Advanced Electrolytic Fluorination for High-Purity Fluoroethylene Sulfate Production and Commercial Scale-Up
Advanced Electrolytic Fluorination for High-Purity Fluoroethylene Sulfate Production and Commercial Scale-Up
The rapid evolution of lithium-ion battery technology demands electrolyte additives that can significantly enhance cycle life and safety, driving intense interest in fluoroethylene sulfate derivatives. Patent CN113215600A introduces a groundbreaking preparation method that utilizes direct electrolytic fluorination to synthesize these critical compounds from vinyl sulfate and anhydrous hydrogen fluoride. This innovative approach represents a paradigm shift from traditional multi-step chemical synthesis, offering a streamlined pathway that achieves high conversion rates and exceptional selectivity under controlled electrochemical conditions. By leveraging precise voltage and current parameters, manufacturers can produce a range of fluorinated species tailored for specific solid electrolyte interface (SEI) film formation requirements. This technical breakthrough not only addresses the growing demand for high-performance battery materials but also aligns with global sustainability goals by minimizing hazardous waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated vinyl sulfates has relied on cumbersome chemical fluorination routes that involve multiple reaction steps and hazardous reagents. Prior art, such as the methods disclosed in patent CN105541789A, typically requires dissolving vinyl sulfate in chlorinated solvents like dichloromethane and reacting it with N-bromosuccinimide (NBS) to form a bromo-intermediate, followed by a separate exchange reaction with sodium fluoride. Similarly, older processes like those in US3454597A utilize chlorine gas and carbon tetrachloride, introducing severe toxicity and environmental liability issues. These conventional pathways suffer from poor atom economy, complex workup procedures involving heavy metal catalysts or cyclodextrins, and significant challenges in waste disposal due to the generation of halogenated organic byproducts. Furthermore, the reliance on stoichiometric amounts of expensive fluorinating agents drives up production costs and complicates supply chain logistics for large-scale manufacturing.
The Novel Approach
In stark contrast, the novel electrolytic fluorination method described in the patent simplifies the entire production workflow into a single, continuous electrochemical process. By directly mixing vinyl sulfate with anhydrous hydrogen fluoride in an electrolytic cell, the process eliminates the need for intermediate isolation and the use of toxic halogenating reagents entirely. The reaction is driven by electricity rather than chemical oxidants, allowing for precise control over the fluorination degree simply by adjusting the molar ratio of reactants and electrochemical parameters. This one-step synthesis not only drastically reduces the equipment footprint and operational complexity but also inherently improves safety profiles by avoiding the handling of volatile and toxic intermediates. The result is a robust, scalable manufacturing route that delivers consistent product quality while significantly lowering the environmental impact associated with traditional fine chemical synthesis.
Mechanistic Insights into Electrolytic Fluorination of Vinyl Sulfate
The core mechanism of this process involves the electrochemical activation of hydrogen fluoride to generate reactive fluorine species that selectively substitute hydrogen atoms on the vinyl sulfate backbone. Under normal pressure and controlled temperatures ranging from -5°C to 50°C, the application of a specific voltage window (5-8V) and current density (30-120A) facilitates the direct introduction of fluorine atoms without disrupting the sensitive sulfate ester functionality. This selective fluorination is critical for maintaining the structural integrity required for the molecule to function effectively as an SEI film former in lithium batteries. The electrochemical environment allows for the tuning of reaction kinetics, ensuring that the fluorination proceeds with high efficiency while minimizing side reactions such as polymerization or over-fluorination that could degrade product performance. Understanding these electrochemical parameters is essential for optimizing yield and ensuring the reproducibility of the synthesis on an industrial scale.
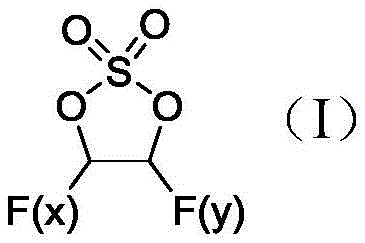
The versatility of this electrolytic method is evident in its ability to produce a spectrum of fluorinated derivatives, ranging from monofluoro- to trifluoro- substituted vinyl sulfates, depending on the reactant stoichiometry. When the molar ratio of anhydrous hydrogen fluoride to vinyl sulfate is maintained between 10:1 and 20:1, the reaction predominantly yields the monofluorinated species, whereas increasing this ratio to 40:1 or higher promotes the formation of di- and tri-fluorinated analogs. This tunability is a significant advantage for R&D teams seeking to optimize battery formulations, as different degrees of fluorination can impart distinct electrochemical properties to the electrolyte system. The ability to access these specific structural variants through a single platform technology simplifies the supply chain for battery manufacturers who require diverse additive portfolios.
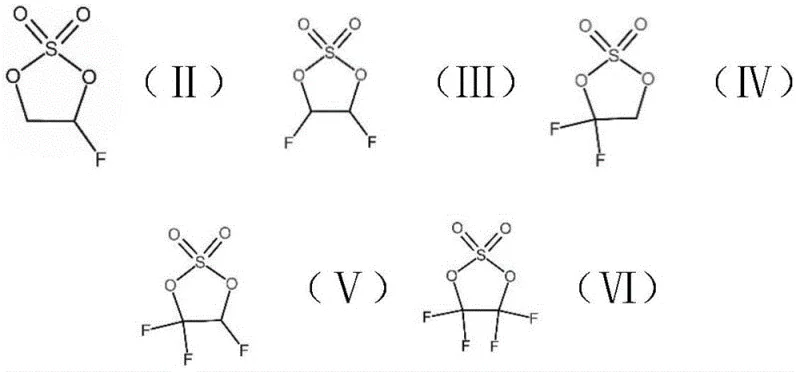
Impurity control is rigorously managed through a combination of electrochemical precision and downstream purification techniques. Following the electrolytic reaction, the crude mixture contains residual hydrogen fluoride, which is effectively neutralized using alkali metal oxides such as calcium oxide or aluminum oxide acting as acid scavengers. This neutralization step is crucial for preventing corrosion in downstream equipment and ensuring the stability of the final product. Subsequent rectification further refines the material, removing any unreacted starting materials or minor byproducts to achieve purity levels exceeding 99.5%. This high level of purity is paramount for battery applications, where trace impurities can lead to gas generation, impedance growth, and reduced cycle life, thereby validating the efficacy of the patented purification protocol.
