Scalable Asymmetric Synthesis of Pyrrolidine Carboxylic Acids for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry's relentless pursuit of effective treatments for metabolic disorders has placed significant emphasis on the development of melanocortin-4 receptor (MC-4R) agonists. Patent CN1774419A discloses a groundbreaking methodology for the preparation of pyrrolidine carboxylic acid compounds, which serve as critical intermediates in the synthesis of these therapeutic agents. These compounds are pivotal for addressing conditions such as obesity, diabetes, and sexual dysfunction, representing a high-value segment within the fine chemical market. The disclosed process offers a robust alternative to traditional methods by integrating asymmetric synthesis techniques that ensure high stereochemical purity from the outset. By leveraging specific catalytic reductions and controlled cyclization strategies, this technology enables the production of chiral pyrrolidine derivatives with exceptional efficiency. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable supply chain for next-generation metabolic drugs. The ability to produce these intermediates without relying on wasteful resolution steps marks a significant advancement in process chemistry, aligning with modern demands for sustainability and cost-effectiveness in active pharmaceutical ingredient (API) manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral pyrrolidine acids relied heavily on the preparation of racemic mixtures followed by chiral high-performance liquid chromatography (HPLC) for separation. This traditional approach is inherently inefficient, as it theoretically discards fifty percent of the produced material corresponding to the unwanted enantiomer. The reliance on chiral chromatography introduces substantial operational costs, including expensive stationary phases, high solvent consumption, and complex equipment maintenance. Furthermore, scaling such resolution processes presents significant engineering challenges, often resulting in bottlenecks that delay clinical supply and commercial launch timelines. The environmental footprint of these methods is also considerable, generating large volumes of chemical waste that require specialized treatment. For procurement managers, the economic implications are severe, as the low overall yield drives up the cost of goods sold (COGS) and creates vulnerability in the supply chain. The inability to consistently produce high-purity intermediates without these cumbersome separation steps has long been a barrier to the widespread availability of MC-4R targeting therapeutics.
The Novel Approach
In stark contrast, the methodology outlined in CN1774419A employs a convergent asymmetric synthesis strategy that circumvents the need for chiral resolution entirely. By utilizing chiral catalysts such as (S)-CBS or (+)-DIP-Chloride during the initial reduction of haloketones, the process establishes the desired stereochemistry early in the synthetic sequence. This proactive approach ensures that the subsequent intermediates maintain high enantiomeric excess, ultimately delivering the final pyrrolidine carboxylic acid with a chiral purity exceeding 99.9% ee. The synthetic sequence is streamlined into five distinct steps, achieving an impressive overall yield of approximately 71%. This efficiency is further enhanced by the use of crystallization techniques for purification, which are far more scalable and cost-effective than chromatographic methods. The elimination of the resolution step not only doubles the theoretical yield compared to racemic synthesis but also drastically reduces solvent usage and waste generation. For supply chain heads, this translates to a more resilient manufacturing process capable of meeting large-scale commercial demands without the volatility associated with complex purification technologies.
Mechanistic Insights into Asymmetric Reduction and Cyclization
The core of this innovative synthesis lies in the precise control of stereochemistry during the reduction of alpha-halo ketones. The process initiates with the asymmetric reduction of a haloketone precursor using a borane reducing agent in the presence of a chiral catalyst, such as (S)-2-methyl-CBS-oxazaborolidine. This catalytic cycle facilitates the hydride transfer to the carbonyl group with high facial selectivity, generating a chiral halohydrin intermediate with exceptional enantiomeric purity. The reaction conditions are meticulously optimized, typically operating at temperatures between 30°C and 50°C in solvents like MTBE or toluene to ensure maximum catalyst turnover and selectivity. Following the reduction, the halohydrin undergoes base-mediated intramolecular substitution to form an epoxide intermediate, which retains the stereochemical information established in the first step. Subsequent ring opening of this epoxide with a primary amine, such as tert-butylamine, proceeds with inversion of configuration at the benzylic position, yielding a chiral aminoalcohol. This sequence is critical for setting the relative stereochemistry required for the final trans-pyrrolidine structure.
