Advanced Synthesis of 9-IHA Cisplatin-like Complexes for Next-Generation Anticancer Drug Development
The pharmaceutical industry is constantly seeking novel molecular entities that can overcome the limitations of established chemotherapeutic agents, particularly regarding drug resistance and systemic toxicity. Patent CN102268046A introduces a significant advancement in this domain by disclosing a novel cisplatin-like complex based on 9-imidanthrone hydrazone (9-IHA). This technology represents a strategic convergence of anthracycline chemistry and platinum coordination chemistry, aiming to create a dual-action therapeutic agent. The core innovation lies in the utilization of 9-IHA as an organic ligand that chelates with Pt(II) ions to form a stable five-membered ring structure. For R&D directors and procurement managers alike, this patent offers a compelling pathway for developing next-generation anticancer intermediates that potentially mitigate the severe nephrotoxicity and neurotoxicity associated with traditional cisplatin while maintaining potent antitumor efficacy. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such coordination compounds is essential for scaling production to meet the demands of modern oncology drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anthracycline antibiotics, while effective, suffer from a lack of selectivity, often causing severe damage to healthy tissues such as cardiac muscle, alongside the rapid development of multidrug resistance in tumor cells due to their单一 mechanism of DNA intercalation. Similarly, while cisplatin has been a cornerstone of chemotherapy for decades, its clinical utility is severely hampered by dose-limiting toxicities, including profound nephrotoxicity and ototoxicity, which restrict the dosage that can be safely administered to patients. Furthermore, tumor cells frequently develop resistance to cisplatin through enhanced DNA repair mechanisms or decreased drug accumulation, rendering the treatment ineffective over time. These inherent drawbacks of single-mechanism agents necessitate the exploration of hybrid molecules that can engage multiple biological targets simultaneously. The conventional synthesis of simple platinum salts or unmodified anthracyclines fails to address these complex biological challenges, leading to a high attrition rate in clinical trials and significant financial losses for pharmaceutical developers seeking cost reduction in API manufacturing.
The Novel Approach
The novel approach detailed in the patent leverages the principles of coordination chemistry to create a hybrid molecule, cis-[PtCl2(9-IHA)], which combines the DNA-intercalating properties of the anthracene moiety with the DNA-crosslinking capability of the platinum center. By coordinating the 9-IHA ligand through its imidazoline nitrogen atoms to the platinum ion, the resulting complex forms a robust five-membered chelate ring that stabilizes the metal center and modifies its reactivity profile. This structural modification is hypothesized to alter the pharmacokinetics and biodistribution of the platinum species, potentially reducing off-target toxicity while enhancing tumor selectivity. The synthesis allows for the use of readily available starting materials like K2PtCl4 or Na2PtCl4, making the process scalable and economically viable for commercial scale-up of complex pharmaceutical intermediates. This strategy effectively transforms a highly toxic ligand into a therapeutically viable candidate, demonstrating how rational molecular design can rescue promising but toxic scaffolds for clinical application.
Mechanistic Insights into N,N-Bidentate Chelation and Stability
The chemical integrity of the cis-[PtCl2(9-IHA)] complex is fundamentally derived from the specific coordination geometry adopted by the platinum(II) ion. In this configuration, the Pt(II) center adopts a square planar geometry, which is characteristic of d8 metal ions, and coordinates with two chloride ions and the two nitrogen atoms of the 9-IHA ligand. The 9-IHA ligand acts as a bidentate chelator, utilizing the nitrogen atoms on the imidazoline ring to form strong coordinate covalent bonds with the metal. This N,N-chelation mode creates a thermodynamically stable five-membered metallacycle, which prevents the premature dissociation of the ligand in physiological conditions before reaching the target site. The stability of this chelate is crucial for minimizing the release of free platinum ions in the bloodstream, which are primarily responsible for the non-specific toxicity observed with cisplatin. Understanding this mechanistic detail is vital for quality control teams ensuring high-purity anticancer intermediates, as any deviation in the coordination sphere could drastically alter the biological profile.
