform stepwise halogenation followed by nucleophilic substitution with alkali metal alkoxides to install the alkoxymethyl group.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain leaders in the agrochemical sector. By eliminating the requirement for highly regulated and hazardous alkylating agents, the process reduces the regulatory burden and insurance costs associated with manufacturing facilities. The use of readily available starting materials and common solvents ensures that the supply chain remains resilient against raw material shortages, thereby enhancing supply chain reliability for long-term production contracts. Additionally, the high yields reported in the experimental examples suggest a material-efficient process that minimizes waste generation and lowers the overall cost of goods sold. These factors combine to create a more sustainable and economically viable manufacturing model for key crop protection intermediates.
- Cost Reduction in Manufacturing: The avoidance of expensive and hazardous specialty reagents like chloromethyl ethyl ether leads to substantial cost savings in raw material procurement and waste disposal. The stepwise nature of the reaction allows for better process control, which reduces the incidence of batch failures and the need for costly reprocessing operations. Furthermore, the ability to use standard industrial solvents simplifies the recovery and recycling infrastructure, contributing to a lower environmental footprint and reduced operational expenditure. These efficiencies translate into a more competitive pricing structure for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as chlorine, bromine, and simple alkoxides ensures that the production process is not vulnerable to the supply disruptions often seen with exotic catalysts or reagents. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global agrochemical companies. The robust nature of the chemistry also allows for flexibility in sourcing, enabling manufacturers to qualify multiple vendors for key inputs to mitigate risk. Consequently, partners can expect a more dependable supply of high-purity intermediates throughout the product lifecycle.
- Scalability and Environmental Compliance: The reaction conditions, which operate at moderate temperatures and pressures, are well-suited for translation from laboratory scale to multi-ton commercial production without significant engineering hurdles. The process design inherently minimizes the generation of toxic byproducts, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. This compliance advantage reduces the risk of regulatory shutdowns and facilitates smoother audits by international customers. The combination of scalability and environmental safety makes this technology an ideal candidate for long-term strategic partnerships in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of the process for potential manufacturing partners. Understanding these details helps stakeholders assess the fit of this intermediate within their existing supply chains and product portfolios. The responses highlight the balance between technical performance and operational practicality that defines this patented approach.
Q: What are the key safety advantages of this synthesis route?
A: The process avoids the use of hazardous chloromethyl ethyl ether by utilizing a stepwise halogenation and etherification strategy, significantly improving operational safety.
Q: How is high purity achieved in the final intermediate?
A: High purity is maintained through controlled incremental addition of halogens and rigorous recrystallization steps using solvents like heptane or isopropanol.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common industrial solvents like acetonitrile and toluene, along with standard temperature ranges, supports feasible commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-2-(p-Chlorophenyl)-1-(Ethoxymethyl)-5-(Trifluoromethyl)Pyrrole-3-Carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like this arylpyrrole synthesis are executed with precision. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications to guarantee that every batch meets the exacting standards required for agrochemical active ingredient synthesis. We understand the critical nature of supply continuity for crop protection manufacturers and have optimized our operations to deliver consistent quality and reliability. Our technical team is ready to collaborate on process validation to ensure seamless integration into your manufacturing workflow.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. By engaging with us, you can access specific COA data and route feasibility assessments that demonstrate the viability of this intermediate for your projects. Let us partner with you to secure a sustainable and efficient supply of this high-value chemical building block for your agrochemical formulations.
