Advanced Pd-Boron Co-Catalysis for Scalable Chiral Tertiary Alcohol and Ether Production
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex chiral architectures, particularly those found in life-saving antifungal medications. Patent CN107382644B introduces a groundbreaking preparation method for chiral tertiary alcohol and tertiary ether compounds, addressing long-standing challenges in asymmetric synthesis. This technology leverages a sophisticated palladium and boron co-catalytic system to transform racemic 4-substituted-4-vinyl-1,3-dioxolane-2-ketone compounds into high-value chiral building blocks. Unlike conventional routes that often struggle with regioselectivity or require harsh conditions, this invention utilizes stable raw materials and operates under mild reaction parameters, making it highly attractive for industrial adoption. The core innovation lies in the direct reaction of these cyclic carbonate precursors with water or alcohols, facilitated by the synergistic action of a palladium complex and a boron compound, yielding products with exceptional enantioselectivity. 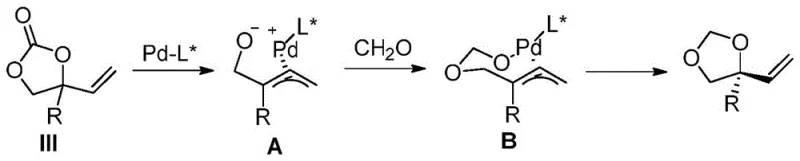
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral tertiary alcohols and ethers has been a formidable obstacle in organic synthesis, primarily because traditional strategies rely heavily on the asymmetric addition of carbon nucleophiles to ketones. This classical approach typically necessitates the activation of the ketone carbonyl to enhance its electrophilicity, and crucially, it demands a significant steric difference between the two substituents on the carbonyl group to achieve high stereoselectivity. When substituents are similar in size, the stereochemical outcome is often poor, limiting the scope of accessible molecules. Furthermore, alternative methods involving asymmetric dihydroxylation or epoxidation of 1,1-disubstituted olefins also suffer from similar steric constraints, requiring bulky groups to differentiate the faces of the double bond effectively. Previous attempts by the applicants, such as those described in patent CN103788056A, utilized formaldehyde as an electrophile but required a multi-step sequence involving the formation of a five-membered ring intermediate before hydrolysis, adding complexity and cost to the manufacturing process. Additionally, methods employing vinyl oxiranes, while effective, utilize unstable raw materials that pose significant safety and handling risks during large-scale production, hindering their practical application in commercial supply chains.
The Novel Approach
The methodology disclosed in CN107382644B represents a paradigm shift by employing racemic 4-substituted-4-vinyl-1,3-dioxolane-2-one compounds as stable and readily available starting materials. This novel route bypasses the need for unstable epoxides or sterically demanding ketones, instead utilizing a direct asymmetric hydroxylation or etherification strategy catalyzed by the unique combination of palladium and boron. The reaction proceeds smoothly in common organic solvents such as tetrahydrofuran or toluene at moderate temperatures ranging from 0°C to 60°C, typically optimized around 40°C. This mild condition profile not only enhances safety but also significantly reduces energy consumption compared to cryogenic processes often required for high-fidelity asymmetric transformations. By directly introducing water or alcohol nucleophiles to the allylic position under palladium catalysis, the method constructs the quaternary carbon chiral center with remarkable precision. The versatility of this approach allows for the synthesis of a wide array of chiral tertiary alcohols and ethers, serving as multifunctional molecular building blocks that can be flexibly transformed into various downstream pharmaceutical intermediates, thereby streamlining the synthesis of complex drug candidates.