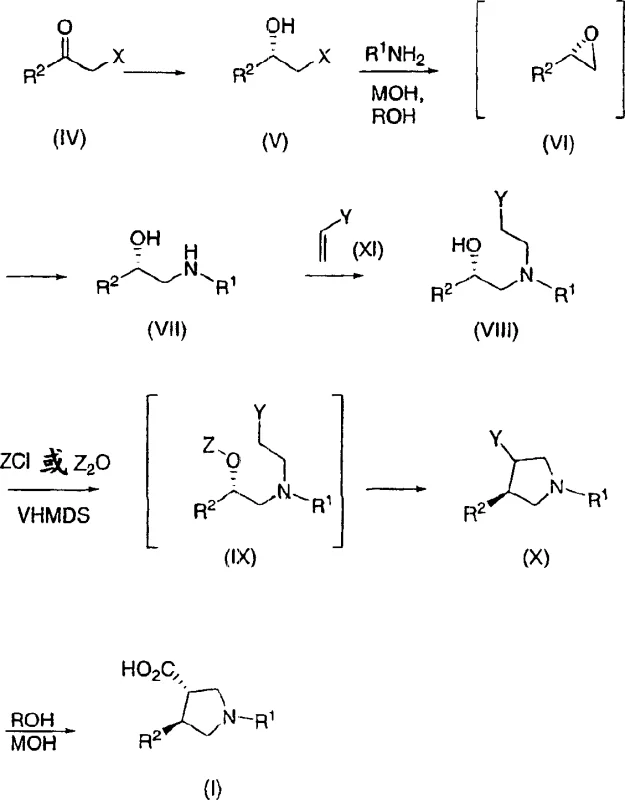
Following the formation of the chiral aminoalcohol, the synthesis proceeds through a conjugate addition reaction with an alpha,beta-unsaturated nitrile or ester, such as acrylonitrile. This Michael addition extends the carbon chain and introduces the functionality necessary for ring closure. The resulting tertiary amine intermediate is then subjected to activation, where the hydroxyl group is converted into a leaving group using reagents like chlorodiethyl phosphate or mesyl chloride. Treatment with a strong base, such as lithium hexamethyldisilazane (LiHMDS), triggers an intramolecular nucleophilic substitution, closing the five-membered pyrrolidine ring. This cyclization step produces a mixture of cis and trans diastereomers, which are subsequently subjected to kinetic hydrolysis and epimerization. The final transformation involves hydrolyzing the nitrile group to a carboxylic acid under basic conditions, coupled with thermodynamic equilibration to favor the more stable trans-isomer. This mechanistic pathway ensures that the final product is obtained as a single diastereomer with high optical purity, suitable for direct use in downstream drug synthesis.
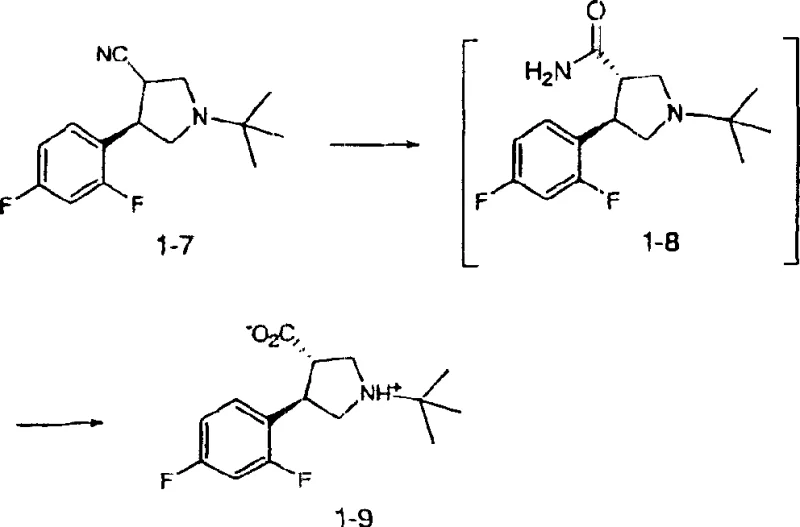
How to Synthesize Pyrrolidine Carboxylic Acid Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the asymmetric reduction and cyclization steps. The process begins with the preparation of the chiral halohydrin, where moisture control is paramount to prevent catalyst deactivation. The subsequent epoxide formation and ring opening should be monitored closely to ensure complete conversion before proceeding to the Michael addition. The cyclization step, driven by the activation of the alcohol and subsequent base treatment, requires low-temperature control to minimize side reactions and optimize the cis/trans ratio. Finally, the hydrolysis and epimerization step is conducted under reflux conditions with aqueous base, followed by pH adjustment to the isoelectric point to isolate the product as a zwitterion. Detailed standardized synthetic steps are provided in the guide below to ensure reproducibility and quality control across different manufacturing sites.
