Optimizing Sultopride Hydrochloride Production: A Technical Analysis of the Novel Esterification Route
Introduction to Advanced Sultopride Hydrochloride Manufacturing
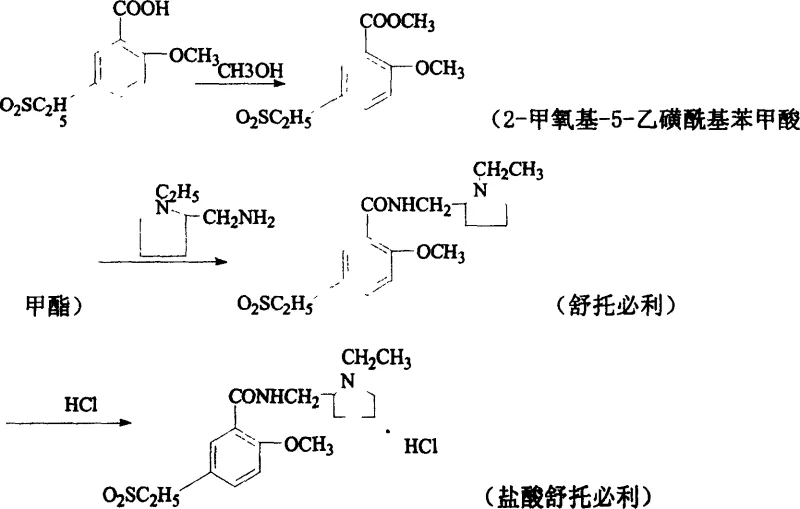
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for antipsychotic agents like Sultopride Hydrochloride. Patent CN1315804C introduces a transformative methodology that redefines the production landscape for this critical active pharmaceutical ingredient (API) intermediate. By shifting away from unstable acid chloride intermediates toward a stable esterification-amidation sequence, this technology addresses long-standing inefficiencies in yield and operational safety. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, understanding the mechanistic superiority of this route is essential for securing long-term supply chain stability. The process leverages common, recyclable solvents and mild reaction conditions to achieve a total yield improvement that fundamentally alters the cost structure of manufacturing.
This technical insight report dissects the proprietary steps outlined in the patent, highlighting how the conversion of 2-methoxy-5-ethanesulfonylbenzoic acid into its methyl ester serves as the pivotal activation step. Unlike traditional methods that rely on hazardous chlorinating agents, this approach utilizes Fischer esterification followed by a high-temperature amidation in polyol solvents. The result is a streamlined workflow that minimizes waste generation while maximizing the recovery of valuable starting materials. As we delve deeper into the specific reaction parameters, it becomes evident that this methodology represents a significant leap forward in cost reduction in API manufacturing, offering a scalable solution for global demand.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sultopride Hydrochloride has been plagued by operational complexities that hinder large-scale production efficiency. Traditional literature describes two primary routes, both of which present substantial drawbacks for industrial application. The first method involves the chlorination of the benzoic acid precursor to form an acid chloride, followed by condensation. This pathway is inherently risky due to the instability of the chlorinated intermediate, which complicates storage and handling while often resulting in inconsistent reaction outcomes. Furthermore, the second conventional method attempts direct condensation between the free acid and the amine component. While avoiding chlorination, this route necessitates the use of excessive amounts of pyridine and chloroform, solvents that are not only toxic but also notoriously difficult to recover and recycle efficiently. Consequently, these legacy processes typically suffer from dismal overall yields, reported as low as 44.9%, rendering them economically unviable for modern commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel synthesis route detailed in the patent employs a strategic esterification pre-step that fundamentally changes the reactivity profile of the substrate. By converting 2-methoxy-5-ethanesulfonylbenzoic acid into its corresponding methyl ester, the process creates a highly electrophilic center that readily undergoes nucleophilic attack by N-ethyl-2-aminomethyltetrahydropyrrolidine. This transformation eliminates the need for hazardous chlorinating reagents and avoids the use of problematic solvents like chloroform. Instead, the amidation step utilizes high-boiling polyols such as ethylene glycol or glycerin, which facilitate the reaction at elevated temperatures without requiring pressurized equipment. The integration of these steps allows for a continuous operation where the condensation and subsequent acidification can be managed with greater fluidity. This innovation not only simplifies the post-treatment workflow but also dramatically enhances the overall throughput, positioning this method as the preferred choice for high-purity pharmaceutical intermediates production.
Mechanistic Insights into Esterification and Polyol-Mediated Amidation
The core of this synthetic breakthrough lies in the precise control of the esterification and subsequent amidation mechanisms. The initial step involves a classic acid-catalyzed Fischer esterification where concentrated sulfuric acid acts as both a catalyst and a dehydrating agent. By maintaining the reaction temperature below 30°C during the addition of sulfuric acid to methanol, the process prevents localized overheating and potential side reactions. Once the 2-methoxy-5-ethanesulfonylbenzoic acid is introduced, the mixture is heated to reflux for approximately 4 hours. This thermal energy drives the equilibrium toward the formation of the methyl ester by continuously removing water, ensuring a high conversion rate that sets the stage for the subsequent coupling reaction. The isolation of the white crystalline ester intermediate provides a pure starting material for the next phase, eliminating impurities that could otherwise propagate through the synthesis.
