Advancing Pharmaceutical Intermediates Manufacturing with Eco-Friendly Biphasic Radical Bromination Technology
The pharmaceutical industry is constantly seeking sustainable methodologies to produce high-value intermediates without compromising on yield or purity. A pivotal advancement in this domain is detailed in Chinese Patent CN103265426A, which discloses an environmentally friendly preparation method for 2-(4-bromomethylphenyl)propionic acid, a critical precursor for the non-steroidal anti-inflammatory drug Loxoprofen Sodium. This technology represents a significant departure from conventional halogenation techniques by employing a novel water-organic two-phase free radical reaction system. By integrating elemental bromine with a biphasic solvent体系 and utilizing hydrogen peroxide as a clean oxidant, the process achieves a closed-loop recycling of bromine atoms. This innovation not only addresses the severe environmental hazards associated with hydrogen bromide emissions but also drastically improves the atom economy of the synthesis. For global procurement and R&D teams, understanding this shift towards greener chemistry is essential for maintaining compliance with increasingly stringent environmental regulations while securing a reliable supply chain for key pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzylic bromides like 2-(4-bromomethylphenyl)propionic acid has relied heavily on methods that are either economically inefficient or environmentally detrimental. Traditional approaches often utilize N-bromosuccinimide (NBS) in solvents like benzene or carbon tetrachloride. While NBS offers selectivity, it is prohibitively expensive for large-scale industrial application and generates stoichiometric amounts of succinimide waste, which complicates downstream purification and crystallization processes. Alternatively, direct bromination using elemental bromine in single-phase organic solvents suffers from poor atom economy; theoretically, only 50% of the bromine is incorporated into the product, while the remaining 50% is released as corrosive hydrogen bromide (HBr) gas. This necessitates complex and costly tail-gas absorption systems to prevent environmental contamination and equipment corrosion. Furthermore, the use of ozone-depleting solvents like carbon tetrachloride is now heavily restricted, forcing manufacturers to seek compliant alternatives that do not sacrifice reaction efficiency or safety standards.
The Novel Approach
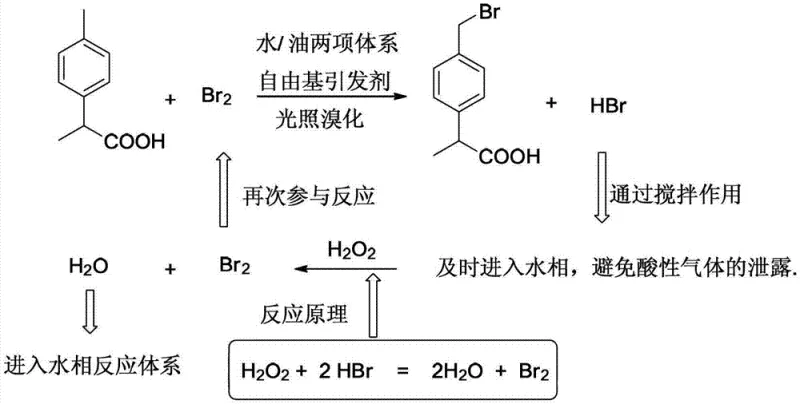
The methodology outlined in the patent introduces a transformative solution by replacing the single-phase organic system with a water-organic biphasic mixture. In this system, the organic phase dissolves the substrate and product, while the aqueous phase serves a dual function: it acts as a heat sink to control the exothermic radical reaction and, more critically, as a trap for the generated HBr gas. By mechanically stirring the two immiscible phases, the acidic HBr is immediately absorbed into the water layer, preventing its escape into the atmosphere. This setup allows for the subsequent addition of hydrogen peroxide, which oxidizes the dissolved hydrobromic acid back into elemental bromine. This regenerated bromine re-enters the organic phase to continue the bromination cycle. Consequently, the theoretical utilization rate of bromine is elevated from the traditional 50% to nearly 100%, significantly reducing raw material costs and eliminating the need for extensive waste neutralization procedures.
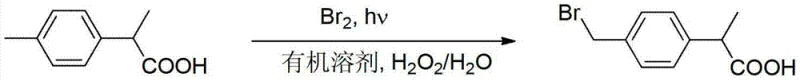
Mechanistic Insights into Two-Phase Free Radical Bromination
The core of this technological breakthrough lies in the intricate interplay between the radical initiation mechanism and the oxidative recycling loop within the biphasic interface. The reaction is initiated either by irradiation with energy-saving lamps (80-100W) or by thermal reflux in the presence of a radical initiator such as benzoyl peroxide (BPO) or AIBN. Upon initiation, bromine radicals abstract a hydrogen atom from the benzylic methyl group of the 2-(4-methylphenyl)propionic acid substrate. This generates a stable benzylic radical intermediate, which subsequently reacts with molecular bromine to form the desired bromomethyl product and a molecule of hydrogen bromide. In a conventional setup, this HBr would be lost; however, in this patented system, the vigorous agitation ensures rapid mass transfer of HBr from the organic interface into the aqueous phase.
