Scalable Synthesis of Neratinib Related Substance Formula I for Global Pharmaceutical Compliance
Scalable Synthesis of Neratinib Related Substance Formula I for Global Pharmaceutical Compliance
The development of robust synthetic routes for drug-related substances is a cornerstone of modern pharmaceutical quality control and regulatory compliance. As epidermal growth factor receptor (EGFR) inhibitors like Neratinib become critical in treating HER2-positive breast cancer, the demand for high-purity reference standards and impurities for method validation has surged. Patent CN111848582A introduces a groundbreaking methodology for preparing the Neratinib related substance known as Formula I. This technical breakthrough addresses the longstanding challenges of low yields and complex purification associated with previous methods. By leveraging a streamlined four-step sequence starting from readily available quinoline derivatives, this process delivers a target product with exceptional purity profiles suitable for rigorous analytical applications.
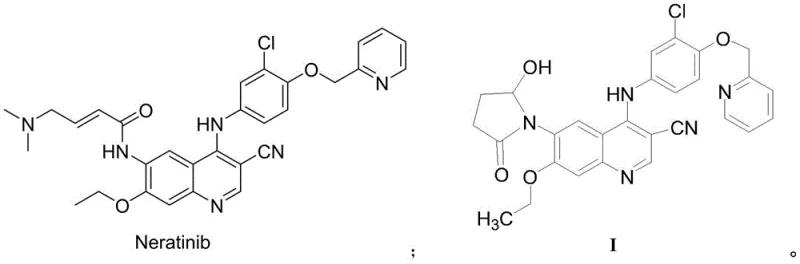
The strategic importance of this synthesis lies in its ability to produce complex heterocyclic impurities without resorting to preparative HPLC or flash column chromatography, which are often bottlenecks in supply chains. For R&D directors and procurement managers alike, the availability of such high-quality intermediates ensures that drug substance characterization meets the stringent requirements of global pharmacopoeias. The patent outlines a pathway that not only optimizes chemical efficiency but also aligns with green chemistry principles by enabling solvent recovery and minimizing waste generation. This report delves deep into the mechanistic nuances and commercial implications of adopting this novel synthetic strategy for your pharmaceutical intermediate sourcing needs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Neratinib related substances have historically suffered from significant operational inefficiencies that hinder commercial viability. As documented in earlier literature, such as the 2015 publication referenced in the patent background, traditional routes often rely on multi-step sequences that result in suboptimal overall yields. A critical drawback of these conventional approaches is the absolute necessity for column chromatography to isolate the final product. In an industrial setting, column chromatography is notoriously difficult to scale, consumes vast quantities of silica and solvents, and introduces variability in product recovery. Furthermore, the reliance on harsh conditions or unstable intermediates in older pathways frequently leads to the formation of difficult-to-remove byproducts, compromising the purity required for analytical reference standards.
These limitations translate directly into increased costs and extended lead times for pharmaceutical manufacturers. The inability to crystallize the product directly means that production batches are smaller and less consistent, creating supply chain vulnerabilities for companies dependent on these specific impurities for stability testing and regulatory filings. Additionally, the environmental footprint of disposing of large volumes of chromatographic waste contradicts modern sustainability goals. Consequently, there has been a pressing industry need for a synthetic route that bypasses these purification hurdles while maintaining high structural fidelity and yield.
The Novel Approach
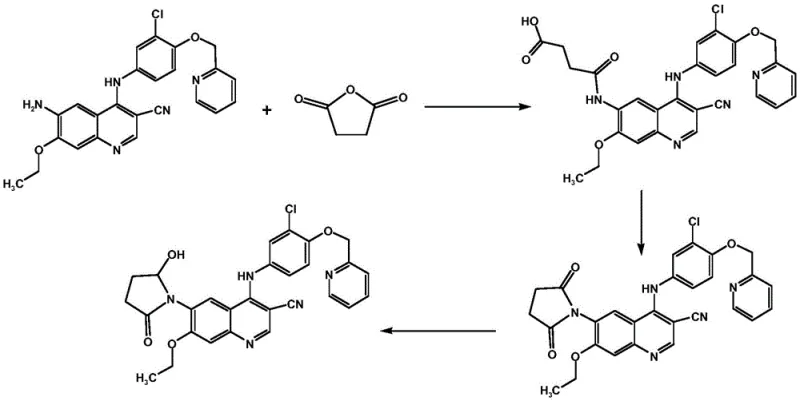
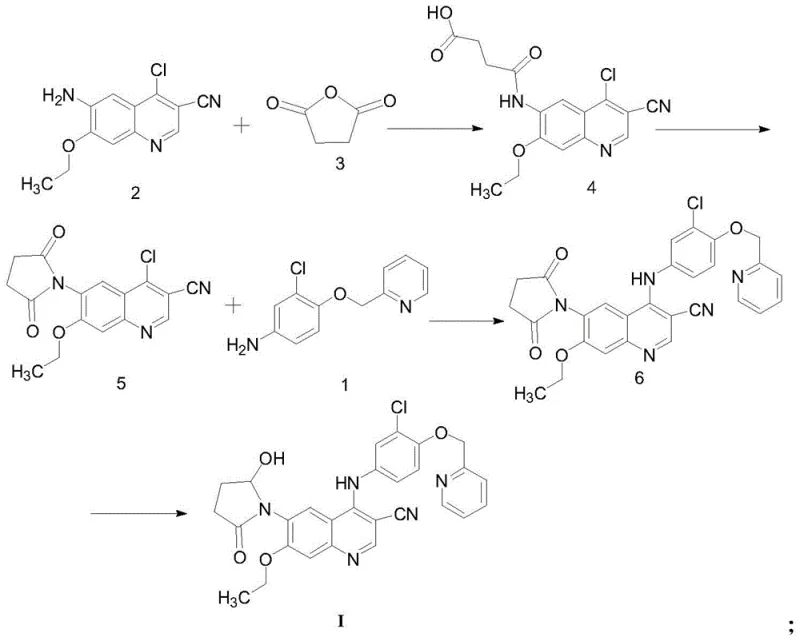
