Scalable Synthesis Of L-Phenylalanine Modified Maslinic Acid Derivatives For Anticancer Drug Development
Introduction To Novel Antitumor Triterpenoid Intermediates
The pharmaceutical industry is constantly seeking novel scaffolds that can offer improved therapeutic indices for oncology applications. A significant breakthrough in this domain is documented in patent CN109517026B, which discloses a series of L-phenylalanine-modified maslinic acid derivatives. Maslinic acid, a natural pentacyclic triterpenoid found in olive oil and hawthorn, possesses inherent biological activities, but its modification with essential amino acids like L-phenylalanine creates entirely new chemical entities with enhanced pharmacological profiles. These derivatives are designed to leverage the metabolic pathways of tumor cells, which have a high demand for amino acids, thereby improving drug uptake and efficacy. The patent outlines the synthesis of eight distinct compounds, ranging from protected intermediates to final hydrochloride salts, providing a comprehensive library for drug discovery teams.
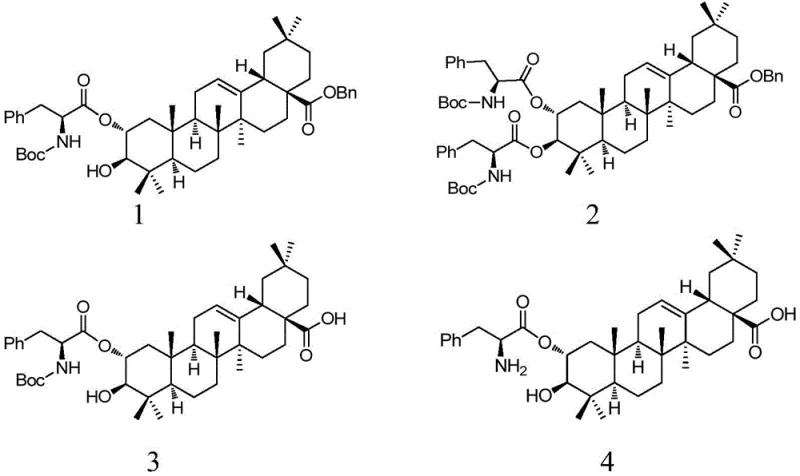
For procurement and supply chain professionals, understanding the structural complexity of these molecules is vital for sourcing strategies. The structures feature a rigid triterpenoid core coupled with flexible peptide chains, requiring precise stereochemical control during manufacturing. As a reliable pharmaceutical intermediate supplier, ensuring the integrity of the chiral centers in the L-phenylalanine moiety is paramount. The ability to produce these high-purity maslinic acid derivatives consistently allows research teams to accelerate their preclinical screening processes without being bottlenecked by material availability. This technology represents a bridge between natural product chemistry and modern peptide-drug conjugate strategies.
The Limitations Of Conventional Methods Vs. The Novel Approach
The Limitations Of Conventional Methods
Traditionally, modifying the carboxylic acid group at the C-28 position of triterpenoids like maslinic acid has been fraught with challenges regarding selectivity and yield. Conventional amidation or esterification methods often require harsh activating agents or extreme temperatures that can degrade the sensitive olefinic bond at the C-12 position or epimerize the chiral centers on the ring system. Furthermore, direct coupling without protecting groups on the hydroxyl functionalities at C-2 and C-3 can lead to a mixture of regioisomers, complicating downstream purification and significantly increasing the cost of goods sold. Older methods also frequently relied on heavy metal catalysts or stoichiometric amounts of toxic reagents, creating substantial environmental burdens and waste disposal issues that modern green chemistry initiatives seek to eliminate.
The Novel Approach
The methodology presented in the patent overcomes these historical hurdles through a sophisticated protection-group strategy. By initially converting maslinic acid into benzyl maslinate, the C-28 carboxylic acid is activated and protected simultaneously, preventing unwanted side reactions. The subsequent coupling with Boc-L-phenylalanine utilizes mild conditions, specifically employing dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) at room temperature. This approach ensures that the stereochemistry of the amino acid is preserved while achieving high conversion rates. The novelty lies not just in the coupling, but in the sequential deprotection steps that allow for the isolation of specific intermediates (Compounds 1-8) with varying degrees of functionality, offering flexibility for further derivatization or direct biological testing.
