Advanced Edaravone Manufacturing: Technical Breakthroughs in Cost and Purity for Global Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for neuroprotective agents, particularly for critical compounds like Edaravone, which serves as a potent free-radical scavenger in the treatment of acute ischemic stroke. Patent CN101830852A introduces a transformative synthetic methodology that addresses long-standing inefficiencies in the production of this vital API intermediate. By shifting the starting material from expensive free phenylhydrazine to the more economical phenylhydrazine hydrochloride, this innovation not only drastically reduces raw material expenditures but also enhances the chemical integrity of the final product. The technical significance of this patent lies in its ability to harmonize cost reduction with stringent quality control, a balance that is often difficult to achieve in fine chemical manufacturing. This report analyzes the technical nuances of this route, providing R&D and procurement leaders with a clear understanding of its commercial viability and supply chain implications.
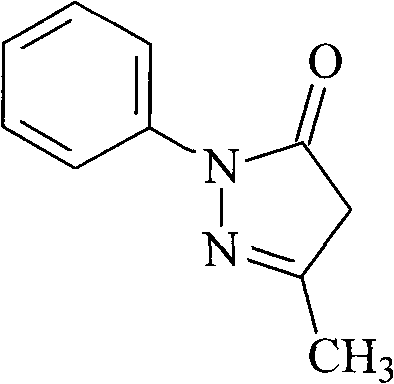
The limitations of conventional Edaravone synthesis methods have long posed challenges for large-scale manufacturers aiming to optimize production costs without compromising pharmaceutical grade standards. Traditional approaches typically involve the direct condensation of free phenylhydrazine with ethyl acetoacetate in solvents such as ethanol or water. While chemically feasible, these legacy processes suffer from inherent economic and technical drawbacks, primarily driven by the high market price and stability issues associated with free phenylhydrazine. Historical data indicates that conventional routes often struggle to achieve yields exceeding 70%, with product purity frequently stagnating around 96% even after multiple recrystallization steps. Furthermore, the reliance on pure ethanol for purification in older methods presents both environmental and cost inefficiencies, as solvent recovery rates are often suboptimal. These factors collectively contribute to a higher cost of goods sold (COGS) and a less reliable supply chain for downstream drug manufacturers who require consistent, high-volume availability of the intermediate.
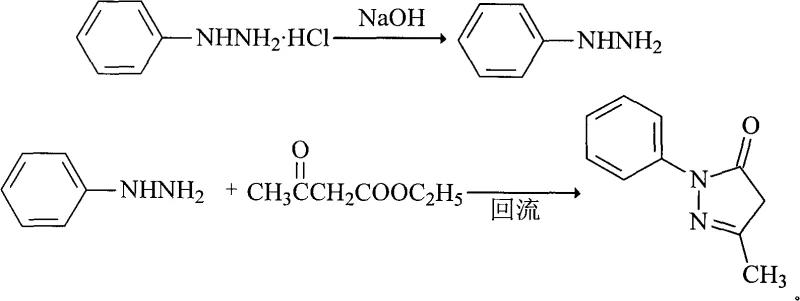
In contrast, the novel approach detailed in the patent data leverages a strategic substitution of raw materials and a refined purification protocol to overcome these historical bottlenecks. By utilizing phenylhydrazine hydrochloride as the primary starting material, the process capitalizes on a significantly lower price point compared to the free base, effectively lowering the entry cost for the synthesis. The method employs an in-situ generation step where sodium hydroxide is used to liberate the reactive phenylhydrazine within the reaction mixture, thereby minimizing handling risks and exposure to air which can degrade the reagent. This modification not only streamlines the operational workflow but also creates a reaction environment that favors higher conversion rates. The subsequent purification stage utilizes a specific isopropanol-water solvent system combined with activated carbon adsorption, a technique that has been proven to remove trace impurities more effectively than traditional ethanol washing. This holistic redesign of the synthesis pathway results in a marked improvement in both yield and purity, establishing a new benchmark for manufacturing efficiency.
Mechanistic Insights into Phenylhydrazine In-Situ Generation and Cyclization
The core chemical innovation of this process revolves around the controlled in-situ generation of phenylhydrazine and its subsequent condensation with ethyl acetoacetate to form the pyrazolone ring. The reaction begins with the neutralization of phenylhydrazine hydrochloride by sodium hydroxide in an aqueous medium, which releases the free amine necessary for nucleophilic attack. This step is critical as it ensures that the reactive species is generated immediately prior to its consumption, reducing the likelihood of side reactions or oxidation that typically plague stored free phenylhydrazine. Following this, the addition of ethyl acetoacetate initiates a condensation reaction that proceeds through a reflux phase, typically maintained for 2 to 4 hours. The thermal energy provided during reflux drives the cyclization forward, facilitating the elimination of ethanol and water to close the five-membered heterocyclic ring. The stoichiometry is carefully balanced, with a molar ratio favoring a slight excess of the hydrazine component to ensure complete consumption of the keto-ester, thereby minimizing residual starting materials in the crude product.
