Advanced Synthesis of 28-(L-Phenylalanine)-Pentacyclic Triterpene Derivatives for Oncology
Advanced Synthesis of 28-(L-Phenylalanine)-Pentacyclic Triterpene Derivatives for Oncology
Introduction to Novel Antitumor Intermediates
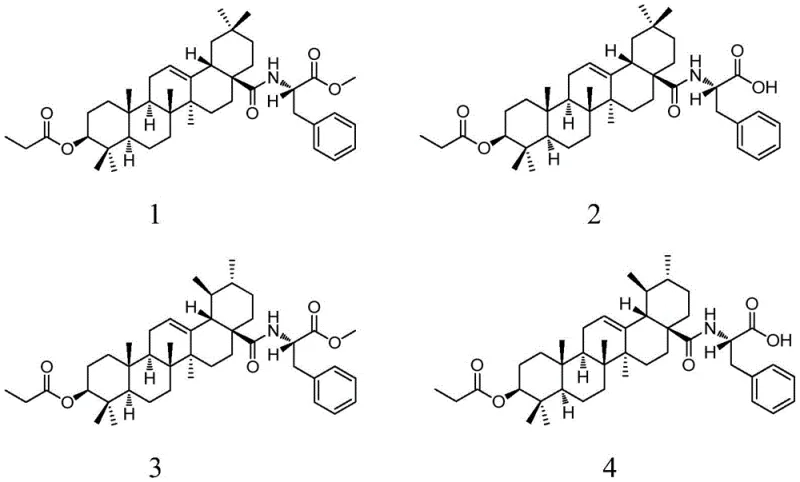
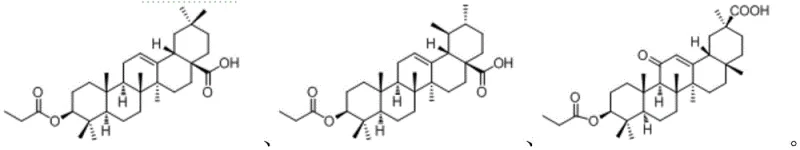
The pharmaceutical landscape is constantly evolving with the demand for more effective antitumor agents that possess higher selectivity and lower toxicity profiles. Patent CN109517025B introduces a groundbreaking series of 28-(L-phenylalanine)-pentacyclic triterpene derivatives, representing a significant leap forward in the field of pharmaceutical intermediates. These compounds, derived from natural triterpenic acid scaffolds such as oleanolic acid, ursolic acid, and glycyrrhetinic acid, are chemically modified at the C-28 position with L-phenylalanine. This strategic conjugation leverages the essential amino acid's metabolic pathways to potentially enhance cellular uptake in tumor cells, which are known for their high nutrient demand. The structural diversity offered by this patent covers six distinct derivatives, providing a robust library for drug discovery teams to explore structure-activity relationships. As a reliable pharmaceutical intermediates supplier, understanding the nuances of this synthesis is critical for scaling these molecules from benchtop research to commercial manufacturing. The integration of amino acid moieties into complex natural product skeletons not only modifies physicochemical properties but also opens new avenues for prodrug design. This report delves deep into the technical feasibility, mechanistic insights, and commercial viability of producing these high-value compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the modification of pentacyclic triterpenes has been fraught with challenges related to regioselectivity and reaction efficiency. Conventional methods often rely on harsh conditions that can degrade the sensitive triterpene skeleton or result in complex mixtures of isomers that are difficult to separate. Many existing protocols for introducing nitrogen-containing functional groups require expensive coupling reagents or transition metal catalysts that leave behind toxic residues, necessitating costly purification steps to meet pharmaceutical standards. Furthermore, the poor solubility of native triterpenic acids in common organic solvents often limits reaction concentrations, leading to lower throughput and higher solvent consumption. In the context of cost reduction in pharmaceutical intermediates manufacturing, these inefficiencies translate directly into higher production costs and longer lead times. The lack of a standardized, scalable route for C-28 amidation has historically been a bottleneck for researchers aiming to develop triterpene-based therapeutics. Without a clear pathway to high-purity derivatives, the commercial potential of these natural products remains underutilized, hindering their progression through clinical pipelines.