Mechanistic Insights Into Peptide Coupling And Catalytic Hydrogenolysis
The core of this synthesis relies on two fundamental organic transformations: Steglich esterification and catalytic transfer hydrogenation. In the first step, the reaction between benzyl maslinate and Boc-L-phenylalanine proceeds via the formation of an O-acylisourea intermediate generated by DCC. DMAP acts as a nucleophilic catalyst, attacking this intermediate to form a highly reactive acylpyridinium species, which is then attacked by the hydroxyl group of the triterpenoid or the carboxyl group depending on the specific target compound. This mechanism is crucial for cost reduction in API manufacturing as it operates efficiently at ambient temperatures, reducing energy consumption. The use of Boc (tert-butyloxycarbonyl) protection on the amine prevents self-polymerization of the amino acid, ensuring that the coupling occurs exclusively at the desired site.
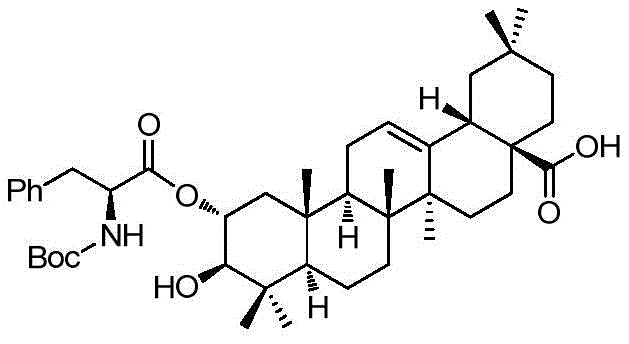
The second critical mechanistic phase involves the removal of the benzyl protecting group to reveal the free carboxylic acid, as seen in the conversion of Compound 1 to Compound 3. Instead of using high-pressure hydrogen gas which requires specialized autoclaves and poses safety risks, the patent employs catalytic transfer hydrogenation using ammonium formate and 10% Pd/C. In this mechanism, ammonium formate decomposes on the palladium surface to generate nascent hydrogen, which reduces the benzyl ester to a toluene byproduct and the free acid. This technique is particularly advantageous for the commercial scale-up of complex polymer additives and pharmaceutical intermediates because it can be performed in standard glass-lined reactors under reflux conditions (40-80°C), drastically simplifying the engineering requirements and enhancing supply chain reliability.
How To Synthesize L-Phenylalanine Modified Maslinic Acid Efficiently
Executing this synthesis requires strict adherence to the stoichiometric ratios and purification protocols outlined in the patent to ensure the high purity required for biological testing. The process begins with the dissolution of benzyl maslinate in a chlorinated solvent like dichloromethane, followed by the sequential addition of the amino acid derivative and coupling agents. Reaction monitoring via thin-layer chromatography (TLC) is essential to determine the endpoint, typically achieved within 8 to 24 hours at room temperature. Following the coupling, the workup involves filtration to remove dicyclohexylurea byproducts and silica gel column chromatography to separate the mono-substituted and di-substituted products. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Perform Steglich esterification of benzyl maslinate with Boc-L-phenylalanine using DCC and DMAP in dichloromethane at room temperature.
- Execute catalytic transfer hydrogenation using ammonium formate and 10% Pd/C in methanol at 55°C to remove benzyl protecting groups.
- Conduct final deprotection using saturated hydrogen chloride ethyl acetate solution to yield the target hydrochloride salts.
Commercial Advantages For Procurement And Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers multiple levers for optimizing the supply chain and reducing overall production costs without compromising quality. The reliance on commercially available starting materials such as maslinic acid (extracted from abundant natural sources like olive pomace) and standard protected amino acids ensures a stable raw material supply base. This reduces the risk of shortages that often plague synthetic routes dependent on exotic or custom-synthesized reagents. Furthermore, the mild reaction conditions translate directly into lower utility costs and reduced wear and tear on manufacturing equipment, contributing to substantial cost savings over the lifecycle of the product.
