Scalable Synthesis of Deuterated Flavonoids for Next-Generation Pharmaceutical Intermediates
The pharmaceutical industry is increasingly recognizing the strategic value of deuterated compounds as a means to optimize pharmacokinetic profiles without altering the fundamental pharmacology of a drug candidate. Patent CN109879847B introduces a groundbreaking preparation method for deuterated flavonoid compounds, addressing a critical need for stable isotopically labeled intermediates in modern drug discovery. This technology leverages a direct hydrogen-deuterium exchange mechanism under relatively mild conditions, utilizing heavy water as the deuterium source and alkali catalysts to achieve high substitution rates. For R&D directors and procurement specialists, this represents a significant opportunity to access high-purity deuterated building blocks such as deuterated baicalein and quercetin, which are essential for developing next-generation therapeutics with improved metabolic stability and reduced toxicity profiles.
The significance of this patent lies in its ability to transform common natural product scaffolds into valuable deuterated assets through a streamlined process. Unlike traditional multi-step syntheses that require constructing the flavonoid core from scratch with deuterated precursors, this method allows for late-stage functionalization. This approach not only preserves the complex stereochemistry and functional groups inherent to flavonoids but also drastically reduces the time and cost associated with sourcing specialized starting materials. By enabling the efficient production of deuterated analogs of widely used bioactive molecules, this technology supports the rapid iteration of drug candidates in preclinical studies, ultimately accelerating the path to clinical trials for cardiovascular, anti-inflammatory, and anticancer therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of deuterated flavonoids has been plagued by significant inefficiencies and economic barriers that hinder their widespread adoption in commercial drug manufacturing. Conventional routes often rely on the total synthesis of the flavonoid skeleton using expensive deuterated reagents at the very beginning of the synthetic sequence. This strategy is not only cost-prohibitive due to the high price of deuterated building blocks but also suffers from poor atom economy, as deuterium atoms can be lost during subsequent protection and deprotection steps. Furthermore, traditional methods frequently require harsh reaction conditions, such as strong acids or cryogenic temperatures, which can lead to the degradation of sensitive phenolic hydroxyl groups commonly found in flavonoid structures. These limitations result in low overall yields, complex purification processes, and a final product that may contain unacceptable levels of isotopic impurities, thereby compromising the reliability of metabolic studies.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct catalytic exchange strategy that fundamentally simplifies the production workflow while enhancing output quality. By mixing the native flavonoid compound directly with heavy water and an alkali catalyst, the method facilitates a robust exchange of hydrogen atoms for deuterium at specific active sites on the aromatic rings. This process operates effectively at temperatures above 90°C, and when augmented with a platinum carbon catalyst, can proceed efficiently between 110°C and 200°C. The innovation here is the dual-catalyst system involving sodium deuteroxide and platinum carbon, which activates the C-H bonds for exchange without necessitating the destruction of the molecular framework. This results in a remarkably simple operation process where the deuterated sodium salt is formed in situ and subsequently acidified to yield the final product, achieving yields of at least 40% and often exceeding 80% in optimized examples.
Mechanistic Insights into Base-Catalyzed H/D Exchange
The core chemical mechanism driving this transformation is a base-catalyzed electrophilic aromatic substitution where the deuteron from heavy water acts as the electrophile. In the presence of a strong base like sodium deuteroxide (NaOD), the phenolic hydroxyl groups on the flavonoid scaffold are deprotonated to form phenoxide ions, which significantly increase the electron density of the aromatic rings. This activation makes the ortho and para positions relative to the hydroxyl groups highly susceptible to attack by deuterium species. When platinum carbon is introduced into the reaction matrix, it serves as a heterogeneous catalyst that further lowers the activation energy for C-H bond cleavage, facilitating the exchange even at sterically hindered positions. The synergy between the homogeneous base catalyst and the heterogeneous metal catalyst ensures a high degree of deuteration across multiple sites, as evidenced by the formation of tetra- and pentadeuterated species in the experimental data.
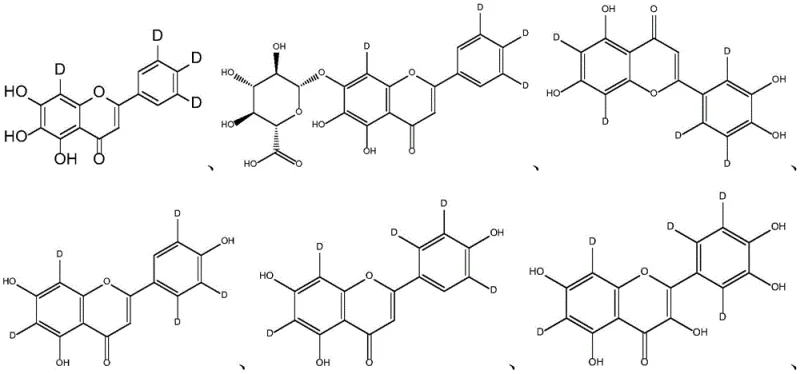
Understanding the regioselectivity of this exchange is crucial for R&D teams aiming to maximize metabolic stability. The patent data indicates that the exchange occurs preferentially at positions activated by electron-donating groups, such as the hydroxyls on the A and B rings of the flavonoid structure. For instance, in the case of baicalein, the reaction successfully targets the C-3', C-4', C-5', and C-8 positions, replacing labile protons with stable deuterium atoms. This specific labeling pattern is highly desirable because it protects the molecule from oxidative metabolism by cytochrome P450 enzymes, which typically target these electron-rich sites. The ability to control the degree of deuteration by adjusting the ratio of platinum carbon to the substrate—ranging from 1:3 to 1:12—provides a tunable parameter for chemists to optimize the isotopic purity versus cost trade-off for their specific application requirements.