The Novel Approach
The methodology outlined in CN109517025B offers a transformative solution by employing a two-step amidation and hydrolysis strategy that is both robust and scalable. By first activating the C-28 carboxylic acid of 3-propionyloxy-triterpenic acids using thionyl chloride or oxalyl chloride, the process creates a highly reactive acyl chloride intermediate that readily reacts with L-phenylalanine methyl ester. This approach avoids the need for expensive peptide coupling reagents in the initial step, significantly simplifying the reaction setup. The subsequent hydrolysis of the methyl ester group under controlled alkaline conditions allows for the precise generation of the free acid derivatives without compromising the integrity of the newly formed amide bond. This novel approach ensures high yields, with specific examples demonstrating conversion rates up to 94% for certain derivatives, indicating a highly efficient process. The use of readily available starting materials and common solvents like dichloromethane and methanol further enhances the practicality of this method for industrial application. This streamlined synthesis not only improves the economic feasibility but also aligns with green chemistry principles by reducing waste and energy consumption compared to traditional multi-step functionalization routes.
Mechanistic Insights into Amidation and Hydrolysis
The core chemical transformation in this patent relies on a classic nucleophilic acyl substitution mechanism, optimized for the steric hindrance inherent in pentacyclic triterpene structures. The initial activation step involves the conversion of the C-28 carboxylic acid into an acid chloride using thionyl chloride (SOCl2) at elevated temperatures, typically around 72°C. This step is crucial as it converts a poor electrophile into a highly reactive species capable of overcoming the steric bulk of the triterpene backbone. The subsequent addition of L-phenylalanine methyl ester in the presence of a weak base, such as saturated sodium bicarbonate, facilitates the nucleophilic attack of the amino group on the carbonyl carbon. The base serves a dual purpose: it neutralizes the hydrochloric acid byproduct generated during the reaction and maintains the amino group in its unprotonated, nucleophilic form. This careful balance of pH is essential to prevent the hydrolysis of the acid chloride before it can react with the amine. The result is the formation of a stable amide bond, linking the triterpene scaffold with the amino acid side chain. This mechanistic pathway is highly reproducible and allows for the synthesis of various derivatives by simply changing the triterpene starting material, as demonstrated by the successful synthesis of oleanolic, ursolic, and glycyrrhetinic acid conjugates.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this patent details specific strategies to ensure high-purity [Pharmaceutical Intermediates]. The reaction mixture often contains excess reagents, such as unreacted L-phenylalanine methyl ester or residual thionyl chloride, which must be rigorously removed. The protocol specifies a workup procedure involving dispersion in dilute hydrochloric acid, which effectively neutralizes excess weak bases and converts unreacted amines into water-soluble salts that can be washed away. Following this, extraction with organic solvents like dichloromethane or ethyl acetate isolates the desired product from the aqueous phase. For further purification, silica gel column chromatography is employed using a gradient of petroleum ether and ethyl acetate. The specific eluent ratios are optimized for each compound; for instance, compounds 1 and 3 require a less polar system (30:1 to 5:1), while compounds 2 and 4, which contain free carboxylic acids, require a more polar system (3:1 to 1:1). This tailored purification approach ensures that the final products meet stringent purity specifications, removing trace impurities that could affect biological activity or safety profiles. The ability to consistently produce high-purity material is a key factor in reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for re-processing.
How to Synthesize 28-(L-Phenylalanine)-Pentacyclic Triterpene Derivatives Efficiently
The synthesis of these complex molecules requires precise control over reaction parameters to maximize yield and purity. The process begins with the dissolution of the 3-propionyloxy-triterpenic acid in a suitable organic solvent, followed by the dropwise addition of the activating agent. Temperature control is critical during this phase to prevent side reactions. Once the acid chloride is formed, the solvent is removed under reduced pressure, and the residue is redissolved for the coupling step. The addition of the amino acid ester must be performed under ice-bath conditions to manage the exothermic nature of the amidation. After the reaction is complete, typically monitored by TLC, the crude product is isolated and subjected to the specific purification protocol outlined in the patent. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different batches and scales.