Impurity control is meticulously managed through the unique recrystallization protocol involving an isopropanol-water mixture. Unlike conventional ethanol-based purification, which may co-crystallize certain organic byproducts or fail to remove colored impurities effectively, the isopropanol-water system offers a different polarity profile that selectively precipitates the target Edaravone molecule. The addition of activated carbon during the reflux of the recrystallization solvent plays a pivotal role in adsorbing high-molecular-weight colored impurities and trace metal residues that could otherwise compromise the safety profile of the pharmaceutical intermediate. This adsorption step is followed by hot filtration, which removes the carbon along with the trapped impurities, leaving a clear solution that yields white crystalline powder upon cooling. The result is a product with purity levels consistently reaching 99.9%, a specification that exceeds the requirements for most regulatory filings and ensures a cleaner impurity profile for the final drug substance.
How to Synthesize Edaravone Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize the benefits of the novel methodology. The process is designed to be scalable, moving seamlessly from laboratory verification to industrial production with minimal adjustment to the core chemical principles. Operators must ensure that the neutralization step is complete before the addition of the keto-ester to prevent acid-catalyzed side reactions. The reflux duration is a critical variable, with 2.5 hours identified as the optimal timeframe to balance conversion efficiency with energy consumption. Detailed standardized synthesis steps see the guide below for specific operational parameters.
- React phenylhydrazine hydrochloride with sodium hydroxide in water to generate free phenylhydrazine in situ.
- Add ethyl acetoacetate to the reaction mixture and reflux for 2 to 4 hours to form crude Edaravone.
- Purify the crude product using an isopropanol-water solution with activated carbon adsorption to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic advantages that extend beyond simple unit cost savings. The shift to phenylhydrazine hydrochloride as a feedstock mitigates the volatility associated with the pricing of free phenylhydrazine, providing a more stable cost basis for long-term contracting. This stability is crucial for maintaining margin integrity in the face of fluctuating raw material markets. Furthermore, the improved yield profile means that less raw material is required to produce the same amount of finished product, effectively amplifying the purchasing power of the procurement budget. The simplified purification process also reduces the demand for high-purity ethanol, substituting it with more readily available isopropanol and water, which further insulates the supply chain from solvent shortages or price spikes.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally stronger due to the substantial price differential between phenylhydrazine hydrochloride and free phenylhydrazine. By eliminating the need to purchase the more expensive free base, manufacturers can achieve significant direct material savings. Additionally, the higher yield reduces the effective cost per kilogram of the final API intermediate, as less waste is generated and less reprocessing is required. The elimination of complex catalytic systems or exotic reagents further simplifies the bill of materials, contributing to a leaner and more cost-effective production structure that enhances overall competitiveness in the global market.
- Enhanced Supply Chain Reliability: Phenylhydrazine hydrochloride is a stable salt that is easier to store and transport than the free base, which is prone to oxidation and degradation. This stability translates into reduced inventory losses and a more reliable supply of starting materials, ensuring that production schedules are not disrupted by raw material quality issues. The robustness of the synthesis route also means that production can be scaled up rapidly to meet surges in demand without the need for extensive process re-validation. This reliability is a key value proposition for pharmaceutical clients who require guaranteed continuity of supply for their critical drug formulations.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing common unit operations such as reflux, filtration, and crystallization that are standard in multi-purpose chemical plants. The use of water as a primary solvent in the initial steps reduces the organic solvent load, aligning with green chemistry principles and simplifying waste treatment protocols. The activated carbon treatment effectively manages organic impurities, reducing the burden on downstream wastewater treatment facilities. These factors collectively make the process easier to permit and operate within strict environmental regulatory frameworks, reducing the risk of compliance-related shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Edaravone synthesis route. These answers are derived directly from the patent specifications and practical manufacturing considerations, providing clarity for stakeholders evaluating this technology for adoption. Understanding these details is essential for making informed decisions about process integration and supply chain optimization.
Q: Why is phenylhydrazine hydrochloride preferred over free phenylhydrazine in this synthesis?
A: Phenylhydrazine hydrochloride is significantly more cost-effective than free phenylhydrazine, reducing raw material costs while allowing for in-situ generation of the reactive species, which minimizes impurity formation and improves overall process safety.
Q: How does the new purification method impact product quality?
A: By utilizing an isopropanol-water recrystallization system combined with activated carbon adsorption, the process achieves purity levels exceeding 99.9%, significantly higher than the 96% typical of conventional ethanol recrystallization methods.
Q: What are the scalability advantages of this synthetic route?
A: The process utilizes common, stable raw materials and avoids complex catalytic systems, making it highly suitable for commercial scale-up from 100 kgs to multi-ton production with consistent yield and minimal waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edaravone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate innovative patent methodologies like CN101830852A into commercial reality. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis route are fully realized in large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Edaravone meets the highest international standards. Our commitment to quality assurance means that clients can rely on us for a consistent supply of high-purity intermediates that facilitate smoother regulatory approvals for their final drug products.
We invite global pharmaceutical partners to engage with our technical procurement team to explore how this optimized synthesis route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to our manufacturing process can impact your bottom line. We encourage you to contact us for specific COA data and route feasibility assessments to verify the compatibility of our Edaravone with your existing formulations. Let us collaborate to drive efficiency and quality in your neuroprotective drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