Following esterification, the mechanism shifts to a nucleophilic acyl substitution facilitated by the unique properties of the solvent system. When the methyl ester is reacted with N-ethyl-2-aminomethyltetrahydropyrrolidine in ethylene glycol or glycerin, the high boiling point of the solvent allows the reaction to proceed at temperatures between 70°C and 110°C. This thermal environment provides the necessary activation energy for the amine nitrogen to attack the carbonyl carbon of the ester, displacing the methoxy group. The use of polyols is particularly advantageous as they stabilize the transition state and solubilize both organic reactants effectively. Upon completion, the reaction mixture is cooled, and the free base is precipitated by acidification with hydrochloric acid to pH 1. This final protonation step ensures the formation of the stable hydrochloride salt, which is then purified via ethanol recrystallization to achieve the stringent purity specifications required for psychiatric medication.
How to Synthesize Sultopride Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding temperature control and reagent stoichiometry to ensure reproducibility. The process is designed to be modular, allowing for the esterification and amidation steps to be performed in sequence with minimal intermediate handling. Detailed standard operating procedures regarding specific molar ratios, stirring rates, and cooling profiles are critical for maintaining the high yield profile observed in the patent examples. Manufacturers aiming to adopt this technology should focus on the efficient recovery of the methanol and polyol solvents to maximize the economic benefits of the process.
- Perform acid-catalyzed esterification of 2-methoxy-5-ethanesulfonylbenzoic acid with methanol under reflux to form the methyl ester intermediate.
- Conduct nucleophilic substitution by reacting the methyl ester with N-ethyl-2-aminomethyltetrahydropyrrolidine in ethylene glycol or glycerin at elevated temperatures.
- Acidify the resulting base with hydrochloric acid to pH 1, followed by recrystallization from ethanol to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible benefits that extend beyond simple yield improvements. The elimination of unstable acid chloride intermediates reduces the risk of batch failures and safety incidents, thereby enhancing the reliability of supply. Furthermore, the ability to recycle solvents such as methanol, ethylene glycol, and glycerin directly impacts the variable costs associated with production. By recovering mother liquors through distillation and reusing them in subsequent batches, manufacturers can significantly lower their raw material consumption. This closed-loop approach to solvent management not only drives down costs but also aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The robustness of this method ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the substantial increase in overall yield compared to legacy methods. By moving from a yield of roughly 44.9% to approximately 80%, the effective cost per kilogram of the final API is drastically reduced. This efficiency gain is compounded by the removal of expensive and hazardous reagents like thionyl chloride or oxalyl chloride, which are required in the traditional chlorination route. Additionally, the replacement of chloroform and pyridine with cheaper, recyclable polyols further decreases the operational expenditure. These cumulative savings allow for a more competitive pricing structure in the global market, making the final medication more accessible while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized or hazardous reagents that may face regulatory restrictions or availability fluctuations. This new method utilizes commodity chemicals such as methanol, sulfuric acid, and ethylene glycol, which are widely available from multiple global sources. The stability of the methyl ester intermediate also allows for flexible production scheduling, as it can be stored safely if needed, unlike the reactive acid chlorides used in older processes. This flexibility empowers supply chain planners to buffer against market volatility and ensure consistent delivery schedules to downstream API formulators. The simplified workflow also reduces the dependency on highly specialized labor, further stabilizing the production timeline.
- Scalability and Environmental Compliance: From an environmental perspective, this process represents a greener alternative to traditional synthesis. The reduction in hazardous waste generation is significant, as the toxic byproducts associated with chlorination are entirely avoided. The solvents used are not only less toxic but are also easily separable and reusable, minimizing the volume of effluent that requires treatment. This alignment with green chemistry principles facilitates easier permitting and regulatory approval in jurisdictions with strict environmental oversight. Moreover, the mild reaction conditions and lack of high-pressure requirements make the process inherently safer and easier to scale from pilot plant to multi-ton commercial production, ensuring that capacity can be expanded rapidly to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: Why is the esterification route preferred over direct condensation for Sultopride?
A: Direct condensation often suffers from low yields (around 44.9%) and requires difficult-to-recycle solvents like pyridine and chloroform. The esterification route activates the carboxylic acid more effectively, leading to significantly higher overall yields and easier purification.
Q: What are the environmental advantages of this new synthesis method?
A: This method utilizes recyclable solvents such as methanol, ethylene glycol, and glycerin. The mother liquor from the filtration steps can be distilled and reused, drastically reducing hazardous waste generation compared to traditional chlorination methods.
Q: What purity levels can be achieved with this process?
A: Analytical data from the patent indicates that the final product achieves a content purity of over 99% as determined by HPLC, with melting points and spectral data consistent with imported reference standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sultopride Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of psychiatric medications depends on the availability of high-quality intermediates produced via efficient routes. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial reality is seamless. We are committed to maintaining stringent purity specifications and utilizing rigorous QC labs to verify that every batch of Sultopride Hydrochloride meets the highest international standards. Our infrastructure is designed to support the complex solvent recovery and temperature control systems required by this advanced synthesis method, guaranteeing consistency and reliability for our partners.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this superior synthesis technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized process can enhance your supply chain resilience and profitability. Let us help you secure a sustainable future for your antipsychotic drug portfolio with our proven expertise in fine chemical manufacturing.
