Advanced Copper-Catalyzed Synthesis of 4-Phenoxybenzoic Acid for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce critical intermediates, and the synthesis of 4-phenoxybenzoic acid stands as a prime example of this technological evolution. As a key building block for the synthesis of Ibrutinib, a potent antitumor drug, as well as various agrochemicals and functional materials, the demand for high-purity 4-phenoxybenzoic acid is escalating. Patent CN115594573A introduces a groundbreaking copper catalytic coupling technology that addresses the longstanding inefficiencies of traditional manufacturing methods. This innovation not only streamlines the synthetic route but also aligns with the modern imperative for greener, more cost-effective chemical production. By leveraging a specific copper catalyst and ligand system, this method achieves superior yields under milder conditions, positioning it as a vital asset for R&D directors and procurement managers aiming to optimize their supply chains for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
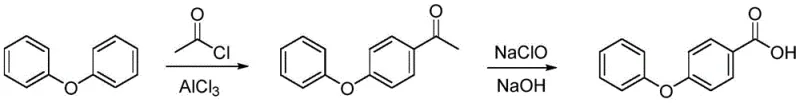
Historically, the industrial production of 4-phenoxybenzoic acid has relied on routes that are increasingly untenable in the context of modern environmental and economic standards. The first conventional route involves a Friedel-Crafts reaction between diphenyl ether and acetyl chloride, followed by oxidation with sodium hypochlorite. This process is fraught with significant drawbacks, primarily the consumption of large quantities of aluminum trichloride and the generation of substantial hazardous waste. Furthermore, the oxidation step using sodium hypochlorite often results in low production capacity and inconsistent quality, driving up the overall manufacturing cost. The second reported route utilizes p-chlorobenzoic acid and sodium phenolate at high temperatures ranging from 150°C to 160°C. While this method shortens the step count, it necessitates the use of tetrahydronaphthalene, an expensive solvent, and requires special equipment to withstand the harsh thermal conditions, thereby creating barriers to efficient industrial scaling and increasing capital expenditure.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a copper-catalyzed coupling reaction between p-chlorobenzoic acid and phenol. This method operates under significantly milder conditions, typically between 110°C and 130°C, which reduces energy consumption and thermal stress on the equipment. The use of a copper catalyst, such as cuprous chloride, in conjunction with specific ligands and bases like sodium hydroxide, allows for a direct coupling that bypasses the need for hazardous oxidants or expensive high-boiling solvents. The reaction system is versatile, accommodating solvent mixtures like water and n-butanol, which are not only cheaper but also easier to recover and recycle. This shift represents a fundamental improvement in process chemistry, offering a pathway that is both economically superior and environmentally more sustainable, directly addressing the pain points of waste management and raw material costs faced by supply chain heads.

Mechanistic Insights into Copper-Catalyzed Coupling
The core of this technological advancement lies in the precise orchestration of the copper catalytic cycle, which facilitates the formation of the carbon-oxygen bond between the aryl chloride and the phenol. The mechanism involves the oxidative addition of the aryl chloride to the copper center, followed by ligand exchange with the phenolate anion generated in situ by the base. The presence of specialized ligands, such as the oxalamide derivatives described in the patent (Formula I), is critical for stabilizing the copper species and accelerating the reductive elimination step that releases the product. This catalytic system is highly tunable; by selecting specific ligands like L1, L2, L3, or L4, chemists can fine-tune the electronic and steric environment around the metal center. This optimization ensures high turnover numbers and minimizes the formation of side products, which is essential for maintaining the stringent purity profiles required in pharmaceutical intermediate manufacturing.
Impurity control is another critical aspect where this mechanism excels, particularly for R&D directors focused on product quality. The mild reaction conditions prevent the thermal degradation of sensitive functional groups, which is a common issue in the high-temperature conventional routes. Furthermore, the specific choice of base and solvent system helps to suppress homocoupling side reactions that often plague copper-catalyzed processes. The post-treatment process, involving pH adjustment and recrystallization from isopropanol, further purifies the crude product by removing residual catalyst and unreacted starting materials. This robust control over the impurity profile ensures that the final 4-phenoxybenzoic acid meets the rigorous specifications necessary for downstream drug synthesis, reducing the risk of batch rejection and ensuring consistent supply continuity for global pharmaceutical partners.