Mechanistic Insights into Pd-Boron Co-Catalyzed Asymmetric Allylic Substitution
The success of this transformation hinges on the intricate interplay between the palladium catalyst and the chiral ligand environment, which dictates the stereochemical outcome of the reaction. The mechanism initiates with the oxidative addition of the palladium source to the vinyl dioxolane substrate, generating a cationic pi-allyl palladium intermediate. In traditional allylic substitution reactions, controlling the regioselectivity of nucleophilic attack on this intermediate is notoriously difficult, often leading to mixtures of branched and linear products. However, the introduction of the boron compound acts as a crucial co-catalyst, likely functioning to activate the nucleophile or stabilize the transition state, thereby enforcing high regioselectivity towards the desired tertiary center. The chiral information is transferred from the specialized phosphine ligands to the substrate through the rigid coordination sphere of the palladium complex. 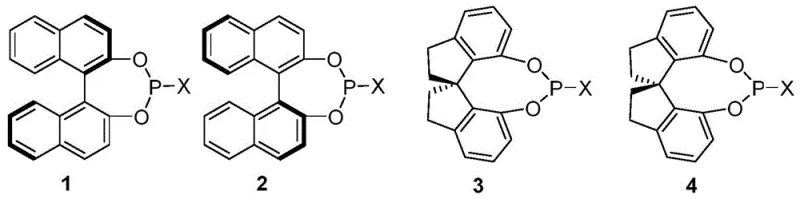
Experimental data within the patent highlights the critical role of ligand architecture, where specific binaphthyl-based phosphine ligands bearing dialkylamino groups demonstrate superior performance in terms of both yield and enantiomeric excess (ee). For instance, ligands such as (R)-3a have been shown to deliver products with up to 96% yield and 95% ee in model reactions, underscoring the importance of fine-tuning the steric and electronic properties of the ligand backbone. The boron component, whether it be triethylboron or phenylboronic acid, appears to facilitate the ionization of the leaving group or modulate the Lewis acidity of the system, ensuring that the nucleophilic attack by water or alcohol occurs exclusively at the more substituted carbon atom. This level of control effectively suppresses the formation of undesired regioisomers and racemic byproducts, resulting in a clean impurity profile that is essential for pharmaceutical grade intermediates. The robustness of this catalytic cycle allows it to tolerate a diverse range of functional groups on the aromatic ring of the substrate, including halogens, alkoxy groups, and heterocycles, further validating its utility for synthesizing complex drug scaffolds.
How to Synthesize Chiral Tertiary Alcohols Efficiently
Implementing this advanced catalytic protocol requires careful attention to reagent quality and reaction conditions to maximize the potential of the Pd-Boron system. The process is designed to be operationally simple, avoiding the need for exotic equipment or extreme temperatures, which facilitates its translation from laboratory scale to commercial manufacturing. Operators should ensure the use of dry solvents and an inert atmosphere to protect the sensitive palladium catalyst from deactivation by oxygen or moisture prior to the intentional addition of the nucleophile. The molar ratios of the catalyst components are critical, with typical loadings of the palladium source around 2.5 mol% and the chiral ligand at 10 mol%, while the boron reagent is used in slight excess to drive the reaction to completion. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction mixture by combining the racemic 4-substituted-4-vinyl-1,3-dioxolane-2-one substrate with a palladium source such as Pd2(dba)3CHCl3 and a chiral phosphine ligand in an organic solvent like THF or toluene.
- Add the boron compound catalyst, such as triethylboron or phenylboronic acid, along with the nucleophile (water for alcohols or alcohol for ethers) to the reaction vessel under inert atmosphere.
- Maintain the reaction temperature between 0°C and 60°C, typically around 40°C, for approximately 16 hours, then isolate the chiral product via column chromatography after solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers compelling strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediates manufacturing achieved through the use of stable, bulk chemical raw materials. Unlike previous methods that relied on unstable vinyl oxiranes or required multi-step sequences with formaldehyde, this single-step direct functionalization minimizes unit operations, reducing labor, energy, and equipment costs significantly. The mild reaction conditions further contribute to operational expenditure savings by eliminating the need for expensive cryogenic cooling systems, allowing the process to run in standard glass-lined or stainless steel reactors commonly available in multipurpose facilities. Moreover, the high catalytic activity means that precious metal loading can be optimized, reducing the overall consumption of palladium, which is a major cost driver in fine chemical synthesis.
- Cost Reduction in Manufacturing: The elimination of unstable reagents and the consolidation of multiple synthetic steps into a single catalytic transformation drastically simplifies the production workflow. This streamlining reduces the accumulation of intermediate inventory and lowers the working capital tied up in work-in-progress materials. Furthermore, the high atom economy of the direct hydroxylation and etherification reactions minimizes waste generation, leading to lower disposal costs and a smaller environmental footprint. The ability to achieve high yields and enantioselectivity in one pot also reduces the need for costly and time-consuming purification steps such as recrystallization or chiral resolution, directly impacting the bottom line by improving overall process mass intensity.