How to Synthesize Fluoroethylene Sulfate Efficiently
The synthesis of fluoroethylene sulfate via this electrolytic route offers a practical and scalable solution for producing high-value battery additives. The process begins with the careful preparation of an anhydrous hydrogen fluoride solution of vinyl sulfate, requiring strict moisture control to prevent side reactions and equipment corrosion. Operators must then execute a two-stage electrolytic procedure: first, an electrolytic dehydration phase to ensure anhydrous conditions, followed by the main fluorination reaction under optimized voltage and current settings. The detailed standardized synthetic steps, including specific parameter ranges for temperature, time, and electrical input, are outlined in the structured guide below to ensure reproducible results.
- Mix anhydrous hydrogen fluoride and vinyl sulfate in an electrolytic cell to form a solution, ensuring moisture content is strictly controlled below 60000 ppm.
- Perform electrolytic dehydration at 4.5-7V and 5-50A for 5-15 hours, followed by electrolytic fluorination at 5-8V and 30-120A with temperatures between -5°C and 50°C.
- Remove residual hydrogen fluoride using an alkali metal oxide scavenger like calcium oxide, then rectify the crude product to obtain fluoroethylene sulfate with purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this electrolytic fluorination technology presents compelling opportunities for cost optimization and risk mitigation. By eliminating the dependency on expensive and hazardous chemical fluorinating agents like NBS or chlorine gas, the process inherently reduces raw material costs and removes the logistical burdens associated with handling dangerous goods. The simplification of the synthesis into a one-step reaction also translates to reduced utility consumption and lower labor requirements, as there are fewer unit operations and intermediate handling steps involved. These operational efficiencies contribute to a more resilient supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents or regulatory changes regarding hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of stoichiometric halogenating reagents and chlorinated solvents significantly lowers the variable cost of production, as electricity is generally a more stable and cost-effective reagent than specialized organic fluorine sources. Furthermore, the high current efficiency and conversion rates reported in the patent minimize raw material waste, ensuring that a greater proportion of the input vinyl sulfate is converted into valuable saleable product. The simplified downstream processing, which avoids complex extraction and washing steps typical of chemical fluorination, further reduces solvent recovery costs and energy consumption during purification. These factors combine to create a highly competitive cost structure that allows for substantial margin improvement or aggressive pricing strategies in the market.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like anhydrous hydrogen fluoride and vinyl sulfate, which are widely available from established industrial suppliers, greatly enhances supply security compared to sourcing niche fluorinating agents. The robustness of the electrochemical process means that production can be easily scaled up or down in response to market demand without the need for complex reactor reconfiguration or catalyst regeneration cycles. Additionally, the reduced generation of hazardous waste simplifies compliance with environmental regulations, mitigating the risk of production shutdowns due to permitting issues or waste disposal bottlenecks. This reliability is critical for maintaining continuous supply to battery manufacturers who operate on just-in-time inventory models.
- Scalability and Environmental Compliance: Electrolytic processes are inherently scalable, as capacity can often be increased by adding more cells or increasing electrode surface area without fundamentally changing the reaction chemistry. The absence of heavy metal catalysts and toxic halogenated byproducts aligns perfectly with modern green chemistry principles and increasingly stringent global environmental standards. This eco-friendly profile not only reduces the cost of waste treatment but also enhances the brand value of the final battery products by contributing to a more sustainable lifecycle. The ability to produce high-purity materials with a lower environmental footprint positions this technology as a future-proof solution for the rapidly growing energy storage sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrolytic production of fluoroethylene sulfate, based on the specific data and embodiments provided in the patent literature. These insights are designed to clarify the operational feasibility and strategic benefits of adopting this novel synthesis route for industrial applications. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing manufacturing frameworks or supply chains.
Q: What are the primary advantages of electrolytic fluorination over traditional chemical fluorination for vinyl sulfate?
A: Electrolytic fluorination eliminates the need for toxic halogenating agents like N-bromosuccinimide (NBS) or chlorine gas, significantly reducing environmental hazards and waste disposal costs while simplifying the process to a one-step reaction with higher selectivity.
Q: How does the process control the degree of fluorination in the final product?
A: The degree of fluorination is precisely controlled by adjusting the molar ratio of anhydrous hydrogen fluoride to vinyl sulfate; lower ratios favor monofluorinated products, while higher ratios facilitate the formation of difluoro- and trifluoro- derivatives.
Q: What purity levels can be achieved with this electrolytic method?
A: Following the removal of hydrogen fluoride and subsequent rectification, the process consistently yields fluoroethylene sulfate products with purity levels exceeding 99.5%, meeting stringent requirements for battery electrolyte applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroethylene Sulfate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN113215600A into commercial reality, offering unparalleled expertise in the scale-up of complex battery additives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of fluoroethylene sulfate meets the exacting standards required by top-tier lithium battery manufacturers. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure a stable supply of critical electrolyte components.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior electrolytic fluorination technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive your business forward in the competitive energy storage market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
