Advanced Cinchona Alkaloid Ligands for High-Efficiency Asymmetric Synthesis and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust solutions for asymmetric synthesis, a critical step in producing chiral active pharmaceutical ingredients (APIs). Patent CN113831366A introduces a groundbreaking class of cinchona alkaloid ligands that address longstanding limitations in stereoselectivity and reaction scope. These novel P,N-ligands, characterized by a quinine or quinidine backbone modified with bulky, electron-rich phosphorus substituents, represent a significant evolution in organocatalysis and transition metal catalysis. By expanding the structural diversity at the phosphorus center to include 3,5-disubstituted phenyl groups and naphthyl moieties, the invention provides a versatile toolkit for chemists aiming to construct carbon-nitrogen bonds with high precision. This technological advancement is particularly relevant for the manufacturing of complex pharmaceutical intermediates where chirality dictates biological activity.
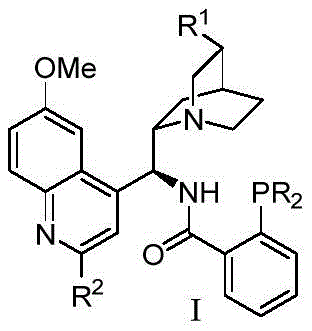
Historically, the development of chiral ligands derived from cinchona alkaloids has been constrained by the availability of simple phosphorus substituents. Conventional methods typically relied on commercially available aryl or alkyl groups attached to the phosphorus atom, which often resulted in limited steric bulk and insufficient electronic differentiation. These structural limitations frequently led to moderate enantioselectivity and narrow substrate scope in asymmetric transformations, particularly in challenging radical reactions. Furthermore, traditional ligands often lacked the necessary rigidity to effectively shield one face of the metal center, resulting in lower ee values that necessitated expensive and wasteful recrystallization or chromatographic purification steps. The inability to finely tune the steric environment around the catalytic center hindered the optimization of reactions involving bulky or electronically deactivated substrates.
The novel approach detailed in the patent overcomes these deficiencies through strategic structural modification of the ligand framework. By introducing sterically demanding groups such as 3,5-di-tert-butylphenyl, 3,5-diphenylphenyl, and various naphthyl derivatives at the phosphorus position, the new ligands create a highly defined chiral pocket. This enhanced steric environment forces the substrate to approach the metal center in a specific orientation, thereby drastically improving stereocontrol. Additionally, the inclusion of an amide linkage capable of hydrogen bonding or deprotonation adds a secondary coordination site, stabilizing the transition state through non-covalent interactions. This dual-activation mode allows the ligand to coordinate effectively with transition metals like copper, facilitating difficult asymmetric cross-coupling and difunctionalization reactions with unprecedented efficiency and selectivity.
Mechanistic Insights into Copper-Catalyzed Asymmetric Radical Reactions
The efficacy of these cinchona-based ligands is rooted in their ability to form stable, chiral complexes with transition metals, specifically copper, which acts as the central hub for radical generation and capture. In the catalytic cycle, the ligand coordinates to the copper center via the phosphorus atom and the quinoline nitrogen, creating a rigid chiral environment. When a radical precursor, such as an alkyl halide, interacts with this complex, single-electron transfer occurs to generate a carbon-centered radical. The bulky substituents on the phosphorus atom prevent the radical from rotating freely, locking it into a specific trajectory relative to the electrophile. This spatial constraint is critical for distinguishing between the pro-chiral faces of the reacting species, ensuring that the new bond forms with high enantiomeric excess. The amide moiety further assists by potentially stabilizing charged intermediates or participating in proton transfer events that lower the activation energy of the rate-determining step.
From an impurity control perspective, the high stereoselectivity of this ligand system inherently minimizes the formation of unwanted enantiomers, which are often the most difficult impurities to remove in chiral synthesis. Traditional catalysts might produce a near-racemic mixture, requiring extensive downstream processing to isolate the desired isomer. In contrast, the optimized steric bulk of the new ligands suppresses the formation of the minor enantiomer at the source. Moreover, the robust nature of the P,C-bond formation in the ligand synthesis ensures that phosphine oxidation, a common degradation pathway for phosphine ligands, is mitigated by the electron-donating nature of the substituents. This stability translates to a cleaner reaction profile with fewer phosphine-oxide byproducts, simplifying the workup and purification processes for the final pharmaceutical intermediate.
How to Synthesize Cinchona Alkaloid Ligands Efficiently
The preparation of these high-performance ligands follows a logical, modular synthetic strategy that is amenable to scale-up. The process begins with the formation of the phosphine oxide backbone via Grignard addition, followed by a crucial deoxygenation step to reveal the reactive phosphine. This phosphine is then coupled with a functionalized benzoic acid derivative, which serves as the linker to the cinchona scaffold. The final step involves amide bond formation with a quinine-derived amine, stitching together the chiral pool material with the synthetic phosphine component. This convergent approach allows for the independent optimization of the phosphine and amine fragments before final assembly. For a detailed breakdown of the standardized synthetic steps, including specific molar ratios and purification techniques, please refer to the guide below.