- Asymmetric reduction of haloketones using CBS catalyst or DIP-Chloride to form chiral halohydrins with high enantiomeric excess.
- Base-mediated epoxide formation followed by ring opening with primary amines to generate chiral aminoalcohols.
- Conjugate addition with acrylonitrile, followed by activation and intramolecular cyclization to form the pyrrolidine ring.
- Kinetic hydrolysis and epimerization of the nitrile group to yield the final trans-pyrrolidine carboxylic acid zwitterion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement and supply chain management within the pharmaceutical sector. The primary benefit stems from the significant reduction in manufacturing costs driven by the elimination of chiral chromatography. By avoiding the need for expensive chiral columns and the associated solvent-intensive processes, manufacturers can achieve substantial cost savings per kilogram of produced intermediate. This efficiency is compounded by the high overall yield of the process, which maximizes the output from raw materials and reduces the cost of goods sold. For procurement managers, this means a more competitive pricing structure for the final API, allowing for better margin management in an increasingly price-sensitive market. Furthermore, the reliance on crystallization for purification simplifies the operational workflow, reducing the need for specialized equipment and skilled labor associated with complex chromatographic separations.
- Cost Reduction in Manufacturing: The process achieves cost optimization by removing the most expensive unit operation in chiral synthesis, which is preparative chiral HPLC. This elimination not only saves on direct material costs for stationary phases but also reduces utility consumption related to solvent recovery and waste disposal. The high yield of 71% across five steps ensures that raw material utilization is maximized, further driving down the variable costs associated with production. Additionally, the use of commercially available reagents like borane complexes and common bases ensures that input costs remain stable and predictable. These factors collectively contribute to a leaner manufacturing model that enhances profitability without compromising on the quality or purity of the intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly improved due to the robustness of the chemical transformations involved. The reagents used, such as sodium hydroxide, methanol, and acrylonitrile, are commodity chemicals with stable global supply networks, reducing the risk of shortages. The process does not rely on bespoke or hard-to-source catalysts that could become bottlenecks. Moreover, the ability to purify intermediates via crystallization rather than chromatography makes the process more tolerant to scale-up variations, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturing, thereby mitigating the risk of drug shortages and ensuring timely market availability for critical metabolic therapies.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for scalability, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial plant scales. The avoidance of chromatographic steps simplifies the engineering requirements for large-scale reactors, allowing for higher throughput in standard vessel configurations. From an environmental standpoint, the process aligns with green chemistry principles by reducing solvent waste and eliminating the disposal challenges associated with spent chiral stationary phases. The high atom economy of the asymmetric reduction and the efficient recycling of solvents like heptane and ethanol further minimize the environmental footprint. This compliance with stringent environmental regulations facilitates smoother regulatory approvals and enhances the sustainability profile of the manufacturing operation, which is increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of pyrrolidine carboxylic acids using this patented methodology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the process's capability to deliver high-purity materials while maintaining operational efficiency and cost-effectiveness.
Q: How does this process avoid chiral chromatography?
A: The process utilizes asymmetric catalysis (CBS or DIP-Cl) and crystallization-induced diastereomeric enrichment to achieve >99.9% ee without requiring expensive chiral HPLC separation.
Q: What is the overall yield of the synthetic route?
A: The patented synthetic sequence comprises five steps and achieves an overall yield of approximately 71%, which is significantly higher than traditional racemic resolution methods.
Q: Is this process suitable for large-scale production?
A: Yes, the method avoids chromatographic purification and uses robust reagents like borane complexes and aqueous bases, making it highly amenable to commercial scale-up in standard reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolidine Carboxylic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We understand the critical importance of stringent purity specifications and rigorous QC labs in the pharmaceutical industry, and our facilities are equipped to handle the demanding requirements of chiral intermediate manufacturing. By partnering with us, you gain access to a robust supply chain capable of delivering high-quality pyrrolidine carboxylic acids that meet the exacting standards required for MC-4R agonist development.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these critical intermediates. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this efficient synthetic route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project's unique requirements. Let us help you accelerate your development timeline and secure a competitive advantage in the metabolic disease therapeutic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