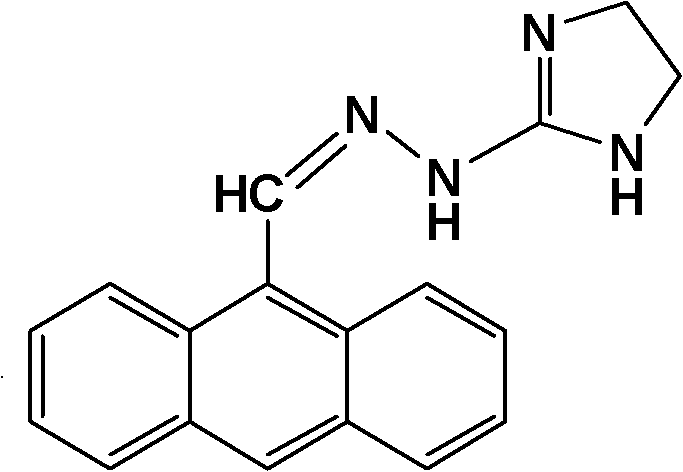
Furthermore, the presence of the bulky anthracene group attached to the hydrazone functionality introduces steric and electronic effects that influence the overall lipophilicity and cellular uptake of the complex. The planar anthracene system is known for its ability to intercalate between DNA base pairs, suggesting that the complex may retain this mode of action in addition to the platinum-mediated DNA crosslinking. This dual mechanism of action—intercalation plus alkylation—provides a synergistic effect that can overwhelm the DNA repair machinery of cancer cells, thereby overcoming resistance mechanisms that are effective against cisplatin alone. The patent data indicates that the complex maintains the structural integrity of the ligand while successfully incorporating the metal, as evidenced by spectroscopic analysis showing characteristic shifts in IR and NMR spectra. For supply chain heads, this implies that the synthesis must be tightly controlled to ensure the correct isomeric form (cis-configuration) is produced, as the trans-isomer would likely lack the requisite biological activity and geometric fit for DNA interaction.
How to Synthesize 9-IHA Platinum Complex Efficiently
The synthesis of this valuable intermediate can be achieved through robust and scalable methodologies that are compatible with industrial manufacturing standards. The patent describes two primary routes: a direct coordination method and an intermediate substitution method, both of which operate under relatively mild conditions ranging from 30°C to 90°C. These temperatures are easily achievable in standard glass-lined reactors, eliminating the need for specialized high-pressure or cryogenic equipment, which significantly lowers the capital expenditure required for production. The process utilizes common polar solvents such as methanol, ethanol, and water, which are inexpensive and easy to recover and recycle, further contributing to cost reduction in API manufacturing. The reaction times, varying from 1 to 48 hours depending on the specific conditions, allow for flexible scheduling in multi-purpose production facilities. Detailed standardized synthesis steps for optimizing yield and purity are provided in the guide below to assist technical teams in replicating these results.
- Dissolve the platinum salt (K2PtCl4 or Na2PtCl4) in distilled water and the 9-IHA ligand in a polar organic solvent like methanol or ethanol.
- Mix the solutions under nitrogen protection and heat the reaction mixture to a temperature range of 30-90°C for 1 to 48 hours.