- Cost Reduction In Manufacturing: The elimination of high-pressure hydrogenation equipment in favor of transfer hydrogenation using ammonium formate represents a significant capital expenditure saving. By avoiding the need for specialized autoclaves and the associated safety infrastructure, manufacturers can utilize existing standard reactor trains. Additionally, the use of DCC/DMAP coupling is a well-understood process with predictable kinetics, minimizing batch failures and rework. The ability to purify intermediates using standard silica chromatography rather than preparative HPLC further drives down the cost of goods, making these high-value intermediates more accessible for early-stage drug development.
- Enhanced Supply Chain Reliability: The robustness of the chemical steps ensures consistent batch-to-batch reproducibility, which is critical for maintaining regulatory compliance. Since the reagents involved (DCC, DMAP, Pd/C, Ammonium Formate) are commodity chemicals with multiple global suppliers, the risk of single-source dependency is minimized. This diversification allows procurement managers to negotiate better pricing and secure longer-term contracts. The stability of the intermediates, such as the Boc-protected compounds, also allows for inventory buffering, enabling the supply chain to absorb demand fluctuations without disrupting the production schedule for downstream API synthesis.
- Scalability And Environmental Compliance: The process is inherently scalable from gram to kilogram and eventually to metric ton quantities. The waste streams generated are primarily organic solvents and urea byproducts, which are easier to treat and dispose of compared to heavy metal waste from other catalytic systems. The transfer hydrogenation method produces nitrogen and carbon dioxide as gaseous byproducts, simplifying off-gas treatment. This alignment with green chemistry principles not only reduces environmental fees but also enhances the corporate sustainability profile of the manufacturing partner, a key metric for modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triterpenoid derivatives. Understanding these details helps stakeholders make informed decisions about integrating these materials into their development pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for R&D and procurement teams evaluating this technology.
Q: What is the biological activity of these L-phenylalanine modified maslinic acid derivatives?
A: According to patent CN109517026B, these compounds exhibit significant proliferation inhibitory activity against various tumor cell lines including human bladder cancer (T24), gastric cancer (MGC-803), and liver cancer (HepG2), with IC50 values demonstrating potent anticancer potential compared to unmodified maslinic acid.
Q: How does the novel synthesis method improve upon conventional triterpenoid modification?
A: The patented method utilizes a strategic protection-deprotection strategy involving benzyl esters and Boc groups, allowing for high regioselectivity and yield. The use of catalytic transfer hydrogenation avoids high-pressure hydrogen gas equipment, enhancing safety and scalability for industrial production.
Q: Are these intermediates suitable for large-scale commercial manufacturing?
A: Yes, the synthesis relies on standard organic transformations such as DCC coupling and palladium-catalyzed reduction which are well-established in process chemistry. The reaction conditions are mild (often room temperature to 60°C), facilitating easy scale-up from laboratory to commercial tonnage.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable L-Phenylalanine Modified Maslinic Acid Supplier
As the demand for specialized oncology intermediates grows, partnering with an experienced CDMO is essential for navigating the complexities of process development. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to market launch. Our rigorous QC labs and stringent purity specifications guarantee that every batch of L-phenylalanine modified maslinic acid meets the highest international standards, minimizing the risk of delays in your regulatory filings. We understand that time-to-market is critical in the competitive landscape of anticancer drug development.
We invite you to contact our technical procurement team to discuss your specific requirements. We can provide a Customized Cost-Saving Analysis tailored to your projected volumes, helping you identify the most economical route for your supply needs. Whether you need specific COA data for a particular batch or route feasibility assessments for analogues, our experts are ready to support your goals. Let us help you secure a reliable supply of these high-purity anticancer intermediates to accelerate your path to commercial success.