Furthermore, the purification mechanism relies on the solubility differences between the deuterated product and the catalyst residues. After the acidification step using organic acids like formic or acetic acid, the deuterated flavonoid precipitates or can be extracted, while the platinum carbon remains as a solid residue that is easily removed by filtration. This simplicity in downstream processing is a major advantage over homogeneous catalytic systems that require complex chromatographic separation to remove metal traces. The use of inert gas environments, typically nitrogen, prevents the oxidation of the sensitive flavonoid core during the high-temperature exchange process, ensuring that the final product retains its antioxidant properties and structural integrity. This robust mechanistic pathway ensures that the resulting deuterated intermediates meet the stringent purity specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize Deuterated Flavonoids Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for producing deuterated flavonoids that can be adapted for both laboratory scale optimization and pilot plant operations. The process begins with the precise weighing of the flavonoid substrate, such as baicalein or luteolin, and mixing it with a calculated volume of heavy water and a stoichiometric amount of sodium deuteroxide solution. The reaction vessel is then purged with nitrogen to create an oxygen-free environment, which is critical for preventing side reactions and ensuring safety at elevated temperatures. Once the mixture is heated to the target temperature range of 110°C to 140°C and maintained for a duration of 5 to 13 hours, the hydrogen-deuterium exchange proceeds to completion, forming the soluble sodium salt of the deuterated compound.
- Mix the target flavonoid compound with alkali (NaOD/NaOH) and heavy water in an inert atmosphere.
- Heat the mixture to temperatures above 90°C (or 110-200°C with Pt/C catalyst) to facilitate hydrogen-deuterium exchange.
- Acidify the resulting sodium salt with organic acid, filter, and purify to obtain the high-purity deuterated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this deuteration technology presents a compelling value proposition centered on cost efficiency and supply reliability. The primary driver for cost reduction is the elimination of expensive deuterated starting materials; instead, the process utilizes commodity chemicals like heavy water and standard flavonoids which are abundantly available from natural extraction or established synthetic routes. This shift significantly lowers the raw material cost basis, making deuterated drugs more economically viable to develop. Additionally, the simplicity of the workup procedure, which involves basic filtration and drying rather than complex column chromatography, reduces solvent consumption and waste disposal costs. The high yields reported, often surpassing 70% and reaching up to 84% in optimized conditions, further enhance the economic feasibility by maximizing the output per batch and minimizing the need for reprocessing off-spec material.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing costly deuterated precursors with inexpensive heavy water and leveraging a reusable platinum carbon catalyst system. The operational simplicity allows for the use of standard stainless steel reactors without the need for specialized cryogenic or high-pressure equipment, thereby reducing capital expenditure and maintenance overheads. Moreover, the high atom economy of the exchange reaction ensures that the majority of the deuterium source is incorporated into the product, minimizing waste and maximizing the return on investment for every gram of heavy water purchased.
- Enhanced Supply Chain Reliability: By relying on widely available flavonoid feedstocks and common industrial reagents, this method mitigates the risk of supply chain disruptions often associated with niche custom synthesis. The robustness of the reaction conditions means that production can be scaled up rapidly to meet surging demand without lengthy process re-validation. The ability to produce a diverse range of deuterated flavonoids from a single platform technology also allows manufacturers to maintain a flexible inventory, quickly switching between products like deuterated quercetin or apigenin based on market needs without retooling the entire production line.
- Scalability and Environmental Compliance: The method is inherently scalable, having been demonstrated to work effectively with increased batch sizes while maintaining consistent yields and purity profiles. The use of nitrogen as a protective gas and the absence of toxic heavy metal catalysts in the final product simplify environmental compliance and waste treatment protocols. The solid platinum carbon catalyst can potentially be recovered and regenerated, further aligning the process with green chemistry principles and reducing the environmental footprint of the manufacturing operation, which is increasingly important for meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuterated flavonoid synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of using deuterated flavonoids in drug development?
A: Deuterated flavonoids exhibit enhanced metabolic stability due to the stronger carbon-deuterium bond compared to carbon-hydrogen bonds, potentially extending the half-life and improving the therapeutic index of the drug candidate.
Q: Can this deuteration method be scaled for industrial production?
A: Yes, the patent describes a simple process using readily available reagents like heavy water and platinum carbon, with yields reaching up to 84%, making it highly suitable for large-scale commercial manufacturing.
Q: Which flavonoid scaffolds are compatible with this synthesis route?
A: The method is versatile and has been successfully demonstrated on baicalein, baicalin, luteolin, apigenin, quercetin, kaempferol, isorhamnetin, hesperetin, puerarin, and daidzein.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Flavonoid Supplier
As the global demand for deuterated pharmaceuticals continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is essential for navigating the complexities of isotopic labeling. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and compliant. Our state-of-the-art facilities are equipped to handle the specific requirements of deuterium chemistry, including dedicated heavy water recovery systems and rigorous QC labs capable of verifying isotopic enrichment levels with high precision. We are committed to delivering high-purity deuterated flavonoid intermediates that meet the stringent purity specifications required by top-tier pharmaceutical companies worldwide.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this advanced deuteration technology into your drug development pipeline. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of how this method can reduce your overall COGS compared to traditional synthetic routes. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecule, helping you make informed decisions that accelerate your project timelines. Contact our technical procurement team today to discuss how we can support your supply chain with reliable, high-quality deuterated intermediates.