Once in the aqueous phase, the hydrogen bromide undergoes an oxidation reaction driven by the introduction of hydrogen peroxide (H2O2). The chemical equation H2O2 + 2HBr → 2H2O + Br2 describes this regeneration process, where the nascent elemental bromine is highly reactive and readily partitions back into the organic phase to participate in further bromination cycles. This catalytic-like behavior of the bromine source means that the initial molar ratio of bromine to substrate can be reduced significantly, typically to a range of 1.0:0.56 to 1.0:0.62, compared to the excess required in traditional methods. The water phase also helps to moderate the reaction temperature, preventing overheating that could lead to poly-bromination or ring substitution impurities. This precise control over the radical environment ensures high selectivity for the mono-brominated product, simplifying the final purification steps and enhancing the overall quality of the pharmaceutical intermediate.
How to Synthesize 2-(4-Bromomethylphenyl)propionic Acid Efficiently
Implementing this green synthesis protocol requires careful attention to the phase ratios and the timing of reagent addition to maximize the recycling efficiency of the bromine species. The process begins by establishing the biphasic system with the substrate dissolved in a suitable organic solvent such as dichloromethane, dichloroethane, or chlorobenzene, alongside a specific volume of water. The reaction is maintained at a controlled temperature, typically between 10°C and 20°C for photochemical initiation, or at reflux for thermal initiation. A solution containing the radical initiator and elemental bromine is added dropwise, ensuring the reaction mixture maintains a pale yellow to micro-red color, indicative of active radical propagation without excessive free bromine accumulation.
- Prepare a reaction vessel with 2-(4-methylphenyl)propionic acid, organic solvent, and water, cooling to 10-20°C.
- Initiate radical reaction via light or heating while dropwise adding a bromine solution containing a radical initiator.
- Add hydrogen peroxide solution after half the bromine is added to oxidize generated HBr back to Br2, continuing until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biphasic radical bromination technology offers substantial strategic benefits beyond mere regulatory compliance. The primary advantage lies in the drastic reduction of raw material consumption and waste disposal costs. By recycling the hydrogen bromide by-product back into useful elemental bromine, the process effectively halves the requirement for fresh bromine feedstock compared to traditional single-pass methods. This reduction in stoichiometric demand translates directly into lower variable costs per kilogram of produced intermediate. Furthermore, the elimination of succinimide by-products (associated with NBS methods) removes a significant bottleneck in the purification workflow, reducing solvent usage for recrystallization and shortening the overall batch cycle time. These efficiencies contribute to a more robust and cost-competitive manufacturing profile for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the superior atom economy and the use of inexpensive, commodity-grade reagents. Elemental bromine is significantly cheaper than specialized brominating agents like NBS, and the ability to recycle the HBr by-product means that less fresh bromine needs to be purchased and transported. Additionally, the avoidance of ozone-depleting solvents like carbon tetrachloride mitigates the risk of regulatory fines and supply disruptions associated with restricted chemicals. The simplified work-up procedure, which avoids the removal of solid succinimide waste, further lowers labor and utility costs associated with filtration and drying operations, resulting in a leaner production cost structure.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that utilizes widely available and stable raw materials enhances supply chain resilience. Hydrogen peroxide and elemental bromine are bulk chemicals with established global supply networks, reducing the risk of shortages that can occur with specialty reagents. The robustness of the two-phase system also allows for greater flexibility in solvent selection; if dichloromethane supply is constrained, the process can be adapted to use dichloroethane or chlorobenzene without significant loss of yield. This adaptability ensures continuous production capability even when specific solvent markets experience volatility, providing a secure and reliable source of critical API intermediates for downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology is designed for seamless scale-up. The containment of corrosive HBr gas within the aqueous phase eliminates the need for massive external scrubbing towers, reducing the capital expenditure required for plant expansion. The generation of water as the only by-product of the oxidation step aligns perfectly with green chemistry principles, minimizing the facility's environmental footprint. This 'zero-pollution' characteristic facilitates easier permitting for new production lines and ensures long-term operational continuity in regions with strict environmental enforcement, making it an ideal choice for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this biphasic bromination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this method for your specific production needs. Understanding these nuances is critical for R&D teams planning technology transfer and for quality assurance teams establishing control strategies.
Q: How does the two-phase system improve atom economy compared to traditional methods?
A: Traditional single-phase bromination utilizes only 50% of the bromine atoms, releasing the rest as HBr waste. This patented two-phase method recycles HBr using hydrogen peroxide to regenerate elemental bromine in situ, theoretically increasing bromine utilization to 100%.
Q: What are the safety advantages of using a water-organic biphasic system?
A: The aqueous phase effectively absorbs corrosive hydrogen bromide (HBr) gas generated during the reaction, preventing its release into the environment. This eliminates the need for complex external scrubbing systems and significantly reduces operator exposure to acidic fumes.
Q: Why is this method preferred over NBS bromination for large-scale production?
A: N-bromosuccinimide (NBS) is significantly more expensive than elemental bromine and generates succinimide by-products that complicate purification. The elemental bromine method described here uses cheaper reagents and avoids solid by-product removal, streamlining downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Bromomethylphenyl)propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is vital for the future of the pharmaceutical industry. Our team of expert chemists has extensively evaluated the biphasic radical bromination technology described in CN103265426A and possesses the technical capability to implement this advanced process at a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative method—such as high atom economy and reduced environmental impact—are fully realized in our manufacturing operations. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-(4-bromomethylphenyl)propionic acid meets the exacting standards required for the synthesis of Loxoprofen Sodium.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. By leveraging our expertise in green chemistry and process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to verify the superior quality and economic advantages of our manufacturing capabilities. Together, we can build a sustainable and efficient supply chain for high-purity pharmaceutical intermediates that drives value for your organization.