- Enhanced Supply Chain Reliability: Sourcing stable raw materials is a critical factor in maintaining supply continuity, and the 4-substituted-4-vinyl-1,3-dioxolane-2-one precursors used in this method are robust and easy to store compared to sensitive epoxides. This stability mitigates the risk of raw material degradation during transport and storage, ensuring consistent quality input for the manufacturing process. The broad substrate scope of the reaction means that a single platform technology can be adapted to produce a variety of different chiral intermediates, providing flexibility to respond to changing market demands for different antifungal agents without requiring entirely new process development campaigns. This versatility strengthens the supply chain resilience against disruptions specific to single-product lines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on homogeneous catalysis in common solvents, which behaves predictably upon scale-up from kilogram to tonne quantities. The mild thermal profile reduces the risk of thermal runaway incidents, enhancing plant safety and simplifying regulatory compliance regarding process safety management. Additionally, the use of water or simple alcohols as nucleophiles generates benign byproducts, aligning with green chemistry principles and facilitating easier waste treatment. This environmental compatibility is increasingly important for meeting stringent global regulatory standards and corporate sustainability goals, making the technology future-proof for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pd-Boron co-catalyzed synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent documentation to provide clarity on its capabilities and limitations. Understanding these aspects is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines.
Q: What are the advantages of Pd-Boron co-catalysis over traditional ketone addition methods?
A: Traditional methods often require activating ketone carbonyls and rely on large steric differences between substituents for selectivity. The Pd-Boron co-catalysis described in CN107382644B utilizes stable cyclic carbonate precursors and achieves high regioselectivity and enantioselectivity under mild conditions without these steric constraints.
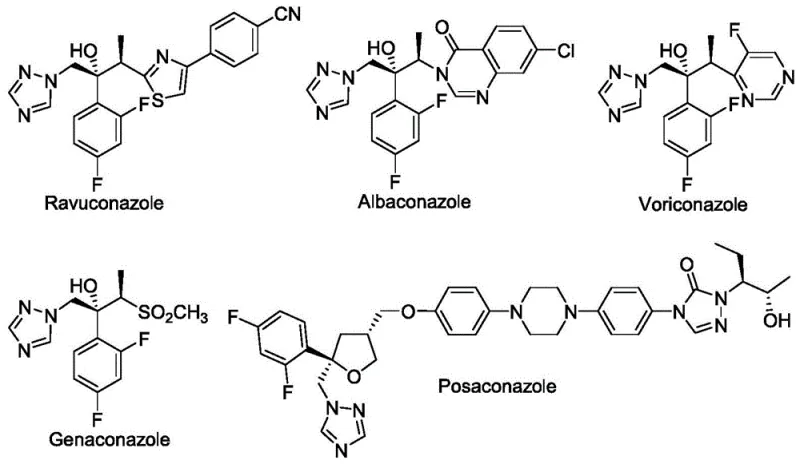
Q: Can this method be used for synthesizing Posaconazole intermediates?
A: Yes, the patent explicitly demonstrates the utility of the resulting chiral tertiary ether compounds as key intermediates for preparing antifungal drugs like Posaconazole, Ravuconazole, and Voriconazole, offering a streamlined route to these complex molecular scaffolds.
Q: What types of boron compounds are compatible with this catalytic system?
A: The process shows broad compatibility with various boron reagents, including trialkylboranes like triethylboron (BEt3) and arylboronic acids like phenylboronic acid (PhB(OH)2), allowing for flexibility in optimizing yield and enantiomeric excess for specific substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tertiary Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Pd-Boron co-catalysis technology described in CN107382644B for the production of high-value antifungal intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch of chiral tertiary alcohol or ether meets the exacting standards required for clinical and commercial pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this catalytic system for your specific needs, delivering cost-effective and reliable supply solutions.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can enhance your supply chain efficiency and product quality. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined process. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules, ensuring a partnership built on transparency, scientific excellence, and mutual growth in the competitive landscape of chiral pharmaceutical intermediates.