Scaling High-Purity Asymmetric Quaternary Ammonium Salts for Next-Generation Energy Storage
Scaling High-Purity Asymmetric Quaternary Ammonium Salts for Next-Generation Energy Storage
The rapid evolution of energy storage technologies has placed unprecedented demand on the purity and stability of electrolyte materials, particularly for double-layer capacitors and high-energy power storage systems. Patent CN101307053B introduces a groundbreaking methodology for the preparation of asymmetric N,N'-1,4-dialkyl triethylene diammonium tetrafluoroborate, a critical component in advanced electrochemical cells. This technical disclosure moves beyond theoretical chemistry to offer a robust, scalable manufacturing protocol that addresses the longstanding challenges of toxicity and prolonged reaction times associated with legacy synthesis routes. By shifting from hazardous ether-based solvents to environmentally benign aqueous and alcoholic systems, this innovation not only enhances operator safety but also streamlines the downstream purification processes required to meet stringent electronic-grade specifications. For industry leaders seeking a reliable electronic chemical supplier, understanding the nuances of this patented pathway is essential for securing a competitive advantage in the burgeoning energy storage market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric quaternary ammonium salts relied heavily on nucleophilic substitution reactions conducted in volatile organic solvents such as diethyl ether or benzene. These traditional protocols, as documented in earlier literature like Bull Chem Soc Jpn (1986), often necessitated excessively long reaction durations, sometimes extending up to 48 hours, to achieve acceptable conversion rates. The reliance on ether introduces significant safety hazards due to its high flammability and tendency to form explosive peroxides, creating substantial liability and insurance costs for manufacturing facilities. Furthermore, the separation of products from these solvent systems often requires complex distillation setups and generates hazardous waste streams that are costly to treat and dispose of in compliance with modern environmental regulations. The cumulative effect of these inefficiencies is a production bottleneck that limits the ability to scale output to meet the growing global demand for high-performance capacitor electrolytes without incurring prohibitive operational expenditures.
The Novel Approach
In stark contrast, the methodology outlined in CN101307053B leverages a stepwise alkylation strategy that utilizes water and lower alcohols as primary reaction media, fundamentally altering the economic and safety profile of the manufacturing process. The initial mono-substitution is carefully controlled at low temperatures around 10°C in ethyl acetate or ethanol, ensuring high selectivity for the monosubstituted intermediate before proceeding to the second alkylation step. The subsequent reaction with a second halohydrocarbon is performed in deionized water under reflux, a condition that drastically accelerates the reaction kinetics compared to anhydrous organic conditions while eliminating the fire risks associated with ether. This transition to aqueous chemistry simplifies the workup procedure, as the inorganic salts and excess reagents can be more easily managed, and the final product isolation is achieved through efficient vacuum distillation and crystallization. This novel approach represents a paradigm shift towards greener chemistry that aligns perfectly with the sustainability goals of modern chemical enterprises.
Mechanistic Insights into Stepwise Alkylation and Ion Exchange
The core chemical transformation involves a precise two-stage nucleophilic attack on the triethylenediamine backbone, followed by a critical anion exchange to install the tetrafluoroborate counterion. Initially, the lone pair on one of the bridgehead nitrogen atoms attacks the first halohydrocarbon (R1X), forming a quaternary ammonium halide salt. The presence of the positively charged nitrogen in this mono-substituted intermediate exerts an electron-withdrawing inductive effect, which slightly deactivates the second nitrogen atom; however, the use of water as a polar protic solvent in the second stage effectively stabilizes the transition state and facilitates the attack by the second halohydrocarbon (R2X). This careful modulation of reaction conditions ensures the formation of the desired asymmetric dialkyl species rather than a statistical mixture of symmetric byproducts. The final conversion to the tetrafluoroborate salt is achieved by treating the di-quaternary halide with tetrafluoroboric acid, driving the equilibrium towards the less soluble or more stable tetrafluoroborate form through ion exchange.
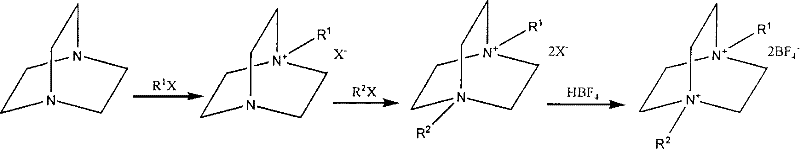
Beyond the primary synthetic steps, the patent details a sophisticated purification mechanism that is vital for achieving the ultra-low impurity profiles required for electronic applications. The crude tetrafluoroborate product is subjected to a decolorization process using activated carbon in an alcoholic solvent, which acts as a powerful adsorbent for trace organic impurities, colored bodies, and potentially catalytic metal residues. This step is crucial because even parts-per-million levels of transition metals like iron or copper can catalyze decomposition reactions within the electrolyte, leading to gas generation and capacitor failure. The subsequent hot filtration and slow crystallization allow for the exclusion of remaining halide ions and solvent inclusions from the crystal lattice. The result is a white crystalline product with purity assays reaching 99.9% and metal ion content suppressed to below 3.0 ppm, demonstrating a level of control over the impurity spectrum that is essential for high-voltage electrochemical stability.