- Filter the resulting dark brown precipitate, wash thoroughly with water and organic solvents to remove impurities, and dry under vacuum.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthesis route offers substantial strategic advantages regarding raw material sourcing and process efficiency. The starting materials, specifically the platinum salts and the anthracene-derived ligands, are commercially available from established chemical suppliers, ensuring a stable and continuous supply chain without reliance on exotic or restricted precursors. The simplicity of the workup procedure, which involves precipitation, filtration, and washing, eliminates the need for complex chromatographic purification steps that are often bottlenecks in large-scale production. This streamlined downstream processing significantly reduces the consumption of silica gel and organic solvents, leading to a drastic simplification of the waste management protocol and a reduction in the overall environmental footprint of the manufacturing process. For supply chain leaders, this translates to reducing lead time for high-purity pharmaceutical intermediates, as the shorter processing time allows for faster turnover of batches and quicker response to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts often required in cross-coupling reactions, replaced here by straightforward coordination chemistry, leads to significant optimization in the bill of materials. The ability to use water as a co-solvent further reduces the volume of organic solvents required, lowering both procurement costs and hazardous waste disposal fees. Additionally, the high yield reported in the patent examples suggests that raw material utilization is efficient, minimizing the loss of valuable platinum resources which are a major cost driver in metal-based drug synthesis. By avoiding complex multi-step organic syntheses for the core scaffold and focusing on a convergent assembly strategy, manufacturers can achieve substantial cost savings while maintaining high product quality standards required for GMP compliance.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity. The use of stable intermediates like cis-[PtCl2(DMSO)2] allows for a modular approach where key components can be stockpiled and reacted on demand, providing flexibility in production planning. This resilience against process variability minimizes the risk of batch failures and subsequent supply disruptions, ensuring that downstream drug formulation teams receive their materials on schedule. Furthermore, the solid nature of the final product facilitates easy storage and transportation, reducing the logistical complexities associated with handling unstable liquid intermediates or sensitive biological materials.
- Scalability and Environmental Compliance: The synthesis protocol is inherently scalable, moving seamlessly from gram-scale laboratory experiments to kilogram or ton-scale commercial production without requiring fundamental changes to the reaction engineering. The precipitation of the product as a solid simplifies isolation and reduces the energy intensity associated with solvent evaporation, aligning with green chemistry principles. The ability to wash the product with common solvents to remove unreacted salts ensures that the final material meets stringent purity specifications with minimal environmental impact. This alignment with sustainability goals is increasingly important for pharmaceutical companies aiming to reduce their carbon footprint and comply with evolving environmental regulations regarding heavy metal discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this cisplatin-like complex. These insights are derived directly from the experimental data and theoretical framework presented in the patent documentation. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their drug discovery or development programs. The answers provide clarity on safety profiles, synthetic versatility, and biological potential, helping to de-risk the investment in this technology.
Q: How does the toxicity of the 9-IHA platinum complex compare to the free ligand?
A: According to patent data, the cis-[PtCl2(9-IHA)] complex exhibits significantly lower cytotoxicity towards normal human liver cells compared to the free 9-IHA ligand, indicating an improved safety profile for potential therapeutic applications.
Q: What are the primary synthesis methods for this cisplatin-like complex?
A: The patent outlines two main approaches: a direct coordination method using potassium or sodium tetrachloroplatinate salts, and an intermediate method utilizing cis-[PtCl2(DMSO)2], both yielding the target complex through N,N-bidentate chelation.
Q: Does this complex show activity against drug-resistant tumor lines?
A: The complex demonstrates selective antitumor activity against specific cell lines like SK-OV-3 and BEL7404, with efficacy comparable to clinical drugs like cisplatin and amsacridine, suggesting potential utility in overcoming certain resistance mechanisms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-IHA Cisplatin Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-organic coordination compounds in modern oncology therapy. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless and efficient. We are committed to delivering high-purity intermediates that meet rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Whether you require custom synthesis of the 9-IHA ligand or the final platinum complex, our facility is equipped to handle the specific challenges of metal-containing pharmaceuticals, including heavy metal control and residual solvent analysis. Partnering with us means gaining access to a supply chain that prioritizes reliability, quality, and regulatory compliance.
We invite you to collaborate with us to optimize your supply chain for this promising anticancer candidate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity specifications. By leveraging our manufacturing expertise, you can accelerate your development timeline and reduce overall project costs. Please contact us today to request specific COA data and route feasibility assessments for the cis-[PtCl2(9-IHA)] complex. Let us help you bring this innovative therapeutic concept to reality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
