Advanced Benzimidazole Derivatives: Technical Insights for Commercial Agrochemical Production
The agricultural chemical industry continuously seeks novel active ingredients that offer superior efficacy and environmental profiles, and patent CN1147255A presents a significant advancement in this domain through the disclosure of substituted [1,4]-dioxino[2,3-f]benzimidazole derivatives. These novel compounds are specifically engineered to address the limitations of existing pest control agents, demonstrating potent fungicidal, insecticidal, and acaricidal activities that surpass structurally similar prior art. For R&D directors and procurement specialists, understanding the chemical architecture and synthesis pathway of these molecules is critical for evaluating their potential integration into existing crop protection portfolios. The patent details a robust synthetic methodology that allows for the precise introduction of sulfonyl groups onto the benzimidazole core, a modification that drastically enhances biological performance against resistant strains of pathogens such as Venturia inaequalis and Pyricularia oryzae. This technical breakthrough not only offers a new mode of action for resistance management but also provides a chemically efficient route that is amenable to large-scale industrial production, ensuring a stable supply of high-purity agrochemical intermediates for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art compounds, such as those disclosed in EP-A0517476, typically feature dimethylaminosulfonyl groups attached to the benzimidazole scaffold, which, while effective, often require higher application rates to achieve satisfactory control under field conditions. The efficacy of these legacy molecules can be inconsistent, particularly when dealing with resistant pest populations or when applied at low dosage rates, leading to increased operational costs and potential environmental burden due to higher chemical loadings. Furthermore, the synthesis of certain prior art analogs may involve complex multi-step sequences or less atom-economical transformations that generate significant waste streams, complicating the manufacturing process and inflating the cost of goods sold. From a supply chain perspective, reliance on older chemistries often means competing for established raw materials that may be subject to market volatility, and the purification of these older compounds can sometimes be challenging due to the formation of difficult-to-remove byproducts, impacting the final purity specifications required for regulatory approval.
The Novel Approach
The innovative strategy outlined in patent CN1147255A overcomes these hurdles by introducing a versatile sulfonylation protocol that allows for the attachment of diverse alkyl, alkenyl, or aryl sulfonyl groups to the benzimidazole nitrogen atom. This structural modification results in derivatives that exhibit unexpectedly high activity, with specific examples showing efficacy rates exceeding 80% at concentrations as low as 10 ppm, significantly outperforming comparative substances which showed efficacy as low as 0% to 43% in identical trials. The synthetic route is streamlined, utilizing a direct reaction between a benzimidazole precursor and a sulfonyl chloride in the presence of a base, which simplifies the process flow and reduces the number of unit operations required. This approach not only enhances the biological profile of the active ingredient but also improves the manufacturability of the compound, offering a clear pathway for cost reduction in agrochemical manufacturing by minimizing waste and maximizing yield through optimized reaction conditions.
Mechanistically, the formation of these high-performance derivatives relies on a nucleophilic substitution reaction where the nitrogen atom of the benzimidazole ring attacks the sulfur atom of the sulfonyl chloride. This reaction is facilitated by the presence of a suitable base, such as sodium hydride or potassium carbonate, which deprotonates the benzimidazole nitrogen, increasing its nucleophilicity and driving the reaction forward to completion. The patent specifies that this transformation can be carried out in a wide range of inert organic solvents, including tetrahydrofuran, acetonitrile, or dichloromethane, providing flexibility for process engineers to select solvents that align with safety and environmental regulations. The reaction temperature can be varied from 0°C to 150°C, allowing for fine-tuning of the kinetics to balance reaction rate with selectivity, ensuring that the desired N-sulfonylated product is formed with minimal formation of side products. This level of control is essential for maintaining a clean impurity profile, which is a key requirement for R&D directors focusing on regulatory compliance and product registration.
Impurity control is further enhanced by the specific workup procedures described, which involve aqueous quenching followed by extraction with organic solvents and subsequent crystallization. For instance, the patent details a process where the reaction mixture is poured into water, extracted with dichloromethane, and the organic layer is dried and concentrated to yield a residue that is then crystallized from diethyl ether. This purification strategy effectively removes unreacted starting materials, inorganic salts, and any minor byproducts, resulting in a final product with high chemical purity suitable for formulation. The ability to achieve high purity through standard crystallization techniques rather than complex chromatographic methods is a significant advantage for commercial scale-up, as it reduces processing time and solvent consumption. This robustness in purification ensures that the supply chain can deliver consistent quality batches, mitigating the risk of batch failures that could disrupt the availability of critical crop protection agents for farmers.