The methodology disclosed in patent CN111848582A represents a paradigm shift in the synthesis of this specific EGFR inhibitor impurity. By utilizing 3-cyano-4-chloro-6-amino-7-ethoxyquinoline as a robust starting material, the inventors have designed a convergent route that maximizes atom economy and operational simplicity. The core innovation lies in the construction of the succinimide ring system followed by a highly selective coupling reaction. Unlike previous methods, this novel approach allows the final product to be isolated via simple recrystallization techniques, completely eliminating the need for column treatment. This capability is a game-changer for scaling, as it transforms a laboratory curiosity into a commercially viable manufacturing process.
Furthermore, the reaction conditions are remarkably mild, utilizing common organic solvents like toluene and acetonitrile which can be easily recovered and reused in subsequent batches. The process achieves a final purity exceeding 99.0% and an overall yield surpassing 35%, figures that are highly competitive for complex heterocyclic synthesis. For supply chain heads, this means a more predictable and reliable source of high-purity pharmaceutical intermediates. The reduction in processing steps and the avoidance of specialized purification equipment significantly lower the barrier to entry for large-scale production, ensuring that the supply of these critical reference materials can meet the growing demands of the global oncology drug market.
Mechanistic Insights into the Four-Step Synthetic Sequence
The chemical elegance of this process is best understood through a detailed dissection of its four distinct transformation stages. The sequence begins with an acylation reaction where the amino group of the quinoline starting material attacks succinic anhydride. This step is conducted in toluene under reflux conditions, typically between 80°C and 120°C, driving the formation of the open-chain amic acid intermediate which subsequently dehydrates or proceeds directly to the next stage. The use of a Dean-Stark trap or similar water removal apparatus is implied by the reflux conditions in toluene, ensuring the equilibrium shifts towards the desired amide formation. This initial functionalization sets the stage for the crucial ring-closing event that defines the core scaffold of the impurity.
Following the initial acylation, the second step involves an intramolecular cyclization to form the succinimide ring. This is achieved by heating the intermediate in acetic anhydride with sodium acetate acting as a mild base catalyst. The conditions promote dehydration and ring closure, generating the bicyclic succinimide structure fused to the quinoline system. The third step is a nucleophilic aromatic substitution where the chlorine atom on the quinoline ring is displaced by an aniline derivative. Uniquely, this patent employs zinc nitrate as a Lewis acid catalyst in acetonitrile to facilitate this coupling. This catalytic activation enhances the electrophilicity of the quinoline ring, allowing the reaction to proceed efficiently at reflux temperatures without degrading the sensitive succinimide moiety.
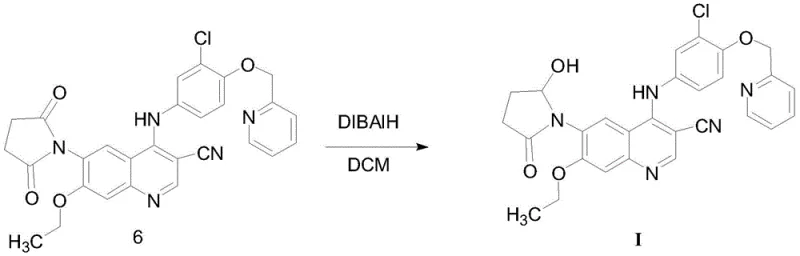
The final and perhaps most chemically sophisticated step is the selective reduction of the succinimide carbonyl to a hydroxy-lactam. This is accomplished using diisobutylaluminum hydride (DIBAL-H) in dichloromethane at cryogenic temperatures ranging from -40°C to -25°C. The control of temperature is paramount here; DIBAL-H is a powerful reducing agent that could potentially reduce the nitrile group or other sensitive functionalities if not carefully managed. By maintaining the reaction at low temperatures, the process achieves chemoselectivity, reducing only one of the carbonyl groups of the imide to the corresponding hemiaminal (which exists in equilibrium with the hydroxy-lactam form). This precise control ensures the structural integrity of the final Formula I compound, matching the specific impurity profile required for Neratinib analysis.