- React Grignard reagent with diethyl phosphite to form intermediate S2, followed by copper-catalyzed deoxygenation to generate the phosphine S3.
- Perform nucleophilic aromatic substitution of intermediate S3 with methyl o-fluorobenzoate to yield ester S4, then hydrolyze to carboxylic acid S5.
- Couple the carboxylic acid S5 with a quinine-derived amine S6 using EDCI and DMAP to finalize the cinchona alkaloid ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ligand technology offers tangible benefits beyond mere chemical performance. The synthetic route described in the patent utilizes readily available starting materials such as Grignard reagents, diethyl phosphite, and commercial quinine derivatives, ensuring a stable and reliable supply chain. Unlike ligands requiring rare earth metals or complex multi-step chiral resolutions, this methodology relies on commodity chemicals and standard unit operations like reflux and column chromatography. This accessibility reduces the risk of supply disruptions and allows for flexible sourcing strategies. Furthermore, the high yields reported in the examples suggest that material throughput can be maximized, reducing the overall cost of goods sold (COGS) for the final catalytic process.
- Cost Reduction in Manufacturing: The implementation of these ligands can lead to significant cost savings by eliminating the need for expensive chiral separation technologies. Because the ligands induce high enantioselectivity directly during the reaction, the burden on downstream purification is drastically reduced. In traditional processes, separating enantiomers often consumes a large portion of the production budget through preparative HPLC or multiple recrystallizations. By achieving high ee values upfront, manufacturers can streamline their workflows, reduce solvent consumption, and shorten production cycles. Additionally, the robustness of the ligand synthesis itself means less waste is generated during the production of the catalyst, contributing to a more economical and sustainable manufacturing footprint.
- Enhanced Supply Chain Reliability: The modular nature of the ligand synthesis enhances supply chain resilience. Since the phosphine component and the cinchona component are synthesized separately and coupled at the end, bottlenecks in one area do not necessarily halt the entire production line. This decoupling allows for inventory buffering of key intermediates. Moreover, the reagents used, such as copper triflate and TMDS, are standard industrial chemicals with established global supply networks. This reduces dependency on single-source suppliers for exotic reagents. The ability to source raw materials from multiple vendors ensures continuity of supply, which is critical for meeting the strict delivery schedules demanded by pharmaceutical clients.
- Scalability and Environmental Compliance: The synthetic protocol is designed with scalability in mind, utilizing solvents like tetrahydrofuran, toluene, and dichloromethane, which are well-understood in large-scale chemical engineering. The reaction conditions, mostly ranging from room temperature to mild reflux, do not require extreme pressures or cryogenic temperatures that would demand specialized equipment. This ease of scale-up facilitates the transition from kilogram-level R&D batches to multi-ton commercial production without significant process re-engineering. From an environmental standpoint, the high atom economy of the coupling reactions and the reduction in purification steps minimize the generation of hazardous waste, aligning with increasingly stringent global environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these cinchona alkaloid ligands in industrial settings. Understanding the operational parameters and compatibility of these catalysts is essential for successful technology transfer. The answers provided are derived directly from the experimental data and structural analysis presented in the patent documentation, ensuring accuracy and relevance for process development teams evaluating this technology for their specific applications.
Q: What structural features distinguish these new ligands from traditional cinchona derivatives?
A: The novel ligands feature expanded phosphorus substituents, specifically 3,5-disubstituted phenyl groups, which provide enhanced steric bulk and electronic tuning compared to simple aryl or alkyl groups found in prior art.
Q: Can this ligand system be scaled for industrial production of API intermediates?
A: Yes, the synthetic route utilizes standard reagents like Grignard reagents and common coupling agents (EDCI), avoiding exotic catalysts, which facilitates straightforward scale-up from laboratory to commercial tonnage.
Q: What represents the primary advantage in terms of reaction efficiency?
A: The ligands demonstrate significantly improved enantioselectivity (ee values often exceeding 90%) and yield in copper-catalyzed radical asymmetric reactions, reducing the need for costly chiral separation downstream.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinchona Alkaloid Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced ligands play in modern drug discovery and process chemistry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess required for your API synthesis. We understand that consistency is key in pharmaceutical manufacturing, and our quality management systems are designed to deliver batch-to-batch reproducibility that meets the highest international standards.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating these novel ligands into your existing processes. Whether you require custom synthesis of specific ligand analogues or large-scale supply of the standard variants, we are prepared to support your needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline and achieve your commercial objectives efficiently.
