Revolutionizing Homopiperazine Production: A Safe, Scalable Route for Fasudil Intermediates
Introduction to Advanced Homopiperazine Manufacturing
The pharmaceutical landscape for cardiovascular treatments continues to evolve, driven by the demand for safer and more efficient synthesis of critical intermediates like homopiperazine. As detailed in the recent patent disclosure CN116143725A, a groundbreaking methodology has emerged that fundamentally shifts the production paradigm away from hazardous high-pressure processes. This innovation addresses the longstanding bottlenecks in manufacturing Fasudil Hydrochloride, a potent vasodilator used to treat cerebral vasospasm. By leveraging a straightforward cyclization and reduction strategy, this technology offers a robust alternative to legacy methods that have plagued the industry with safety concerns and operational complexity. For global supply chain leaders, understanding this shift is crucial for securing reliable sources of high-quality pharmaceutical intermediates.
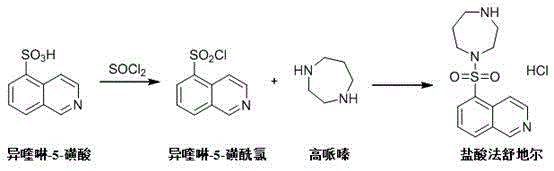
The significance of this patent extends beyond mere chemical novelty; it represents a strategic opportunity for cost optimization and risk mitigation in fine chemical manufacturing. Traditional routes often involve multiple protection-deprotection steps or dangerous hydrogenation reactions that require specialized infrastructure. In contrast, the disclosed method utilizes readily available starting materials to construct the seven-membered heterocyclic ring with remarkable efficiency. This report delves deep into the mechanistic advantages, commercial implications, and scalability of this new process, providing R&D directors and procurement managers with the insights needed to evaluate its adoption for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of homopiperazine has been fraught with significant technical and safety challenges that hinder efficient large-scale production. Conventional pathways, such as those utilizing N-(2-cyanoethyl)ethylenediamine or N-(β-hydroxyl)-1,3-propanediamine, typically rely on catalytic hydrogenation under high temperature and high pressure conditions. These processes necessitate the use of expensive noble metal catalysts, which not only drive up raw material costs but also introduce complexities regarding catalyst recovery and heavy metal residue control. Furthermore, the requirement for high-pressure reactors increases capital expenditure and imposes stringent safety protocols, creating potential bottlenecks in supply continuity. The inherent risks associated with handling hydrogen gas at elevated pressures make these traditional methods less attractive for modern, safety-conscious manufacturing facilities seeking to minimize operational hazards.
The Novel Approach
The innovative route described in patent CN116143725A circumvents these issues by employing a mild, two-step synthesis that avoids high-pressure hydrogenation entirely. This method initiates with the cyclization of ethylenediamine and malonyl chloride to form a cyclic diamide, followed by a reduction step using lithium aluminum hydride. By operating at atmospheric pressure and moderate temperatures, this approach drastically simplifies the equipment requirements and enhances process safety. The elimination of noble metal catalysts reduces the dependency on volatile precious metal markets, offering a more stable cost structure for long-term production planning. This streamlined pathway not only improves the economic viability of homopiperazine manufacturing but also aligns with green chemistry principles by reducing energy consumption and waste generation associated with harsh reaction conditions.
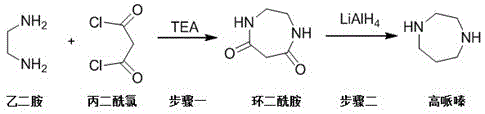
Mechanistic Insights into Cyclization and Hydride Reduction
The core of this technological breakthrough lies in the precise control of nucleophilic acyl substitution and subsequent amide reduction. In the first step, ethylenediamine acts as a nucleophile, attacking the electrophilic carbonyl carbons of malonyl chloride in the presence of a base like triethylamine. This reaction is conducted at low temperatures, specifically between -5°C and 5°C, to suppress intermolecular polymerization and ensure the formation of the desired seven-membered cyclic diamide rather than linear polyamides. The careful stoichiometric balance, with a molar ratio of triethylamine to ethylenediamine ranging from 1:1 to 3:1, is critical for scavenging the hydrochloric acid byproduct and driving the equilibrium towards cyclization. This controlled environment minimizes the formation of oligomeric impurities, setting a high baseline for the purity of the intermediate before the reduction step even begins.
Following the isolation of the cyclic diamide, the second stage involves a powerful reduction using lithium aluminum hydride (LiAlH4) in anhydrous tetrahydrofuran. This reagent is capable of reducing the robust amide bonds within the ring structure directly to amines, effectively converting the cyclic diamide into homopiperazine. The reaction is heated to reflux for approximately 12 hours to ensure complete conversion, with the molar ratio of LiAlH4 to substrate carefully optimized between 1:1 and 6:1 to account for reagent decomposition and ensure excess reducing power. The mechanism proceeds through the formation of an iminium ion intermediate, which is subsequently reduced to the secondary amine. This direct reduction pathway avoids the need for activating groups or protecting groups, thereby reducing the total number of unit operations and potential points of failure in the synthesis, ultimately leading to the reported gas chromatography purity of 99.99%.