How to Synthesize Asymmetric N,N'-1,4-dialkyl triethylene diammonium tetrafluoroborate Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing temperature control and stoichiometric precision to maximize yield. The process begins with the dropwise addition of the first alkylating agent to a cooled solution of triethylenediamine, followed by a standing period to ensure complete crystallization of the mono-salt. This intermediate is then redissolved in water for the second alkylation, where reflux conditions drive the reaction to completion over a period of 4 to 24 hours depending on the specific alkyl chain length. Following the removal of water and excess reagents via vacuum distillation, the resulting di-quaternary halide undergoes ion exchange with aqueous tetrafluoroboric acid. The detailed standardized synthesis steps see below guide.
- Perform mono-substitution by dripping halohydrocarbon into triethylenediamine and solvent at 10°C to form the monosubstituted salt.
- React the monosubstituted salt with a second halohydrocarbon in deionized water under reflux to generate the disubstituted halide salt.
- Conduct ion exchange using tetrafluoroboric acid solution followed by purification with activated carbon to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend far beyond simple chemical yield improvements. The substitution of expensive and hazardous solvents like ether with commodity chemicals such as water and ethanol significantly reduces the raw material cost base and minimizes the need for specialized explosion-proof infrastructure. This shift lowers the barrier to entry for scaling production, allowing manufacturers to utilize standard stainless steel reactors rather than exotic alloys required for handling corrosive or highly reactive organic media. Furthermore, the simplified waste stream, primarily consisting of aqueous saline solutions and recoverable alcohols, reduces the burden on wastewater treatment facilities and lowers the overall environmental compliance costs associated with hazardous waste disposal. These factors combine to create a more resilient and cost-effective supply chain capable of withstanding market volatility.
- Cost Reduction in Manufacturing: The elimination of toxic ether and benzene from the process flow removes the necessity for complex solvent recovery systems designed to handle high-volatility, flammable liquids, thereby reducing both capital expenditure on equipment and operational expenditure on energy. By utilizing water as the primary medium for the second alkylation step, the process leverages the high heat capacity of water to manage exothermic reactions more safely and efficiently, reducing cooling costs. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate symmetric byproducts, which increases the overall mass efficiency of the process and reduces the loss of valuable starting materials. This streamlined approach ensures that the cost reduction in electronic chemical manufacturing is driven by fundamental process intensification rather than temporary market fluctuations.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically triethylenediamine and various haloalkanes, are commodity chemicals produced on a massive global scale, ensuring a stable and continuous supply without the risk of single-source bottlenecks. The robustness of the aqueous reaction conditions means that the process is less sensitive to minor variations in moisture content or ambient temperature compared to strictly anhydrous organometallic syntheses, leading to more consistent batch-to-batch quality and fewer production delays. This reliability is critical for maintaining the just-in-time delivery schedules required by major battery and capacitor manufacturers who cannot afford interruptions in their assembly lines. Consequently, partnering with a supplier utilizing this technology mitigates the risk of supply disruptions caused by raw material scarcity or complex logistical hurdles.
- Scalability and Environmental Compliance: The transition to a water-based system inherently simplifies the scale-up process, as heat and mass transfer issues that often plague large-scale organic reactions are mitigated by the favorable physical properties of the aqueous medium. The process generates significantly less hazardous waste, aligning with increasingly strict global environmental regulations regarding volatile organic compound (VOC) emissions and toxic effluent discharge. This environmental compatibility not only future-proofs the manufacturing facility against regulatory tightening but also enhances the brand reputation of the end-product as a 'green' electronic material. The ability to scale from kilogram quantities in the lab to multi-ton annual production without fundamental changes to the chemistry demonstrates a high degree of commercial maturity and readiness for immediate industrial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized electrolyte salts. The answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing supply chains and product formulations. The focus remains on the practical implications of the synthesis method for quality, safety, and cost.
Q: What distinguishes this synthesis method from conventional ether-based processes?
A: Unlike traditional methods requiring toxic ether or benzene and reaction times up to 48 hours, this patented process utilizes safer alcoholic solvents and water, significantly reducing reaction time and environmental hazards while maintaining high yields.
Q: How does the process ensure electronic-grade purity for capacitor applications?
A: The method incorporates a rigorous purification step using activated carbon decolorization and recrystallization, effectively reducing metal ion impurities like Na, K, and Fe to levels below 3.0 ppm, which is critical for preventing self-discharge in supercapacitors.
Q: Is this manufacturing route suitable for large-scale industrial production?
A: Yes, the use of water as a reaction medium for the second alkylation step and common haloalkanes as raw materials simplifies the supply chain and waste treatment, making the process highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric N,N'-1,4-dialkyl triethylene diammonium tetrafluoroborate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN101307053B can be reliably replicated on an industrial scale. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify that every batch meets the sub-ppm metal ion limits required for high-performance supercapacitors. Our commitment to quality assurance means that clients receive a product that not only performs optimally in electrochemical tests but also ensures the long-term reliability of their energy storage devices.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this greener, more efficient manufacturing process. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your electronic chemical supply chain.