How to Synthesize 4-Phenoxybenzoic Acid Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to maximize yield and purity. The process begins with the precise charging of reactants, where the molar ratios of p-chlorobenzoic acid, phenol, catalyst, and ligand must be carefully controlled to drive the equilibrium towards the product. The patent specifies that a catalyst loading as low as 0.5 mol% can be effective, which is a significant advantage for cost management. The reaction is typically conducted in a mixed solvent system, such as water and n-butanol, which provides a balance between solubility and ease of product isolation. Detailed standardized synthesis steps are crucial for reproducibility, especially when scaling from laboratory to pilot plant.
- Charge p-chlorobenzoic acid, phenol, copper catalyst (e.g., CuCl), ligand, and base into a reactor with an inert solvent system such as water and n-butanol.
- Heat the reaction mixture to 110-130°C and maintain stirring for approximately 10 hours to ensure complete coupling conversion.
- Cool the mixture, adjust pH to 3-5 with acid to precipitate the crude product, then recrystallize from isopropanol to obtain high-purity 4-phenoxybenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed technology offers tangible strategic benefits that extend beyond simple chemistry. The elimination of expensive and hazardous reagents like aluminum trichloride and tetrahydronaphthalene directly translates to a reduction in raw material procurement costs and waste disposal fees. Moreover, the use of common solvents like water and alcohols simplifies the supply chain logistics, as these materials are readily available globally and are not subject to the same regulatory restrictions as specialized chlorinated solvents. This enhanced availability ensures a more resilient supply chain, reducing the risk of production delays caused by raw material shortages. The milder reaction conditions also mean that existing standard reactor infrastructure can be utilized without the need for costly upgrades to high-pressure or high-temperature equipment, further lowering the barrier to entry for commercial production.
- Cost Reduction in Manufacturing: The economic impact of this new method is profound, primarily driven by the substitution of costly reagents with inexpensive copper salts and common bases. By avoiding the use of sodium hypochlorite and aluminum trichloride, manufacturers can significantly lower the variable costs associated with each production batch. Additionally, the simplified workup procedure, which involves straightforward filtration and recrystallization, reduces labor hours and energy consumption compared to the complex separation processes required for the old oxidation routes. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Reliability is a cornerstone of effective supply chain management, and this process enhances it by utilizing widely available starting materials. P-chlorobenzoic acid and phenol are commodity chemicals with stable global supply networks, unlike some specialized intermediates required for other synthetic routes. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in operational parameters, leading to higher batch success rates and more predictable delivery schedules. This stability is crucial for long-term supply agreements with multinational pharmaceutical companies, where consistency and on-time delivery are often weighted more heavily than marginal price differences.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this copper-catalyzed method is inherently designed for industrial scalability. The use of aqueous solvent systems reduces the fire hazard and environmental toxicity associated with organic solvents, facilitating easier regulatory approval and permitting. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, minimizing the risk of compliance-related shutdowns. Furthermore, the high atom economy of the coupling reaction ensures that a greater proportion of raw materials are converted into the desired product, reducing the overall environmental footprint and supporting the sustainability goals of modern chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-phenoxybenzoic acid using this advanced copper-catalyzed technology. These insights are derived directly from the patent data and are intended to clarify the feasibility and benefits of adopting this route for commercial manufacturing. Understanding these details is essential for stakeholders evaluating the potential integration of this technology into their existing production portfolios.
Q: What are the advantages of the copper-catalyzed route over traditional Friedel-Crafts methods?
A: The copper-catalyzed method eliminates the need for hazardous aluminum trichloride and sodium hypochlorite, significantly reducing waste generation and improving environmental compliance compared to traditional oxidation routes.
Q: What is the expected purity level achievable with this synthesis method?
A: Experimental data from the patent indicates that recrystallization from isopropanol yields 4-phenoxybenzoic acid with purity levels exceeding 99%, meeting stringent specifications for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and easily available catalysts and solvents like water and alcohols, avoiding high-pressure or specialized equipment, which makes it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Phenoxybenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the pharmaceutical supply chain. Our team of CDMO experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless. We are committed to delivering high-purity 4-phenoxybenzoic acid that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing our partners with a secure and reliable source of critical intermediates for their drug development programs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with us, you gain access to not just a product, but a comprehensive solution that enhances your competitive edge in the global pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