- Activation of 3-propionyloxy-triterpenic acids using thionyl chloride or oxalyl chloride in organic solvents like dichloromethane.
- Coupling the activated acid intermediate with L-phenylalanine methyl ester under weak alkaline conditions to form the amide bond.
- Hydrolysis of the methyl ester group using strong alkaline solutions to yield the final free acid derivatives with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthesis route described in CN109517025B offers substantial strategic advantages. The reliance on commodity chemicals such as thionyl chloride, L-phenylalanine, and common organic solvents means that the raw material supply chain is robust and less susceptible to market volatility. This stability is crucial for ensuring supply continuity for long-term drug development projects. Furthermore, the elimination of expensive transition metal catalysts or specialized coupling reagents significantly reduces the bill of materials, leading to substantial cost savings in manufacturing. The high yields reported for several derivatives, such as the 94% yield for Compound 2, indicate a process that is material-efficient, minimizing waste disposal costs and maximizing output per batch. These factors combined make the commercial scale-up of complex pharmaceutical intermediates much more feasible and economically attractive for large-scale production.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of precious metal catalysts and expensive coupling reagents like HATU for the majority of the derivatives, relying instead on cost-effective thionyl chloride activation. This substitution drastically lowers the raw material costs associated with each batch. Additionally, the high conversion efficiencies observed in the patent data mean that less starting material is wasted, further driving down the cost per gram of the final active intermediate. The simplified workup procedures also reduce labor and solvent consumption, contributing to an overall leaner manufacturing process that enhances profit margins without compromising quality.
- Enhanced Supply Chain Reliability: All key reagents, including 3-propionyloxy-triterpenic acids and L-phenylalanine derivatives, are commercially available from multiple global suppliers. This multi-sourcing capability mitigates the risk of supply disruptions that can occur with proprietary or single-source reagents. The use of standard solvents like dichloromethane and methanol ensures that procurement teams can leverage existing supply contracts and logistics networks. This reliability is essential for maintaining consistent production schedules and meeting the demanding timelines of pharmaceutical clients who require just-in-time delivery of critical intermediates for their clinical trials.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry, such as reflux, distillation, and column chromatography. The absence of toxic heavy metals simplifies the environmental compliance landscape, reducing the burden on wastewater treatment facilities and lowering the cost of waste disposal. The ability to scale from 100 kgs to 100 MT annual commercial production is supported by the robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without significant loss in yield. This scalability ensures that the supply chain can grow in tandem with the clinical and commercial demands of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triterpene derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is vital for stakeholders evaluating the feasibility of incorporating these intermediates into their drug discovery pipelines. The information covers aspects ranging from structural characteristics to purification standards, ensuring a comprehensive understanding of the product profile.
Q: What are the primary structural advantages of these triterpene derivatives?
A: The conjugation of L-phenylalanine at the C-28 position enhances water solubility and biological targeting compared to native triterpenes, potentially improving antitumor efficacy.
Q: Is the synthesis process scalable for commercial production?
A: Yes, the protocol utilizes standard reagents like thionyl chloride and common organic solvents, avoiding exotic catalysts, which facilitates straightforward scale-up from grams to tons.
Q: What purification methods ensure high purity for pharmaceutical use?
A: The patent specifies silica gel column chromatography with petroleum ether and ethyl acetate systems, alongside acid-base washes to remove unreacted amines and acids effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 28-(L-Phenylalanine)-Pentacyclic Triterpene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation antitumor therapies. Our team of expert chemists has extensively analyzed the synthetic pathways described in CN109517025B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that verify every batch against the highest industry standards. We understand that the success of your drug candidate depends on the reliability of your supply chain, and we are committed to being a partner you can trust for long-term collaboration.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current sourcing strategy or require specific COA data to validate our capabilities, we are ready to provide the information you need. We encourage you to request route feasibility assessments to explore how our manufacturing expertise can optimize your production timeline and reduce overall costs. By partnering with us, you gain access to a wealth of technical knowledge and industrial capacity that can accelerate your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