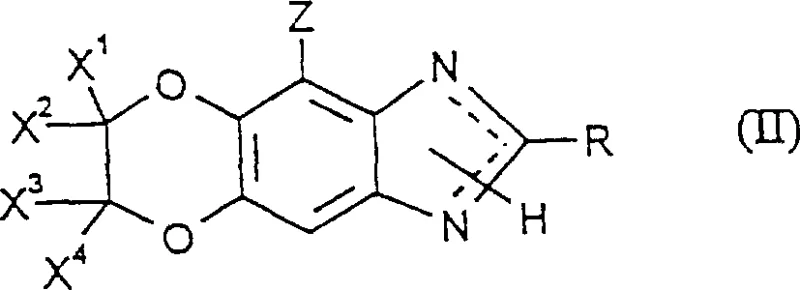
How to Synthesize 2-Cyano-3-methanesulfonyl-benzimidazole Efficiently
The synthesis of specific embodiments, such as 2-cyano-6,7-dichloro-6,7-difluoro-3-methanesulfonyl-[1,4]-dioxino-[2,3-f]-benzimidazole, exemplifies the practical application of this novel chemistry in a laboratory and pilot plant setting. The process begins with the preparation of the benzimidazole starting material, which is then reacted with methanesulfonyl chloride under controlled conditions to ensure high conversion. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-yield pathway.
- Prepare the benzimidazole starting material (Formula II) and select the appropriate sulfonyl chloride (Formula III).
- React the starting materials in an inert organic solvent with a suitable base auxiliary such as sodium hydride or potassium carbonate.
- Isolate the final product (Formula I) through extraction, drying, and crystallization to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and operational efficiency, primarily driven by the simplicity and robustness of the chemistry involved. The elimination of transition metal catalysts from the key sulfonylation step removes the need for expensive metal scavenging processes and reduces the risk of heavy metal contamination in the final product, which is a critical quality attribute for agrochemicals. This simplification of the process flow translates directly into lower manufacturing costs and a reduced environmental footprint, aligning with the increasing demand for sustainable production practices in the chemical industry. Furthermore, the use of commodity chemicals such as sulfonyl chlorides and common inorganic bases ensures that raw material supply is secure and less susceptible to the geopolitical fluctuations that often impact specialized reagents.
- Cost Reduction in Manufacturing: The synthetic pathway described avoids the use of precious metal catalysts and complex multi-step sequences, relying instead on readily available sulfonyl chlorides and simple base-mediated conditions. This streamlined approach significantly reduces the cost of goods by minimizing raw material expenses and lowering energy consumption through moderate reaction temperatures. The high yields reported in the patent examples, such as the 85% theoretical yield obtained in specific preparations, indicate an efficient use of resources that maximizes output per batch. Additionally, the straightforward workup procedure involving extraction and crystallization reduces solvent usage and waste disposal costs, contributing to substantial cost savings in the overall production lifecycle without compromising on the quality of the active ingredient.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials like methanesulfonyl chloride and common solvents such as tetrahydrofuran or acetonitrile ensures a resilient supply chain that is less vulnerable to disruptions. Unlike processes that depend on bespoke intermediates with long lead times, this method allows for rapid procurement of raw materials from multiple global suppliers, reducing the risk of production delays. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, provides flexibility for manufacturing sites to adapt to local resource availability without needing to revalidate the entire process. This adaptability ensures continuous production capability, allowing suppliers to meet fluctuating market demands for crop protection agents reliably and consistently throughout the growing season.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard unit operations like reflux, extraction, and crystallization that are easily transferred from laboratory to commercial scale. The absence of hazardous reagents or extreme pressure conditions simplifies the engineering requirements for production facilities, lowering capital expenditure for new capacity. From an environmental perspective, the atom economy of the sulfonylation reaction is favorable, and the ability to recover and recycle solvents further minimizes waste generation. This aligns with stringent environmental regulations and corporate sustainability goals, making the technology attractive for manufacturers seeking to reduce their ecological impact while maintaining high production volumes of effective pest control solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel benzimidazole derivatives, based on the data provided in the patent documentation. These insights are intended to clarify the operational benefits and chemical characteristics for stakeholders evaluating this technology for potential licensing or procurement.
Q: What distinguishes these benzimidazole derivatives from prior art compounds?
A: The novel derivatives feature specific sulfonyl substituents that demonstrate significantly higher fungicidal and acaricidal activity at lower application rates compared to prior art dimethylaminosulfonyl analogs.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, the process utilizes readily available sulfonyl chlorides and benzimidazole precursors, which facilitates scalable manufacturing and reduces supply chain risks.
Q: Does the synthesis require expensive transition metal catalysts?
A: No, the described sulfonylation process relies on inorganic or organic bases, eliminating the need for costly transition metal catalysts and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the sulfonylation of benzimidazoles are executed with precision and efficiency. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of agrochemical intermediate meets the highest international standards, providing our partners with the confidence needed to register and commercialize new crop protection products. We understand the critical nature of supply continuity in the agricultural sector and have optimized our operations to deliver high-purity benzimidazole derivatives consistently, supporting your R&D and production timelines effectively.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By collaborating with us, you can leverage our expertise to conduct a Customized Cost-Saving Analysis that identifies opportunities to optimize your supply chain and reduce manufacturing expenses. Let us help you secure a reliable source of high-performance agrochemical intermediates that drive innovation and profitability in your product portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