How to Synthesize Homopiperazine Efficiently
Implementing this novel synthesis route requires strict adherence to the specified reaction parameters to maximize yield and safety. The process is designed to be operationally simple, making it highly accessible for facilities equipped with standard glass-lined or stainless steel reactors without the need for specialized high-pressure autoclaves. The initial cyclization step demands precise temperature control to manage the exothermic nature of the acid chloride reaction, while the subsequent reduction requires rigorous exclusion of moisture to maintain the efficacy of the lithium aluminum hydride. Detailed standard operating procedures regarding quenching protocols and workup strategies are essential to handle the aluminum salts generated during the reduction phase. For a comprehensive guide on the exact experimental conditions and workup procedures validated by the patent data, please refer to the technical instructions below.
- Cyclization: React ethylenediamine with malonyl chloride in dichloromethane using triethylamine as a base at -5 to 5°C to form cyclic diamide.
- Reduction: Reduce the cyclic diamide using lithium aluminum hydride in anhydrous tetrahydrofuran under reflux conditions for 12 hours.
- Purification: Quench the reaction carefully, filter salts, concentrate the filtrate, and perform vacuum distillation to obtain high-purity homopiperazine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this new manufacturing method offers compelling benefits that directly address the pain points of cost volatility and supply chain fragility. By shifting away from hydrogenation-based technologies, manufacturers can decouple their production costs from the fluctuating prices of noble metals like palladium and platinum. This transition allows for a more predictable budgeting model and reduces the exposure to supply disruptions often caused by geopolitical tensions affecting precious metal mining. Additionally, the use of commodity chemicals such as ethylenediamine and malonyl chloride ensures that raw material availability remains high, as these are produced on a massive global scale for various industries. This abundance of feedstock significantly lowers the risk of production stoppages due to raw material shortages, ensuring a steady flow of intermediates to downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts represents a direct and substantial saving in variable production costs. Traditional hydrogenation routes require significant capital investment in catalyst loading and subsequent recovery systems to meet regulatory limits on heavy metal residues. By replacing these with stoichiometric chemical reagents like lithium aluminum hydride, the process removes the need for complex catalyst filtration and metal scavenging steps. Furthermore, the mild reaction conditions reduce energy consumption associated with maintaining high pressures and temperatures, leading to lower utility bills. These cumulative efficiencies translate into a more competitive pricing structure for the final homopiperazine product, allowing buyers to negotiate better terms without compromising on quality standards.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup enhances the overall reliability of the supply chain by reducing the likelihood of equipment failure and safety incidents. High-pressure hydrogenation units are complex assets that require frequent maintenance and specialized operator training, creating potential single points of failure in a production schedule. In contrast, the atmospheric pressure operations of this new method can be performed in standard reactors that are widely available in most fine chemical plants. This flexibility allows for easier technology transfer between different manufacturing sites and enables rapid scaling of production capacity to meet surges in demand. Consequently, procurement managers can secure longer-term contracts with greater confidence in the supplier's ability to deliver on time.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is inherently safer and more straightforward due to the absence of explosive hydrogen gas handling. The waste profile of the reaction is also more manageable, primarily consisting of aluminum salts and organic solvents that can be treated using standard effluent treatment protocols. This aligns well with increasingly stringent environmental regulations, reducing the compliance burden and the risk of regulatory shutdowns. The ability to scale up without proportional increases in safety risks makes this route ideal for meeting the growing global demand for Fasudil and related cardiovascular medications. It supports sustainable manufacturing goals by minimizing the carbon footprint associated with high-energy高压 processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on safety, purity, and operational feasibility. Understanding these details is vital for technical teams evaluating the integration of this route into existing production lines. The responses highlight the specific advantages over prior art, focusing on the practical implications for quality control and process safety management systems.
Q: What are the safety advantages of this new homopiperazine synthesis method?
A: Unlike conventional methods that require high-pressure catalytic hydrogenation, this novel route operates under mild atmospheric conditions using standard reduction chemistry, significantly lowering explosion risks and equipment costs.
Q: Does this process rely on expensive noble metal catalysts?
A: No, the process utilizes commercially available cheap raw materials like ethylenediamine and malonyl chloride, completely eliminating the need for costly palladium or platinum catalysts often found in traditional hydrogenation routes.
Q: What purity levels can be achieved with this synthetic route?
A: The patented method demonstrates the capability to achieve extremely high purity, with gas chromatography analysis showing results up to 99.99%, making it highly suitable for sensitive pharmaceutical applications like Fasudil production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homopiperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global pharmaceutical market. We are committed to delivering homopiperazine with stringent purity specifications, utilizing advanced analytical techniques to verify that every batch meets the highest standards required for API synthesis. Our rigorous QC labs are equipped to detect and quantify trace impurities, guaranteeing the consistency and reliability necessary for your regulatory filings and clinical trials.
We invite you to collaborate with us to leverage this innovative synthesis technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization. Let us be your trusted partner in navigating the complexities of fine chemical sourcing and accelerating your path to market.