How to Synthesize Neratinib Related Substance Formula I Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the reduction phase and stoichiometry during the coupling step. The patent provides a clear roadmap for transitioning from bench-scale experiments to pilot production, emphasizing the reproducibility of each stage. The standardized protocol ensures that the critical quality attributes of the intermediate are maintained throughout the batch cycle. For technical teams looking to adopt this route, the following guide outlines the operational framework derived directly from the patent examples, serving as a foundation for process validation and scale-up activities.
- React 3-cyano-4-chloro-6-amino-7-ethoxyquinoline with succinic anhydride in toluene under reflux to form the amide intermediate.
- Perform ring-closing cyclization using sodium acetate in acetic anhydride to generate the succinimide core structure.
- Couple the succinimide intermediate with the aniline derivative using zinc nitrate catalyst in acetonitrile, followed by selective reduction with DIBAL-H.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain strategists focused on cost optimization and risk mitigation. The most significant value driver is the elimination of column chromatography, which is traditionally one of the most expensive and time-consuming unit operations in fine chemical manufacturing. By relying on crystallization for purification, the process drastically reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in raw materials and waste disposal. This efficiency gain allows for a more competitive pricing structure for the final API intermediate, making it an attractive option for budget-conscious procurement departments.
- Cost Reduction in Manufacturing: The economic benefits extend beyond simple material savings. The ability to recycle solvents like toluene and acetonitrile further compresses the variable costs associated with production. Since the process does not require specialized equipment such as large-scale chromatography columns or high-pressure reactors, capital expenditure for setting up production lines is significantly minimized. The high yield of each individual step contributes to a robust overall mass balance, reducing the amount of starting material required per kilogram of finished product. These factors combine to create a lean manufacturing model that maximizes margin potential while minimizing resource intensity.
- Enhanced Supply Chain Reliability: Supply continuity is often jeopardized by complex purification steps that are prone to failure or variability. By simplifying the isolation procedure to filtration and recrystallization, the risk of batch rejection is markedly lowered. The use of commodity chemicals and widely available catalysts like zinc nitrate ensures that the supply of raw materials is stable and not subject to the volatility of niche reagent markets. This reliability translates into shorter lead times and more dependable delivery schedules for downstream pharmaceutical customers who rely on these impurities for critical regulatory submissions and quality control testing.
- Scalability and Environmental Compliance: The scalability of this process is inherent in its design, which avoids unit operations that do not translate well from the lab to the plant. The mild reaction conditions and the absence of hazardous reagents enhance workplace safety and simplify environmental compliance protocols. Waste streams are easier to manage and treat due to the reduced volume of solvent waste and the absence of silica sludge. For organizations committed to sustainable manufacturing practices, this route offers a pathway to reduce the carbon footprint of their supply chain while maintaining the highest standards of product quality and purity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this Neratinib related substance. These insights are derived from the specific technical disclosures within the patent documentation, providing clarity on the feasibility and advantages of the described methodology. Understanding these details is crucial for stakeholders evaluating the integration of this intermediate into their broader quality assurance and supply strategies.
Q: What is the primary advantage of this new synthesis route for Neratinib impurities?
A: The primary advantage is the elimination of column chromatography purification. The process achieves over 99.0% purity through recrystallization alone, significantly reducing production costs and processing time compared to prior art methods.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial production. It utilizes mild reaction conditions, common solvents like toluene and acetonitrile that can be recycled, and avoids the need for specialized equipment, ensuring high safety and scalability.
Q: What is the overall yield and purity profile of the target compound?
A: The process yields the target Formula I compound with a total yield exceeding 35%. Crucially, the final product achieves a purity of greater than 99.0% without requiring complex purification steps, minimizing byproduct formation throughout the four-step sequence.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Neratinib Related Substance Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurities play in the development and approval of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN111848582A can be executed with precision at any volume. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to replicate and optimize such complex synthetic routes ensures that your supply of Neratinib related substances remains uninterrupted and compliant with global regulatory standards.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall procurement spend. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal quality benchmarks. Let us be your strategic partner in securing a reliable and cost-effective supply chain for your pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
